Advanced Copper-Catalyzed Synthesis of Tranylcypromine Intermediates for Commercial Scale-Up
Introduction to Next-Generation Anticoagulant Intermediate Synthesis
The pharmaceutical industry continuously seeks robust and scalable pathways for critical anticoagulant precursors, particularly those required for next-generation P2Y12 inhibitors like ticagrelor. Patent CN112479895A introduces a transformative synthetic methodology for producing tranylcypromine intermediates, addressing long-standing inefficiencies in chiral alcohol synthesis. This innovative approach replaces hazardous and costly reagents with a streamlined copper-catalyzed system, offering a compelling solution for manufacturers aiming to optimize their supply chains for high-value cardiovascular therapeutics. By leveraging accessible raw materials and achieving exceptional stereocontrol, this technology represents a significant leap forward in process chemistry, enabling reliable production of complex chiral scaffolds essential for modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of tranylcypromine intermediates has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional routes heavily rely on expensive chiral oxazaborolidine catalysts and toxic borane dimethyl sulfide compounds, which pose severe safety risks and disposal challenges in an industrial setting. Furthermore, these legacy processes often suffer from overly complicated operation steps, requiring stringent anhydrous conditions and multiple purification stages that drastically reduce overall throughput. The low yields associated with these conventional methods result in substantial raw material waste, inflating the cost of goods sold and creating bottlenecks in the supply of critical anticoagulant ingredients. Additionally, the use of high-boiling solvents like DMF complicates solvent recovery and increases the environmental footprint of the manufacturing process, making it less attractive for sustainable production initiatives.
The Novel Approach
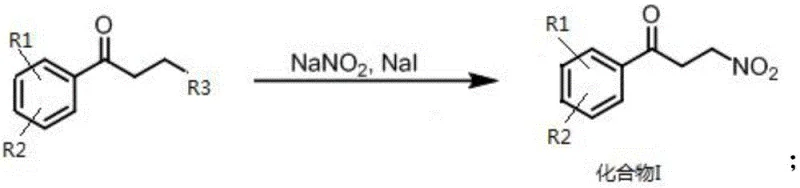
In stark contrast, the novel methodology disclosed in the patent utilizes a sophisticated yet practical sequence starting with a substitution reaction on substituted aromatic ketones under mild conditions. This initial step employs sodium nitrite and sodium iodide in common solvents like acetone, avoiding the need for exotic reagents while ensuring high conversion rates. The core innovation lies in the subsequent copper-hydrogen catalytic reaction, which achieves asymmetric reduction with remarkable precision using low-cost metal precursors and ligands. This approach not only simplifies the operational workflow but also delivers yields reaching up to 99% with enantiomeric excess values of 95.5%, far surpassing the performance of prior art. By eliminating toxic boranes and optimizing solvent systems, this new route offers a safer, more economical, and environmentally friendly pathway for producing high-purity pharmaceutical intermediates.
Mechanistic Insights into Copper-Hydrogen Catalytic Asymmetric Reduction
The heart of this synthetic breakthrough is the copper-hydrogen catalytic system, which facilitates the enantioselective reduction of the nitro-ketone intermediate to a chiral alcohol. Mechanistic studies highlight the critical role of the ligand environment, where SegPhos demonstrates superior performance over BINAP in maintaining high enantiomeric purity throughout the catalytic cycle. The reaction proceeds through a well-defined copper-hydride species generated in situ from phenylsilane, which transfers hydride to the carbonyl group with high facial selectivity dictated by the chiral ligand. Optimization data reveals that copper acetate monohydrate serves as a more effective metal precursor than its anhydrous counterpart, likely due to subtle coordination effects that stabilize the active catalytic species. Furthermore, the choice of solvent is paramount, with toluene providing the optimal balance of solubility and stereocontrol, whereas polar solvents like THF or dichloromethane lead to significant drops in both yield and ee value.

Impurity control is another vital aspect of this mechanism, particularly regarding the stability of the nitro group under varying pH conditions. The process is designed to avoid basic environments that could trigger the premature removal of the nitro functionality, ensuring the integrity of the substrate for downstream cyclization. The subsequent cyclization step, utilizing DIAD and triphenylphosphine, efficiently constructs the cyclopropane ring with inversion of configuration, setting the stage for the final reduction to the amine. This mechanistic understanding allows for precise tuning of reaction parameters, such as temperature and stoichiometry, to minimize side reactions and maximize the formation of the desired trans-cyclopropylamine stereoisomer. Such deep mechanistic insight is crucial for scaling this chemistry from the laboratory bench to multi-ton commercial production without compromising quality.
