Advanced Synthetic Route for Tazobactam Acid: Scalable Manufacturing and Cost Optimization
Introduction to the Novel Synthetic Pathway
The pharmaceutical industry continuously seeks robust and efficient methodologies for producing critical beta-lactamase inhibitors, among which Tazobactam Acid stands out as a pivotal component in combating bacterial resistance. Patent CN110357903B discloses a groundbreaking synthetic method that fundamentally restructures the production landscape for this high-value active pharmaceutical ingredient (API) intermediate. By leveraging 6-aminopenicillanic acid (6-APA) as the foundational starting material, this innovation bypasses the complex and costly esterification steps typical of legacy processes. The structural integrity of the final molecule, characterized by its specific stereochemistry and sulfone functionality, is meticulously preserved through a series of controlled transformations. 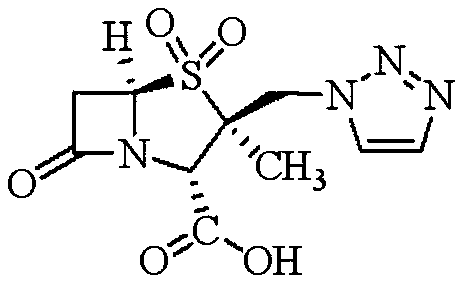
This technical breakthrough is particularly significant for global supply chains aiming to secure reliable sources of high-purity intermediates. The disclosed route not only streamlines the synthesis into fewer operational units but also drastically improves the overall economic feasibility by utilizing commodity chemicals. For R&D directors and procurement strategists, understanding the nuances of this pathway is essential for evaluating potential partnerships and optimizing manufacturing costs. The method ensures that the final white solid powder of Tazobactam Acid achieves a purity level greater than 99.5 percent, meeting the stringent quality standards required for modern antibiotic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tazobactam Acid has been plagued by inefficiencies that hinder large-scale commercial viability. Traditional routes, such as those reported by Deng Yong et al., often involve lengthy sequences starting from 6-APA that require diazotization, bromination, and the preparation of key intermediates like penicillanic acid diphenylmethyl ester-1 alpha-oxide. These multi-step processes frequently result in dismal total yields, sometimes as low as 16.3 percent, due to cumulative losses at each stage. Furthermore, alternative methods described by Taniguchi et al. necessitate the use of hazardous reagents like mercury oxide, posing severe environmental risks and complicating waste management protocols. The reliance on expensive starting materials, such as penicillane-3α carboxylic acid diphenylmethyl ester, further inflates production costs, making these routes economically unattractive for mass manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110357903B introduces a streamlined architecture that prioritizes atom economy and operational simplicity. By initiating the synthesis directly from 6-APA and employing a strategic deamination followed by a highly selective oxidation system, the process eliminates the need for protecting groups like diphenylmethyl esters entirely. This reduction in synthetic steps directly correlates to a substantial increase in total molar yield and a significant reduction in solvent consumption and processing time. The novel approach utilizes a cerium-based catalytic system that allows for precise control over the oxidation state of the sulfur atom, preventing over-oxidation or degradation of the sensitive beta-lactam ring. 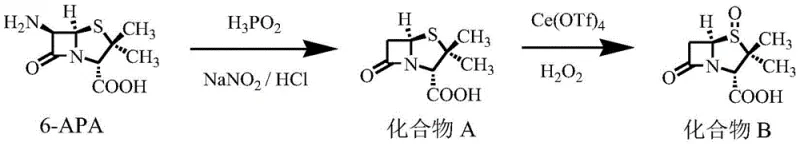
Moreover, the subsequent transformation steps, including the formation of the triazole ring, are optimized for high conversion rates under mild conditions. This ensures that the intermediate products maintain high purity levels throughout the sequence, minimizing the need for extensive recrystallization or chromatographic purification. For supply chain managers, this translates to a more predictable production schedule and reduced risk of batch failures. The ability to achieve such high purity (>99.5%) directly from the synthesis demonstrates a level of process control that is superior to conventional methods, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ce(OTf)4-Catalyzed Selective Oxidation
The cornerstone of this synthetic advancement lies in the sophisticated application of the Ce(OTf)4/H2O2 oxidation system. In traditional sulfide oxidations, achieving selectivity for the sulfoxide or sulfone without affecting other sensitive functional groups, such as the beta-lactam carbonyl or the carboxylic acid, is a formidable challenge. The use of Cerium(IV) triflate acts as a potent Lewis acid catalyst that activates hydrogen peroxide, facilitating a nucleophilic attack on the sulfur atom with exceptional specificity. This mechanistic pathway ensures that the sulfur atom in Compound A is selectively oxidized to the sulfoxide in Compound B, and later to the sulfone in the final product, without inducing ring-opening or epimerization side reactions that commonly plague penicillin derivatives. The reaction conditions are tightly controlled, with the addition of hydrogen peroxide occurring at 0-10 ℃ to manage exothermicity and maintain selectivity.
