Advanced Copper-Catalyzed Synthesis of Minodronic Acid Key Intermediates for Commercial Scale-Up
Introduction to Next-Generation Minodronic Acid Synthesis
The pharmaceutical landscape for osteoporosis treatment continues to evolve, driven by the demand for high-purity bisphosphonates like minodronic acid. A pivotal breakthrough in this domain is documented in patent CN112390794B, which discloses a robust preparation method for the key intermediate, imidazo[1,2-a]pyridine-3-acetic acid. This molecule serves as the critical scaffold for constructing the final active pharmaceutical ingredient (API), necessitating a synthesis route that balances high yield with stringent impurity control. The structural complexity of minodronic acid, characterized by its nitrogen-containing heteroaryl bisphosphonate framework, demands precise chemical engineering to ensure therapeutic efficacy.
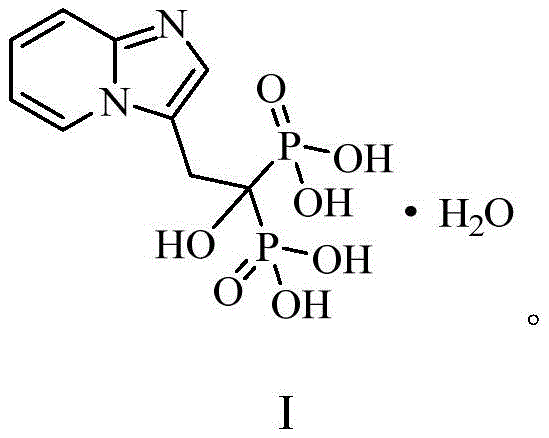
Traditional manufacturing pathways have often struggled with toxic reagents and multi-step inefficiencies, creating bottlenecks for reliable API intermediate suppliers. The disclosed technology addresses these challenges by introducing a streamlined, copper-catalyzed approach that leverages readily available starting materials. By focusing on the fundamental chemistry of ring closure and functional group transformation, this method offers a pathway to high-purity pharmaceutical intermediates that aligns with modern green chemistry principles. For R&D directors and procurement specialists, understanding the nuances of this patented process is essential for securing a competitive edge in the supply of bone resorption inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for minodronic acid precursors have been plagued by significant safety and economic drawbacks that hinder industrial adoption. Early methodologies, such as those utilizing sodium cyanide for nucleophilic substitution, present severe occupational health risks and require complex waste treatment protocols to manage cyanide residues. Other approaches rely on hazardous halogenating agents like liquid bromine or trimethylchlorosilane, which are not only corrosive to reactor equipment but also generate substantial amounts of acidic waste streams. Furthermore, many prior art sequences involve lengthy protection-deprotection strategies or high-temperature decarboxylation steps exceeding 190°C, which promote the formation of difficult-to-remove regioisomers and degrade overall yield.
From a supply chain perspective, several legacy routes depend on specialized starting materials like 3-bromoimidazo[1,2-a]pyridine or 2,2-dimethoxyacetaldehyde, which suffer from limited market availability and volatile pricing. The cumulative effect of these factors is a manufacturing process that is fragile, expensive, and environmentally burdensome. The reliance on stoichiometric amounts of dangerous reagents increases the cost of compliance and insurance, while the low atom economy of multi-step sequences inflates the cost of goods sold (COGS). Consequently, there has been a persistent industry need for a method that eliminates these toxicological and logistical barriers without compromising on chemical quality.
The Novel Approach
The methodology outlined in CN112390794B represents a paradigm shift by employing a direct, catalytic cyclization strategy that bypasses the need for toxic cyanides or harsh halogenation. The core innovation lies in the reaction of ethyl 4-chloroacetoacetate with ammonia to form an enamine intermediate, which subsequently undergoes a copper-catalyzed condensation with 2-halopyridine. This telescoped process allows for the formation of the imidazo[1,2-a]pyridine core in a single pot, drastically reducing solvent usage and processing time. The use of inexpensive copper salts, such as cuprous oxide or cuprous iodide, replaces precious metal catalysts, offering a distinct economic advantage for cost reduction in pharmaceutical intermediate manufacturing.
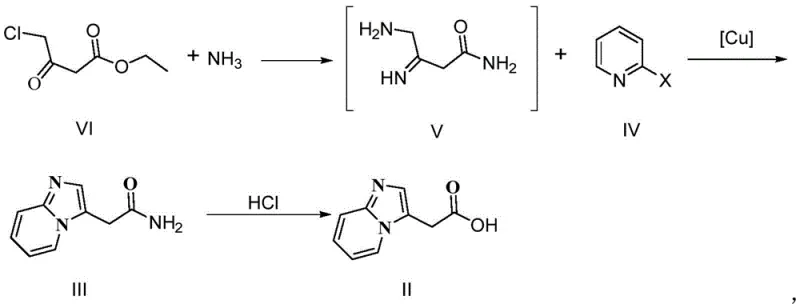
Moreover, the reaction conditions are remarkably mild, operating effectively between 50°C and 80°C, which minimizes energy consumption and thermal degradation of sensitive intermediates. The final hydrolysis step utilizes concentrated hydrochloric acid to convert the acetamide to the corresponding acetic acid, achieving yields consistently above 96% with HPLC purity exceeding 99.5%. This level of efficiency transforms the production economics, enabling commercial scale-up of complex pharmaceutical intermediates with a significantly reduced environmental footprint. By simplifying the workflow to essentially two main chemical transformations, the process enhances operational reliability and facilitates easier regulatory approval due to the cleaner impurity profile.
