Revolutionizing Racecadotril Production: A Deep Dive into the Novel Amidation-First Industrial Route
The pharmaceutical landscape for antidiarrheal agents continues to evolve, driven by the relentless demand for higher purity and more efficient manufacturing processes. Patent CN110283109B, published in early 2021, introduces a transformative methodology for the industrial preparation of Racecadotril, a critical prodrug enkephalinase inhibitor. This intellectual property addresses long-standing bottlenecks in the legacy synthesis routes, specifically targeting the notorious difficulties associated with oily intermediates and unpredictable crystallization behaviors. By fundamentally reordering the synthetic sequence—prioritizing amidation before thiol addition—the disclosed technology offers a pathway to significantly enhanced yield and purity profiles. For global procurement leaders and R&D directors, this represents a pivotal shift towards more robust supply chains for this essential gastrointestinal therapeutic.
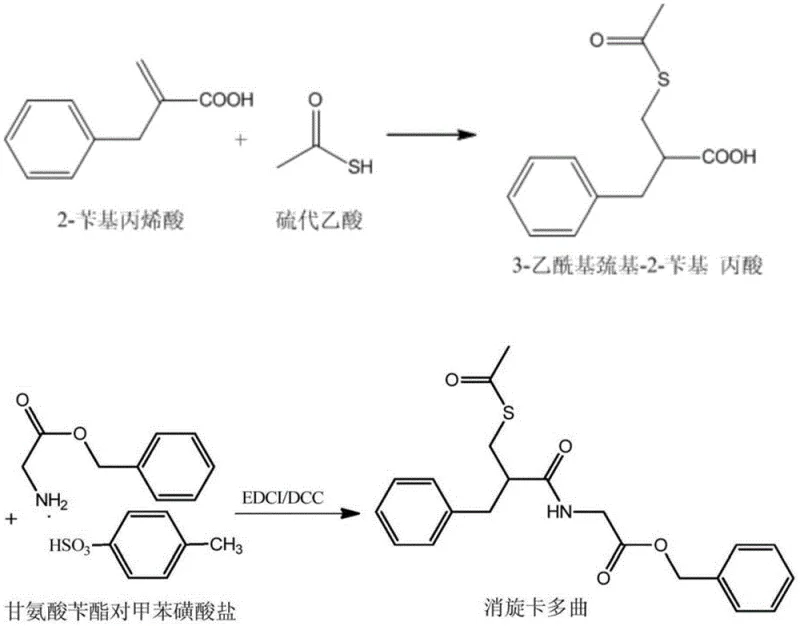
For decades, the conventional synthesis of Racecadotril has been plagued by the inherent instability and purification challenges of its precursors. The traditional approach typically involves reacting 2-benzyl acrylic acid with thioacetic acid first to generate 3-acetylmercapto-2-benzyl propionic acid. Unfortunately, this intermediate exists as an oil that stubbornly retains 5-7% of unknown impurities which cannot be removed via distillation. Furthermore, the specific structure of this oily substance makes it nearly impossible to find a suitable crystallization solvent, leading to low overall yields and a final product burdened with difficult-to-remove byproducts. The situation is exacerbated during the final crystallization of Racecadotril itself, where 'explosive crystallization' phenomena often occur, making material discharge difficult and limiting reactor capacity due to safety concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy manufacturing route suffers from a fundamental flaw in its reaction sequence design, creating a purification bottleneck right at the midpoint of the synthesis. When the thio-group is introduced early, the resulting carboxylic acid intermediate lacks the structural rigidity required for effective crystallization, forcing manufacturers to rely on chromatographic or distillation techniques that are costly and inefficient at scale. The presence of persistent unknown impurities at the 5-7% level not only drags down the final yield but also complicates the regulatory filing process, as identifying and qualifying these degradants becomes a resource-intensive endeavor. Additionally, the final step involving the coupling with glycine benzyl ester often requires complex activation strategies that introduce further impurities, compounding the purification burden.
