Revolutionizing BBIT Production: A Halogen-Free Route for Scalable Polymer Additive Manufacturing
Revolutionizing BBIT Production: A Halogen-Free Route for Scalable Polymer Additive Manufacturing
The global demand for high-performance antimicrobial agents in polymer matrices has necessitated a re-evaluation of synthetic pathways for key intermediates like N-n-butyl-1,2-benzisothiazolin-3-one (BBIT). Patent CN103012308A introduces a transformative methodology that replaces hazardous halogen-based oxidation with a benign hydrogen peroxide system. This innovation addresses critical pain points in the fine chemical sector, specifically targeting the toxicity and equipment corrosion associated with traditional chlorine or bromine usage. By shifting to an oxidative disproportionation mechanism in an alkaline medium, this technology not only enhances operational safety but also streamlines the purification process, offering a compelling value proposition for manufacturers of polymer additives and industrial biocides seeking sustainable production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzisothiazolinone derivatives has relied heavily on aggressive halogenating agents to effect the necessary ring closure and oxidation steps. As illustrated in the traditional reaction pathways, processes often involve the use of elemental chlorine or liquid bromine, which are classified as highly toxic and corrosive substances. These reagents necessitate the use of specialized, expensive corrosion-resistant reactor materials, such as glass-lined steel or Hastelloy, drastically inflating capital expenditure for production facilities. Furthermore, the handling of toxic gases poses significant occupational health risks and requires complex scrubbing systems to manage hazardous off-gases, creating a substantial environmental burden that complicates regulatory compliance and increases operational overheads for chemical manufacturers.
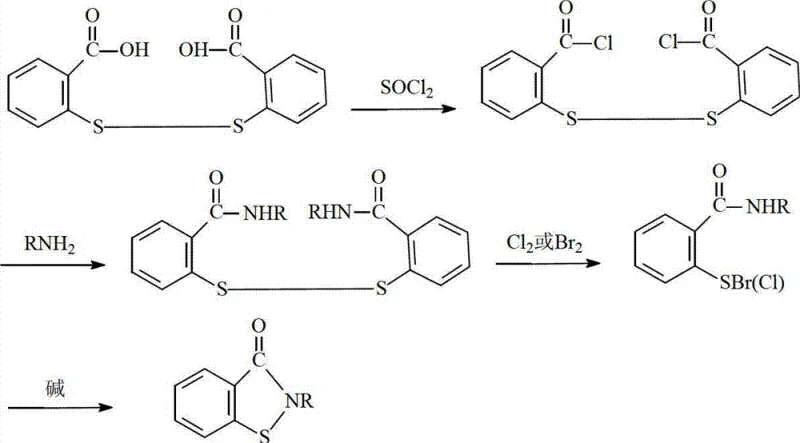
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel approach detailed in the patent utilizes hydrogen peroxide as the primary oxidant within a carefully controlled alkaline environment. This method initiates with the amidation of 2,2'-dithiobis(benzoyl chloride) with n-butylamine, followed by a sophisticated oxidative cyclization. The substitution of halogens with hydrogen peroxide fundamentally alters the safety profile of the reaction, eliminating the generation of corrosive hydrohalic acid byproducts. This shift allows for the use of standard stainless steel equipment, significantly reducing infrastructure costs. The reaction proceeds via a clean disproportionation mechanism that yields water as the primary inorganic byproduct, aligning perfectly with modern green chemistry principles and simplifying downstream waste treatment protocols for industrial facilities.
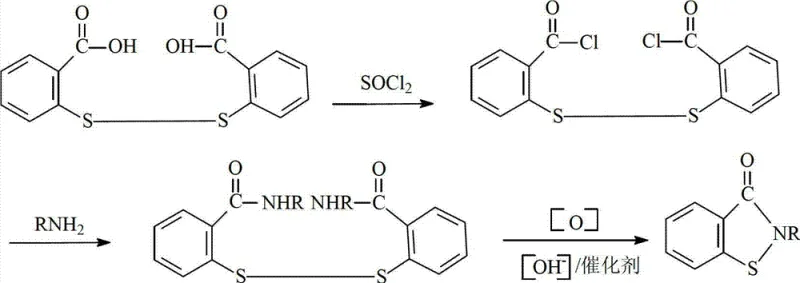
Mechanistic Insights into Oxidative Disproportionation Cyclization
The core of this technological advancement lies in the mechanistic elegance of the oxidative cyclization step. Upon formation of the N-n-butyl-2,2'-dithiobis(benzamide) intermediate, the introduction of an organic base catalyst and an alkaline solution creates a nucleophilic environment conducive to sulfur-sulfur bond activation. The hydrogen peroxide acts as an oxygen donor, facilitating the cleavage of the disulfide bridge and the simultaneous formation of the sulfur-nitrogen bond required for the isothiazolinone ring structure. This disproportionation reaction is kinetically favored at moderate temperatures between 50°C and 70°C, ensuring high selectivity for the desired cyclic product while minimizing the formation of over-oxidized sulfone or sulfoxide impurities that often plague harsher oxidation methods.