Furthermore, the deamination step utilizing hypophosphorous acid (H3PO2) and sodium nitrite (NaNO2) in an acidic medium is critical for establishing the correct carbon skeleton early in the synthesis. This reaction replaces the amino group at the 6-position of the penicillin nucleus with a hydrogen atom, a transformation that must proceed with high fidelity to preserve the stereochemical integrity of the adjacent chiral centers. The patent specifies a molar ratio of 6-APA to H3PO2 and NaNO2 of 1:1.5-2.5, ensuring complete conversion while minimizing the formation of nitrosamine byproducts. 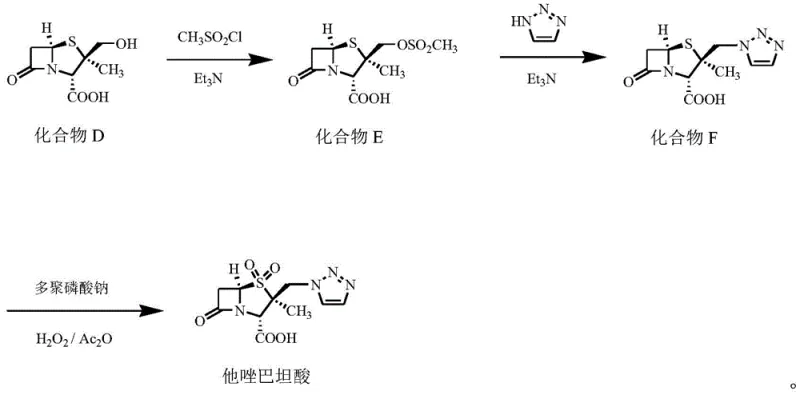
Impurity control is inherently built into this mechanism through the use of specific solvents like dimethyl carbonate and acetone-water mixtures, which favor the desired reaction pathways while suppressing side reactions. The subsequent mesylation and triazole substitution steps are equally precise, utilizing triethylamine as an acid scavenger to drive the nucleophilic substitution to completion. The final oxidation step, employing sodium polyphosphate as a stabilizer alongside hydrogen peroxide and acetic anhydride, ensures the clean conversion of the sulfide/triazole intermediate into the final sulfone product. This comprehensive control over the reaction mechanism results in an impurity profile that is significantly cleaner than that of prior art, reducing the burden on quality control laboratories.
How to Synthesize Tazobactam Acid Efficiently
The synthesis of Tazobactam Acid via this patented route involves a logical sequence of transformations that can be executed with standard pharmaceutical manufacturing equipment. The process begins with the deamination of 6-APA in an acetone-water solvent system at low temperatures (-20 to -10 ℃), followed immediately by the extraction and oxidation to Compound B. The detailed standardized synthesis steps, including specific reagent ratios, temperature profiles, and workup procedures, are provided in the guide below to ensure reproducibility and safety.
- Perform deamination on 6-APA using H3PO2 and NaNO2/HCl to obtain Compound A, followed by selective oxidation with Ce(OTf)4/H2O2 to yield Compound B.
- React Compound B with acetic anhydride under reduced pressure reflux to form Compound C, then treat with hydrazine hydrate to generate Compound D.
- Convert Compound D to mesylate Compound E, substitute with triazole to form Compound F, and finalize with H2O2/Ac2O oxidation to obtain Tazobactam Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical superiority. The shift away from expensive, specialized starting materials like diphenylmethyl esters to the commodity chemical 6-APA represents a fundamental change in the cost structure of Tazobactam Acid production. Since 6-APA is produced on a massive global scale for the penicillin industry, its availability is high, and its price is relatively stable compared to niche intermediates. This switch effectively decouples the production of Tazobactam Acid from the volatility of specialized reagent markets, ensuring a more resilient supply chain capable of withstanding raw material shortages.
- Cost Reduction in Manufacturing: The elimination of multiple protection and deprotection steps, along with the removal of expensive catalysts like mercury oxide, leads to a drastic simplification of the manufacturing process. By reducing the number of unit operations, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. The high yield of each step means that less raw material is wasted, directly improving the cost of goods sold (COGS). Furthermore, the avoidance of heavy metals simplifies the wastewater treatment process, removing the need for costly heavy metal scavenging resins and specialized disposal protocols, which contributes to substantial operational savings.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as acetic anhydride, hydrazine hydrate, and triazole ensures that production is not bottlenecked by single-source suppliers. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic high-pressure or cryogenic setups (beyond standard cooling), enhances the flexibility of manufacturing sites. This adaptability allows for easier technology transfer between different facilities, mitigating the risk of supply disruptions caused by site-specific issues. The high purity of intermediates also reduces the likelihood of batch rejection, ensuring a consistent flow of finished goods to meet market demand.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with steps like reduced pressure reflux at 95-105 ℃ being easily manageable in large reactors. The use of greener solvents and the absence of toxic heavy metals align with increasingly strict global environmental regulations, such as REACH and EPA guidelines. This compliance reduces the regulatory burden on manufacturers and minimizes the risk of production shutdowns due to environmental violations. The ability to scale from pilot batches to multi-ton production without significant process re-engineering makes this route ideal for meeting the growing global demand for beta-lactamase inhibitor combinations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These answers are derived directly from the experimental data and process descriptions within the patent, providing a clear picture of the operational realities and benefits associated with this technology.
Q: What are the primary advantages of this new Tazobactam Acid synthesis route?
A: The patented method offers significantly shorter reaction steps compared to traditional routes, utilizes cheap and readily available starting materials like 6-APA, and achieves a final product purity exceeding 99.5% with a high total molar yield.
Q: How does this process address environmental and safety concerns in pharmaceutical manufacturing?
A: Unlike older methods that rely on toxic mercury oxide or expensive diphenylmethyl esters, this route employs safer reagents and avoids heavy metal contamination, simplifying waste treatment and enhancing operational safety.
Q: Is this synthetic method suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability, featuring simple operation steps, standard reaction conditions (such as reduced pressure reflux), and high intermediate purity which reduces the burden on downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tazobactam Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of Tazobactam Acid for the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of Tazobactam Acid meets or exceeds the >99.5% purity benchmark established by this innovative patent.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and enhance your competitive edge in the antibiotic market.