The Novel Approach
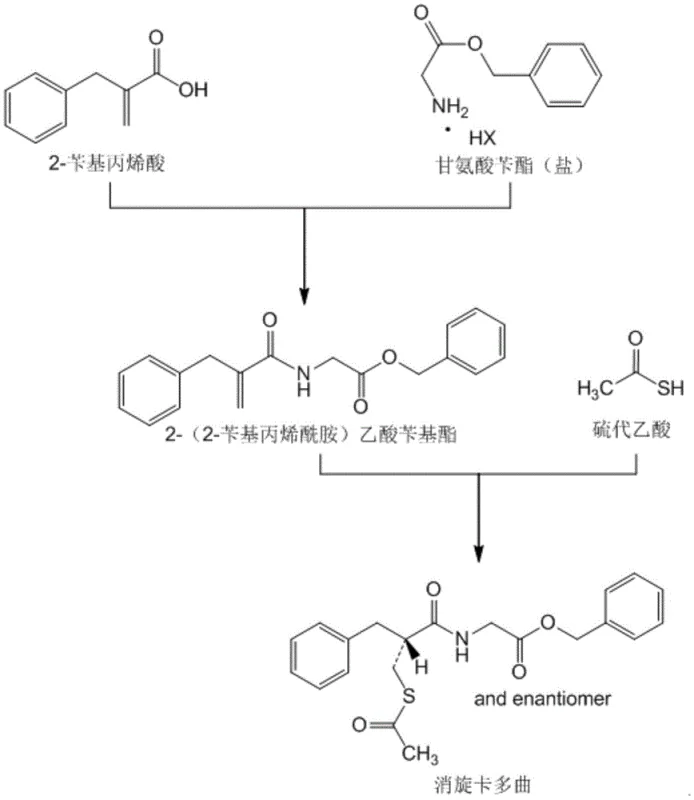
In a brilliant inversion of standard retrosynthetic logic, the inventors of CN110283109B propose performing the amidation reaction first. By coupling 2-benzyl acrylic acid directly with benzyl glycinate salts using DCC (dicyclohexylcarbodiimide) as a condensing agent, the process generates benzyl 2-(2-benzyl acrylamide) acetate as a stable, crystalline solid. This strategic pivot transforms the purification paradigm; instead of struggling with an intractable oil, manufacturers now handle a solid intermediate that can be purified to >99.5% purity through simple standing crystallization. The byproduct of this coupling, dicyclohexylurea (DCU), is poorly soluble and can be mechanically removed via centrifugation, streamlining the workflow and eliminating the need for complex aqueous workups at this stage.
Mechanistic Insights into DCC-Mediated Amidation and Thiol Addition
The core of this technological breakthrough lies in the precise control of the carbodiimide-mediated coupling mechanism. In the first step, the carboxylic acid group of 2-benzyl acrylic acid reacts with DCC to form an O-acylisourea intermediate, which is highly susceptible to nucleophilic attack by the amine group of the benzyl glycinate salt. The presence of an acid-binding agent, such as triethylamine or pyridine, is crucial here to neutralize the protonated amine salt, freeing the nucleophile to attack the activated carbonyl. This reaction proceeds efficiently at low temperatures (-2 to 5°C) to minimize racemization and side reactions, followed by a slow warm-up to 25-35°C to drive the reaction to completion over 12-14 hours. The result is a clean formation of the amide bond, setting the stage for the subsequent functionalization.
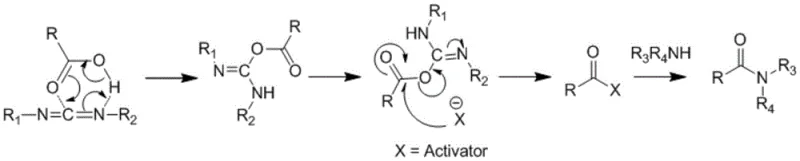
Following the isolation of the pure amide intermediate, the second phase involves a Michael-type addition of thioacetic acid across the alpha-beta unsaturated double bond. This step is conducted in an alcohol solvent at elevated temperatures (70-82°C), facilitating the conjugate addition of the sulfur nucleophile. A key mechanistic advantage here is the stability of the amide backbone during this thermal treatment, which prevents hydrolysis or degradation. Furthermore, the patent describes a sophisticated workup strategy where water is added post-reaction to hydrolyze any trace unreacted DCC into insoluble DCU. This ensures that the final crystallization of Racecadotril from the alcohol-water mixture yields a product virtually free of coupling reagents, addressing one of the most common purity failure modes in peptide-like syntheses.
