Scalable Synthesis of 4-Piperazine-3-Trifluoromethylaniline Hydrochloride for Global Pharma Supply Chains
Scalable Synthesis of 4-Piperazine-3-Trifluoromethylaniline Hydrochloride for Global Pharma Supply Chains
The pharmaceutical industry constantly demands more efficient, safer, and cost-effective pathways for synthesizing complex heterocyclic intermediates, particularly those containing trifluoromethyl groups which are ubiquitous in modern drug design. Patent CN102731437A discloses a robust preparation method for 4-piperazine-3-trifluoromethylaniline hydrochloride, a critical building block in the development of various bioactive molecules. This technology represents a significant advancement over traditional methods by optimizing reaction conditions to enhance yield and operational simplicity. By utilizing a strategic sequence of nucleophilic aromatic substitution, catalytic hydrogenation, and controlled deprotection, the process minimizes impurity formation and maximizes throughput. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The following analysis details the technical merits and commercial viability of this synthesis, highlighting its potential to reduce manufacturing costs and improve supply continuity for global partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted piperazine anilines has been plagued by challenges related to regioselectivity and the stability of intermediates under harsh reaction conditions. Conventional routes often require extreme temperatures or aggressive reagents that can lead to the degradation of the trifluoromethyl group or the formation of difficult-to-remove byproducts. Furthermore, traditional protection and deprotection strategies sometimes involve multiple tedious purification steps, such as column chromatography, which are impractical for large-scale industrial applications. The use of unstable intermediates can also pose significant safety risks during scale-up, leading to inconsistent batch quality and extended lead times. These inefficiencies not only drive up the cost of goods sold but also create bottlenecks in the supply chain, making it difficult for manufacturers to meet the rigorous demands of downstream API production. Consequently, there is a pressing need for a streamlined approach that balances chemical efficiency with operational safety.
The Novel Approach
The methodology outlined in CN102731437A introduces a refined synthetic pathway that addresses these historical pain points through careful optimization of reaction parameters and reagent selection. The process initiates with the nucleophilic substitution of 4-nitro-2-trifluoromethyl-fluorobenzene with tert-butoxycarbonyl-piperazine under mild alkaline conditions, ensuring high conversion rates without compromising the integrity of the sensitive nitro group. 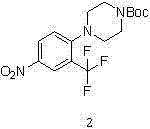 . This is followed by a highly selective catalytic hydrogenation step that reduces the nitro functionality to an amine while leaving the Boc protecting group intact. The final deprotection is achieved using hydrogen chloride in a controlled manner, yielding the target hydrochloride salt with exceptional purity. This stepwise approach eliminates the need for complex purification techniques, thereby significantly simplifying the overall workflow and enhancing the feasibility of commercial scale-up for complex pharmaceutical intermediates.
. This is followed by a highly selective catalytic hydrogenation step that reduces the nitro functionality to an amine while leaving the Boc protecting group intact. The final deprotection is achieved using hydrogen chloride in a controlled manner, yielding the target hydrochloride salt with exceptional purity. This stepwise approach eliminates the need for complex purification techniques, thereby significantly simplifying the overall workflow and enhancing the feasibility of commercial scale-up for complex pharmaceutical intermediates.
Mechanistic Insights into Catalytic Hydrogenation and Deprotection
The core of this synthesis lies in the precise control of the catalytic hydrogenation step, which transforms the nitro-intermediate into the corresponding aniline derivative. Using 10% Palladium on Carbon (Pd/C) as the catalyst, the reaction proceeds efficiently in methanol solvent under a hydrogen pressure of 10 to 20 atmospheres. The choice of methanol is critical, as it provides excellent solubility for the substrate while facilitating the transfer of hydrogen to the catalyst surface. Maintaining the temperature between 50 and 60 degrees Celsius is vital; this range is high enough to overcome the activation energy barrier for nitro reduction but low enough to prevent the premature cleavage of the Boc group or hydrogenolysis of the carbon-fluorine bonds. 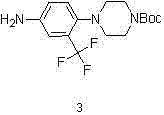 . This selectivity ensures that the piperazine ring remains protected, preventing potential side reactions such as intermolecular coupling or polymerization that could occur if the free amine were generated prematurely. The result is a clean conversion that minimizes the generation of colored impurities often associated with nitro reductions.
. This selectivity ensures that the piperazine ring remains protected, preventing potential side reactions such as intermolecular coupling or polymerization that could occur if the free amine were generated prematurely. The result is a clean conversion that minimizes the generation of colored impurities often associated with nitro reductions.
Following the reduction, the removal of the tert-butoxycarbonyl (Boc) protecting group is executed with high precision to yield the final hydrochloride salt. The process involves dissolving the amino-intermediate in ethyl acetate and cooling the solution to approximately 0 degrees Celsius before introducing excess hydrogen chloride gas. This low-temperature condition is crucial for controlling the exothermic nature of the deprotection reaction and ensuring the formation of a well-defined crystalline solid. The use of ethyl acetate as the solvent facilitates the precipitation of the product, allowing for easy isolation via filtration. This mechanism effectively suppresses the formation of oligomeric byproducts that can arise from the reactivity of the free piperazine amine. By strictly controlling the stoichiometry and temperature during this final step, the process guarantees a product with a consistent impurity profile, which is paramount for meeting the stringent quality specifications required by regulatory bodies for pharmaceutical raw materials.