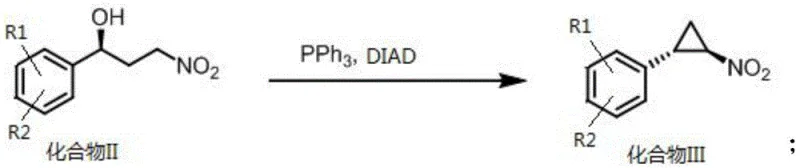
How to Synthesize Tranylcypromine Intermediate Efficiently
Implementing this advanced synthetic route requires careful attention to reaction conditions and reagent quality to fully realize its potential for high-yield production. The process begins with the preparation of the nitro-ketone substrate, followed by the critical asymmetric reduction step which demands strict exclusion of moisture and oxygen to maintain catalyst activity. Detailed standard operating procedures for each transformation, including specific workup protocols and purification strategies, are essential for ensuring consistent batch-to-batch reproducibility. For a comprehensive guide on executing these transformations with maximum efficiency and safety, please refer to the standardized synthesis protocol outlined below.
- Perform substitution reaction on substituted aromatic ketone using sodium nitrite and sodium iodide to synthesize Compound I (1-aryl-3-nitro-propan-1-one).
- Execute copper-hydrogen catalytic reaction on Compound I using a metal precursor (copper acetate), SegPhos ligand, and phenylsilane to generate chiral Compound II.
- React Compound II with DIAD and triphenylphosphine to form the cyclopropane ring structure of Compound III, followed by reduction with zinc powder to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic methodology offers profound advantages by fundamentally restructuring the cost drivers associated with chiral intermediate manufacturing. The elimination of proprietary and expensive chiral borane reagents removes a significant variable cost component, allowing for more predictable budgeting and reduced exposure to volatile raw material markets. Moreover, the use of commodity chemicals such as acetone, toluene, and zinc powder ensures a stable and diversified supply base, mitigating the risk of single-source dependency that often plagues specialty chemical supply chains. The robustness of the reaction conditions, particularly the air-stability of the initial acylation step, further enhances operational flexibility, allowing for simpler facility requirements and reduced downtime.
- Cost Reduction in Manufacturing: The transition to a copper-catalyzed system drastically lowers the cost of goods by replacing precious chiral catalysts with abundant base metals and simple ligands. The high atom economy of the reaction sequence minimizes waste generation, leading to substantial savings in waste disposal and raw material consumption. Additionally, the ability to achieve near-quantitative yields in key steps reduces the need for extensive recycling or reprocessing, directly impacting the bottom line through improved material efficiency and lower energy consumption per kilogram of product.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials and avoiding reagents with complex regulatory or sourcing constraints, this route significantly de-risks the supply chain. The simplified process flow reduces the number of unit operations required, shortening the overall manufacturing cycle time and improving responsiveness to market demand fluctuations. This reliability is further bolstered by the method's tolerance to minor variations in reaction conditions, ensuring consistent output quality even in large-scale production environments where perfect control is challenging.
- Scalability and Environmental Compliance: The replacement of high-boiling, difficult-to-remove solvents like DMF with lower-boiling alternatives like acetone simplifies solvent recovery and reduces the energy burden of distillation processes. The absence of toxic borane byproducts simplifies effluent treatment and aligns the process with increasingly stringent environmental regulations, facilitating smoother permitting and operational continuity. This green chemistry profile not only reduces compliance costs but also enhances the sustainability credentials of the final pharmaceutical product, a key factor for modern healthcare providers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tranylcypromine intermediate synthesis. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supply chain integration.
Q: What are the advantages of this copper-catalyzed method over traditional borane methods?
A: This method eliminates the need for expensive chiral oxazaborolidine and toxic borane dimethyl sulfide compounds. It utilizes low-cost metal precursors and ligands, achieving yields up to 99% and ee values of 95.5%, significantly reducing raw material waste and operational complexity.
Q: How does the process ensure high enantioselectivity during the reduction step?
A: High enantioselectivity is achieved through the precise selection of the SegPhos ligand and copper acetate monohydrate precursor. Optimization studies indicate that SegPhos outperforms BINAP in ee value retention, and maintaining anhydrous, oxygen-free conditions with toluene as the solvent is critical for maximizing stereochemical purity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. Key steps like the Friedel-Crafts acylation are air-stable, and the substitution reaction uses common solvents like acetone instead of high-boiling DMF. The process avoids complex purification steps, making it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tranylcypromine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of life-saving anticoagulants. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this copper-catalyzed method are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of tranylcypromine intermediate meets the highest standards required for API synthesis, providing our partners with unwavering confidence in product quality and consistency.
We invite you to engage with our technical procurement team to explore how this innovative synthesis can drive value in your specific applications. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic impact of switching to this superior manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines and volume requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
