Scalable One-Pot Synthesis of 4-Amino-5-Aryl-1,2,4-Triazole-3-Thione Derivatives for Global Markets
Scalable One-Pot Synthesis of 4-Amino-5-Aryl-1,2,4-Triazole-3-Thione Derivatives for Global Markets
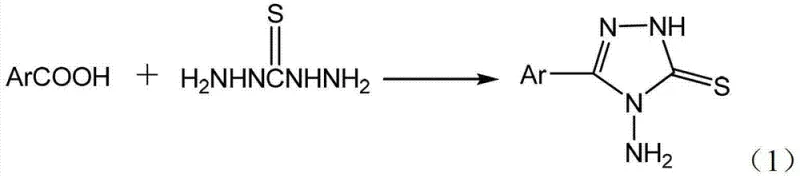
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A significant breakthrough in this domain is documented in patent CN103130731A, which details a novel method for preparing 4-amino-5-aryl-1,2,4-triazole-3-thione derivatives. These compounds serve as critical building blocks in the synthesis of various bioactive molecules, including antibiotics, antimicrobials, and anticancer agents, making their reliable production essential for the pharmaceutical and agrochemical industries. The disclosed technology represents a paradigm shift from traditional multi-step syntheses to a streamlined one-pot protocol that utilizes concentrated sulfuric acid as a dual-function medium. By eliminating the need for intermediate isolation and reducing reaction times, this methodology offers substantial improvements in process mass intensity and operational simplicity. For R&D directors and procurement specialists alike, understanding the nuances of this catalytic system is vital for optimizing supply chains and reducing the cost of goods sold for downstream active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-5-aryl-1,2,4-triazole-3-thiones has been plagued by inefficiencies inherent in multi-step linear sequences. Prior art, such as the methods reported by V. Mathew et al., typically involves a tedious three-stage process: first, the formation of aromatic hydrazides from aromatic acids and hydrazine hydrate under reflux; second, the conversion of these hydrazides into dithioformates using carbon disulfide under alkaline conditions; and finally, the cyclization with hydrazine hydrate to yield the target triazole. This conventional pathway is not only time-consuming, often requiring cumulative reaction times exceeding 20 hours, but it also necessitates the isolation and purification of unstable intermediates. Each isolation step introduces potential yield losses, increases solvent consumption, and generates significant volumes of chemical waste, thereby inflating both the environmental footprint and the manufacturing costs. Furthermore, alternative solid-state methods, while shorter in step count, often demand extreme thermal conditions ranging from 170°C to 180°C, which can lead to the decomposition of sensitive substrates and pose safety hazards in large-scale reactors.
The Novel Approach
In stark contrast to these cumbersome legacy procedures, the innovative approach described in CN103130731A achieves the target transformation in a single operational step with remarkable efficiency. By employing concentrated sulfuric acid (98%) as both the solvent and the catalyst, the reaction drives the direct condensation and cyclization of aromatic acids with 1,3-diamino thiourea (thiosemicarbazide). This eliminates the necessity for pre-forming hydrazide intermediates or handling hazardous reagents like carbon disulfide. The reaction proceeds smoothly at moderate temperatures between 90°C and 110°C, completing within 4 to 6 hours, which represents a drastic reduction in processing time compared to traditional methods. The simplicity of the workup procedure, involving merely cooling, neutralization with a carbonate solution, and recrystallization, further enhances the practicality of this method for industrial application. This streamlined workflow not only accelerates the time-to-market for new drug candidates but also significantly lowers the barrier for commercial scale-up, making it an attractive option for contract development and manufacturing organizations (CDMOs) seeking to optimize their production capabilities.
Mechanistic Insights into Sulfuric Acid-Mediated Cyclization
The efficacy of this synthesis relies heavily on the unique physicochemical properties of concentrated sulfuric acid, which acts as a powerful dehydrating agent and a strong Brønsted acid catalyst. In the initial phase of the reaction, the carboxylic acid group of the aromatic substrate is activated by protonation, facilitating a nucleophilic attack by the amino group of the thiosemicarbazide. This leads to the formation of an acyl thiourea intermediate, a crucial precursor that is typically unstable and difficult to isolate in other solvent systems. However, in the highly acidic and dehydrating environment provided by the sulfuric acid medium, water is continuously removed, driving the equilibrium towards the formation of the cyclized product. The acid also promotes the subsequent intramolecular cyclization by activating the thiocarbonyl group and facilitating the elimination of ammonia or water, depending on the specific tautomeric pathway involved. This dual role ensures that the reaction proceeds to completion with high conversion rates, minimizing the presence of unreacted starting materials and side products that could complicate downstream purification.
From an impurity control perspective, the robustness of this acidic medium is particularly advantageous for managing the quality of the final pharmaceutical intermediate. The high solubility of many organic impurities in hot concentrated sulfuric acid, combined with the selective precipitation of the target triazole-thione upon neutralization, provides a built-in purification mechanism. When the reaction mixture is poured into an ice-water bath and neutralized to pH 7 using sodium carbonate or bicarbonate, the target product precipitates out as a solid filter cake, while many polar byproducts and inorganic salts remain in the aqueous phase. Subsequent recrystallization from ethanol or aqueous ethanol further refines the purity, effectively removing trace colored impurities and residual acids. This inherent ability to produce high-purity material (often exceeding 98% purity without chromatography) is a critical factor for R&D directors who must ensure that impurity profiles meet stringent regulatory guidelines for drug substance manufacturing. The consistency of yields across diverse substrates, ranging from electron-deficient nitrobenzoic acids to electron-rich methoxybenzoic acids, underscores the versatility and reliability of this mechanistic pathway.