How to Synthesize 4-Piperazine-3-Trifluoromethylaniline Hydrochloride Efficiently
To implement this synthesis effectively, operators must adhere to strict procedural guidelines regarding solvent quality and reaction monitoring. The initial substitution reaction benefits from the use of anhydrous DMF and high-purity potassium carbonate to drive the equilibrium towards the desired product. Subsequent hydrogenation requires careful monitoring of hydrogen uptake to determine the reaction endpoint accurately, preventing over-reduction or catalyst poisoning. The final deprotection step demands precise temperature control to ensure the product precipitates in a filterable form. Detailed standardized operating procedures are essential to replicate the high yields reported in the patent data consistently.
- React 4-nitro-2-trifluoromethyl-fluorobenzene with tert-butoxycarbonyl-piperazine in DMF using potassium carbonate as a base at room temperature.
- Perform catalytic hydrogenation of the nitro-intermediate using 10% Palladium on Carbon (Pd/C) in methanol under 10-20 atm pressure at 50-60°C.
- Remove the Boc protecting group by treating the amino-intermediate with excess hydrogen chloride in ethyl acetate at 0°C to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The reliance on readily available starting materials, such as 4-nitro-2-trifluoromethyl-fluorobenzene and Boc-piperazine, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the use of common industrial solvents like DMF, methanol, and ethyl acetate simplifies solvent recovery and recycling processes, contributing to a more sustainable and cost-efficient manufacturing footprint. The elimination of complex purification steps, such as column chromatography, drastically reduces processing time and labor costs, enabling faster turnaround times for custom synthesis orders. These factors collectively position this route as a highly attractive option for cost reduction in pharmaceutical intermediates manufacturing, providing a competitive edge in a price-sensitive market.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived primarily from the high atom economy and the avoidance of expensive reagents or specialized equipment. By utilizing a heterogeneous catalyst like Pd/C, which can potentially be recovered and reused, the process minimizes the consumption of precious metals. Additionally, the straightforward workup procedures, involving simple filtration and concentration, reduce the energy consumption associated with distillation and drying. The high yield obtained at each step means less raw material is wasted, leading to a significant decrease in the overall cost per kilogram of the final active intermediate. This efficiency allows suppliers to offer more competitive pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the robustness of the reaction conditions, which are tolerant to minor variations in input quality. The use of stable intermediates reduces the need for cryogenic storage or expedited shipping, lowering logistics costs and risks. Since the process does not rely on exotic or single-source reagents, procurement teams can diversify their supplier base for raw materials, reducing dependency risks. The scalability of the method ensures that production can be ramped up quickly to meet sudden spikes in demand, ensuring continuous availability of this critical building block for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in modern chemical plants. The waste streams generated are primarily aqueous and organic solvents that can be treated using conventional effluent treatment protocols, ensuring compliance with increasingly strict environmental regulations. The absence of heavy metal contaminants in the final product, due to the efficient filtration of the Pd/C catalyst, simplifies the regulatory filing process for drug manufacturers. This environmental compatibility not only reduces disposal costs but also aligns with the sustainability goals of major pharmaceutical companies, making it a preferred choice for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-piperazine-3-trifluoromethylaniline hydrochloride. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation step utilizes 10% Pd/C catalyst in methanol solvent. Critical parameters include maintaining hydrogen pressure between 10 to 20 atmospheres and controlling the temperature between 50 to 60 degrees Celsius to ensure complete reduction of the nitro group without affecting other sensitive functionalities.
Q: Why is the Boc protection group utilized in this synthesis route?
A: The tert-butoxycarbonyl (Boc) group protects the piperazine nitrogen during the nucleophilic substitution and hydrogenation steps. This prevents unwanted side reactions, such as polymerization or over-alkylation, ensuring high regioselectivity and simplifying the purification of the intermediate before the final deprotection step.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It employs common industrial solvents like DMF, methanol, and ethyl acetate, and uses standard unit operations such as filtration and crystallization. The mild reaction temperatures and robust catalyst system facilitate safe amplification from kilogram to multi-ton production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Piperazine-3-Trifluoromethylaniline Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver this high-value intermediate with unmatched consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorinated chemistry and catalytic hydrogenation, ensuring that every batch meets stringent purity specifications. We understand that in the pharmaceutical sector, the reliability of your supply chain is as critical as the quality of the molecule itself. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every shipment, providing you with the confidence needed to advance your clinical or commercial programs without interruption.
We invite you to collaborate with us to optimize your sourcing strategy for this essential building block. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and delivery schedules. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-effective supply of 4-piperazine-3-trifluoromethylaniline hydrochloride, empowering your organization to focus on innovation while we manage the complexities of chemical manufacturing.