How to Synthesize Racecadotril Efficiently
The implementation of this novel route requires careful attention to solvent selection and temperature gradients to maximize the benefits of the solid-state intermediate. The process begins with the dispersion of reactants in solvents like dichloromethane, ethyl acetate, or toluene, followed by the controlled addition of the DCC mixture. The critical innovation is the standing crystallization step, which avoids the hazards of rapid precipitation. Detailed standardized operating procedures for temperature control, solvent ratios, and filtration protocols are essential for reproducing the high yields reported in the patent examples. For a comprehensive breakdown of the exact operational parameters, please refer to the technical guide below.
- Disperse 2-benzyl acrylic acid and benzyl glycinate salt in solvent, then add DCC and acid-binding agent at low temperature to form the solid intermediate benzyl 2-(2-benzyl acrylamide) acetate.
- Remove the DCU byproduct via centrifugation and purify the intermediate through standing crystallization using alkane solvents.
- React the purified solid intermediate with thioacetic acid in alcohol solvent at elevated temperatures, followed by water-induced crystallization to isolate pure Racecadotril.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift from an oily to a solid intermediate strategy offers profound implications for cost structures and supply reliability. The ability to purify the intermediate via crystallization rather than chromatography or distillation drastically reduces solvent consumption and processing time. This simplification of the unit operations translates directly into lower manufacturing costs and a reduced environmental footprint, aligning with modern green chemistry initiatives. Moreover, the elimination of the 'explosive crystallization' risk in the final step enhances plant safety and allows for larger batch sizes, effectively increasing throughput without requiring additional capital investment in new reactors.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and energy-intensive purification steps associated with oily intermediates. By utilizing DCC, which generates a solid byproduct (DCU) that is easily filtered off, the process avoids the complex aqueous extractions often required with other coupling agents like EDCI. This reduction in downstream processing complexity leads to substantial cost savings in labor, utilities, and waste disposal, making the final API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the solid intermediate ensures consistent quality output, reducing the risk of batch failures that can disrupt supply schedules. The use of common, commodity solvents such as toluene, ethyl acetate, and alcohols ensures that raw material availability is not a bottleneck. Furthermore, the improved yield and purity profiles mean that less starting material is required to produce the same amount of finished goods, optimizing inventory turnover and reducing the working capital tied up in raw materials.
- Scalability and Environmental Compliance: The standing crystallization technique described in the patent is inherently safer and more scalable than the aggressive agitation required for traditional methods. This allows for smoother technology transfer from pilot plant to commercial production scales. Additionally, the efficient removal of byproducts minimizes the load on wastewater treatment facilities, facilitating compliance with increasingly stringent environmental regulations regarding organic solvent discharge and chemical waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this novel synthesis route. Understanding these nuances is critical for quality assurance teams evaluating potential suppliers who utilize this patented technology. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation.
Q: How does the new amidation-first route improve impurity control compared to traditional methods?
A: Traditional methods generate an oily thio-intermediate that traps 5-7% unknown impurities and resists distillation. The new route creates a solid amide intermediate early, allowing rigorous purification via crystallization and centrifugal removal of DCU before the final step, ensuring >99.5% purity.
Q: What are the scalability advantages of the standing crystallization technique described in the patent?
A: The patent replaces dangerous 'explosive crystallization' with controlled standing crystallization. This allows for safe material discharge even at high solute-to-solvent ratios (5-7), significantly reducing processing time and equipment bottlenecks during commercial scale-up.
Q: How is the difficult-to-remove DCC byproduct managed in this process?
A: The process utilizes a clever conversion strategy where trace residual DCC is hydrolyzed into insoluble DCU by adding water during the final workup. The DCU is then easily removed via filtration, ensuring the final API meets stringent residual solvent and impurity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racecadotril Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN110283109B requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this amidation-first strategy are fully realized in a GMP environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities like DCC and DCU at ppm levels, guaranteeing that every batch of Racecadotril meets the highest international pharmacopoeial standards.
We invite pharmaceutical manufacturers and procurement specialists to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthesis route, we can offer a supply solution that balances premium quality with economic efficiency. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can strengthen your supply chain for this critical antidiarrheal agent.