Impurity control is inherently superior in this system due to the mild nature of the oxidant and the specific catalytic role of tertiary amines like triethylamine. In traditional halogen routes, electrophilic aromatic substitution can lead to ring-halogenated byproducts which are difficult to separate and detrimental to the thermal stability of the final polymer additive. The peroxide-mediated pathway avoids electrophilic attack on the aromatic ring entirely, preserving the integrity of the benzene moiety. Additionally, the alkaline conditions ensure that any acidic byproducts are immediately neutralized, preventing acid-catalyzed degradation of the sensitive isothiazolinone ring, thereby resulting in a crude product with a significantly cleaner impurity profile that requires less intensive purification.
How to Synthesize N-n-butyl-1,2-benzisothiazolin-3-one Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing BBIT with high consistency. The process begins with the preparation of the disulfide diamide precursor through a straightforward amidation reaction at low temperatures, ensuring minimal side reactions. Following isolation, the key cyclization step is performed by suspending the intermediate in an organic solvent such as toluene and treating it with aqueous alkali and a catalytic amount of organic base. The controlled addition of hydrogen peroxide drives the ring closure to completion. For a detailed breakdown of the specific molar ratios, temperature gradients, and workup procedures required to replicate this high-efficiency synthesis, please refer to the standardized technical guide below.
- Perform amidation by reacting 2,2'-dithiobis(benzoyl chloride) with n-butylamine in an organic solvent at 0-30°C to form the disulfide diamide intermediate.
- Prepare the cyclization mixture by adding the intermediate to an alkaline solution containing an organic base catalyst and organic solvent.
- Add hydrogen peroxide continuously at 50-70°C to induce oxidative disproportionation and ring closure, yielding the final BBIT product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this halogen-free synthesis represents a strategic opportunity to de-risk the supply of critical polymer additives. The reliance on commodity chemicals like hydrogen peroxide and sodium hydroxide, rather than regulated toxic gases, ensures a more stable and resilient raw material supply chain. This stability is crucial for maintaining continuous production schedules, as it eliminates the logistical complexities and safety inspections associated with transporting and storing hazardous halogen cylinders. Consequently, manufacturers can achieve a more predictable production cadence, reducing the risk of supply interruptions that often occur with tightly controlled hazardous reagents.
- Cost Reduction in Manufacturing: The elimination of corrosive halogens removes the necessity for exotic, high-cost construction materials in reaction vessels and piping systems. Facilities can utilize standard grade stainless steel reactors, which significantly lowers both initial capital investment and long-term maintenance costs associated with corrosion repair. Furthermore, the simplified waste treatment process, driven by the absence of heavy halogen loads in the effluent, reduces the operational expenditure related to environmental compliance and hazardous waste disposal, leading to substantial overall cost savings in the manufacturing lifecycle.
- Enhanced Supply Chain Reliability: By utilizing widely available oxidants and bases, the production process becomes less susceptible to regional regulatory restrictions on toxic chemical transport. This flexibility allows for a more diversified supplier base for raw materials, mitigating the risk of single-source dependency. The simplified logistics also translate to faster turnaround times for raw material procurement, enabling the production team to respond more agilely to market demand fluctuations without being bottlenecked by the lead times of specialized hazardous chemical deliveries.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, removing the engineering challenges associated with high-pressure reactors required for some gas-phase halogenations. This inherent safety margin facilitates easier scale-up from pilot to commercial production volumes. Moreover, the generation of water as a primary byproduct aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tightening emission standards and reducing the carbon footprint associated with the production of these specialty polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of safety, efficiency, and product quality for industrial applications.
Q: Why is the hydrogen peroxide method superior to traditional halogen oxidation for BBIT?
A: Traditional methods utilize toxic and corrosive halogens like chlorine or bromine, requiring specialized anti-corrosion equipment and posing severe safety risks. The patented hydrogen peroxide method eliminates these hazards, reduces equipment costs, and generates water as a benign byproduct, significantly lowering environmental compliance burdens.
Q: What are the critical reaction conditions for the oxidative cyclization step?
A: The cyclization requires a strongly alkaline environment (pH 10-14) and temperatures between 50-70°C. An organic base catalyst, such as triethylamine, is essential to facilitate the disproportionation of the disulfide bond and subsequent ring closure efficiently.
Q: Can this process be scaled for industrial production of polymer additives?
A: Yes, the process operates under mild pressure and temperature conditions without requiring high-pressure reactors. The use of commodity chemicals like hydrogen peroxide and common solvents like toluene makes the supply chain robust and highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-n-butyl-1,2-benzisothiazolin-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes for high-value polymer additives. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify the absence of halogenated impurities and ensure consistent thermal stability for your polymer formulations.
We invite you to collaborate with us to leverage this advanced halogen-free technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product performance while reducing your total cost of ownership.