How to Synthesize 4-Amino-5-Aryl-1,2,4-Triazole-3-Thione Efficiently
Implementing this synthesis on a pilot or commercial scale requires careful attention to mixing dynamics and thermal management to ensure reproducibility and safety. The protocol dictates a specific molar ratio of aromatic acid to thiosemicarbazide to concentrated sulfuric acid, typically ranging from 1.0:1:7.3 to 1.3:1:11.2, ensuring that the acid is present in large excess to function effectively as a solvent. Vigorous stirring at speeds between 1500 and 2500 r/min is essential to maintain homogeneity in the viscous acid medium and to facilitate efficient heat transfer during the exothermic mixing and heating phases. The detailed standardized synthetic steps, including precise addition rates and quenching protocols, are outlined below to guide process engineers in establishing robust manufacturing procedures.
- Mix aromatic acid and 1,3-diamino thiourea in a reaction vessel, then add concentrated sulfuric acid (98%) acting as both solvent and catalyst.
- Heat the reaction mixture to 90°C–110°C with vigorous stirring (1500–2500 r/min) for 4 to 6 hours to complete the cyclization.
- Cool the mixture, neutralize to pH 7 using a carbonate solution in an ice-water bath, filter the precipitate, and recrystallize from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. The consolidation of three distinct reaction steps into a single vessel operation drastically reduces the requirement for intermediate storage, handling equipment, and labor hours associated with multi-step processing. This simplification of the manufacturing workflow inherently lowers the operational expenditure (OPEX) by minimizing the number of unit operations required to produce a kilogram of the final intermediate. Furthermore, the elimination of carbon disulfide, a toxic and volatile reagent used in conventional routes, removes the need for specialized containment systems and expensive scrubbing technologies, thereby reducing capital investment in safety infrastructure and lowering the long-term costs associated with environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the reduction in raw material consumption and solvent usage. By avoiding the isolation of the hydrazide intermediate, manufacturers save on the solvents and energy typically required for drying and purifying that specific step. Additionally, the use of sulfuric acid, a commodity chemical available at low cost, replaces more expensive catalysts or reagents found in alternative pathways. The high atom economy of the direct cyclization ensures that a greater proportion of the input mass is converted into the valuable product, reducing the cost per unit of output. While specific percentage savings depend on local utility costs, the structural elimination of entire processing stages guarantees a significant reduction in the overall cost of goods sold, enhancing the competitiveness of the final API in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction conditions. The tolerance of the method to a wide variety of substituted aromatic acids means that suppliers are not locked into a single, potentially scarce starting material; instead, they can source from a broad pool of commercially available benzoic acid derivatives. The moderate reaction temperature of 90°C to 110°C reduces the risk of thermal runaway incidents that could halt production, ensuring consistent batch-to-batch availability. Moreover, the shortened reaction time of 4 to 6 hours allows for faster turnaround times in multipurpose reactors, increasing the overall capacity of the manufacturing facility to respond to sudden spikes in demand without the need for additional capital investment in new reactor trains.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to metric tons is facilitated by the homogeneous nature of the reaction mixture and the straightforward workup procedure. The absence of gaseous reagents or highly sensitive intermediates simplifies the engineering controls required for large-scale vessels. From an environmental standpoint, the process generates less hazardous waste compared to methods utilizing carbon disulfide or high-temperature solid-state grinding. The aqueous waste stream generated during neutralization can be treated using standard effluent treatment protocols, and the sulfuric acid can potentially be recovered or neutralized safely. This alignment with green chemistry principles not only mitigates regulatory risks but also appeals to end-clients who are increasingly prioritizing sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, substrate scope, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this sulfuric acid-mediated method over conventional multi-step routes?
A: The primary advantage is the reduction of synthetic steps from three distinct stages to a single one-pot operation. Conventional methods often require the isolation of hydrazide intermediates and subsequent reaction with carbon disulfide, which increases processing time and waste. This novel approach utilizes concentrated sulfuric acid to drive the cyclization directly, significantly simplifying the workflow and improving overall throughput.
Q: What is the typical yield range for 4-amino-5-aryl-1,2,4-triazole-3-thione derivatives using this protocol?
A: Experimental data indicates consistently high yields ranging from 85% to 87% across a broad spectrum of substituted aromatic acids. This includes electron-withdrawing groups like nitro and chloro, as well as electron-donating groups like methoxy and hydroxyl, demonstrating the robustness of the catalytic system against varying electronic environments.
Q: How does the reaction temperature compare to solid-state synthesis alternatives?
A: This solution-phase method operates at a moderate temperature range of 90°C to 110°C. In contrast, alternative solid-state grinding methods often require temperatures exceeding 170°C to 180°C. The lower thermal requirement reduces energy consumption and minimizes the risk of thermal degradation for sensitive functional groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-5-Aryl-1,2,4-Triazole-3-Thione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process validation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot synthesis are fully realized in practice. Our state-of-the-art facilities are equipped to handle corrosive media like concentrated sulfuric acid safely, and our stringent purity specifications guarantee that every batch of 4-amino-5-aryl-1,2,4-triazole-3-thione meets the exacting standards required for pharmaceutical applications. With our rigorous QC labs and commitment to continuous process improvement, we provide a secure and reliable source for this critical heterocyclic intermediate.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for your proprietary analogs. By leveraging our manufacturing capabilities, you can accelerate your development timelines and secure a competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