Mechanistic Insights into Copper-Catalyzed Cyclization
The success of this synthesis hinges on the mechanistic efficiency of the copper-catalyzed ring closure, which facilitates the construction of the fused heterocyclic system with high regioselectivity. In the initial phase, ethyl 4-chloroacetoacetate reacts with ammonia to generate a reactive beta-enamino ester species. This intermediate acts as a 1,3-dinucleophile, poised for attack on the electrophilic carbon of the 2-halopyridine ring. The copper catalyst plays a dual role: it activates the carbon-halogen bond of the pyridine derivative through oxidative addition or coordination, and it stabilizes the transition state during the nucleophilic attack by the enamine nitrogen. This catalytic cycle ensures that the cyclization proceeds rapidly at moderate temperatures, avoiding the high-energy barriers associated with thermal uncatalyzed reactions.
Impurity control is inherently built into this mechanism due to the specificity of the copper-ligand interaction and the thermodynamic stability of the resulting aromatic imidazo[1,2-a]pyridine system. Unlike non-catalytic thermal cyclizations that often produce mixtures of 2-substituted and 3-substituted isomers, this method favors the formation of the 3-acetamide derivative with exceptional selectivity. The subsequent hydrolysis step is equally controlled; by using concentrated hydrochloric acid under reflux, the amide bond is cleaved efficiently without affecting the integrity of the heterocyclic ring or inducing unwanted phosphorylation side reactions at this stage. This precise control over reaction pathways ensures that the final product meets the rigorous purity specifications required for downstream bisphosphonate synthesis, minimizing the burden on purification units.
How to Synthesize Imidazo[1,2-a]pyridine-3-acetic Acid Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and temperature control to maximize the benefits of the one-pot design. The process begins with the ammonolysis of the chloro-keto ester, followed immediately by the introduction of the copper catalyst and the pyridine substrate. This continuity prevents the degradation of the unstable enamine intermediate and streamlines the workflow. Detailed standard operating procedures regarding molar ratios, solvent selection, and workup protocols are critical for reproducing the high yields reported in the patent data. For process chemists looking to adopt this technology, the following guide outlines the critical operational parameters derived from the experimental examples.
- React ethyl 4-chloroacetoacetate with an ammonia organic solution at 50-70°C to form the intermediate enamine species.
- Add a copper catalyst (e.g., cuprous oxide) and 2-chloropyridine directly to the mixture without isolation, heating to 60-80°C for cyclization.
- Hydrolyze the resulting acetamide intermediate with concentrated hydrochloric acid under reflux to obtain the final acetic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing ethyl 4-chloroacetoacetate and 2-chloropyridine, both of which are commodity chemicals produced on a massive global scale, manufacturers can insulate themselves from the supply volatility associated with specialized custom synthons. This shift to widely available feedstocks ensures a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of toxic cyanides and corrosive bromine simplifies logistics, as these hazardous materials no longer require specialized transport and storage infrastructure.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven principally by the removal of expensive and hazardous reagents. Traditional routes often necessitate the use of stoichiometric amounts of sodium cyanide or lithium aluminum hydride derivatives, which carry high purchase prices and disposal costs. In contrast, the copper-catalyzed method uses catalytic quantities of inexpensive metal salts, significantly lowering the direct material cost per kilogram. Additionally, the one-pot nature of the reaction reduces solvent consumption and energy usage by eliminating intermediate isolation and drying steps. These operational efficiencies translate into a lower cost base, allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for API manufacturers, and this route strengthens reliability by decoupling production from scarce reagents. The reliance on bulk chemicals like ammonia and chloropyridines means that sourcing can be diversified across multiple global vendors, mitigating the risk of single-source dependency. The mild reaction conditions also reduce wear and tear on reactor vessels and ancillary equipment, leading to higher asset utilization rates and fewer unplanned maintenance downtimes. This robustness ensures that delivery schedules for critical bone health medications can be met consistently, fostering stronger long-term partnerships between chemical suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for industrial expansion. The absence of highly toxic byproducts simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. The process operates at atmospheric or low pressure with moderate heating, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This ease of scale-up facilitates the rapid transition from pilot plant batches to multi-ton commercial production, ensuring that market demand for minodronic acid can be met swiftly while adhering to increasingly stringent environmental, social, and governance (ESG) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: What are the primary safety advantages of this new synthesis route compared to traditional methods?
A: Unlike conventional routes that utilize highly toxic sodium cyanide or corrosive liquid bromine, this patent employs mild ammonolysis and copper catalysis, significantly reducing operational hazards and environmental toxicity.
Q: How does the one-pot strategy impact production costs?
A: By eliminating the isolation and purification steps between ammonolysis and cyclization, the process reduces solvent consumption, labor hours, and equipment occupancy time, leading to substantial operational cost savings.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the use of commercially available raw materials like ethyl 4-chloroacetoacetate and mild reaction temperatures (50-80°C) ensures high scalability and supply chain stability for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,2-a]pyridine-3-acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced therapeutics like minodronic acid depends on a partner capable of navigating complex chemistry with precision and scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence allows us to optimize processes like the copper-catalyzed cyclization described here, delivering superior quality at optimized costs.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in pharmaceutical intermediates can accelerate your development timeline and secure your supply chain for the future.
