Scalable Synthesis of Cyclopentenyl and Cyclohexenyl Pinacol Borates for Commercial Drug Manufacturing
Scalable Synthesis of Cyclopentenyl and Cyclohexenyl Pinacol Borates for Commercial Drug Manufacturing
The pharmaceutical industry's relentless pursuit of novel therapeutic agents has placed cycloalkenyl structural units at the forefront of medicinal chemistry, particularly for their utility in constructing complex biaryl and heteroaryl scaffolds via palladium-catalyzed cross-coupling reactions. Patent CN103145746A discloses a robust and versatile process method for synthesizing cyclopentene and hexene-1-boronic acid pinacol cyclic esters, which serve as critical building blocks in the assembly of active pharmaceutical ingredients (APIs). This technology addresses the growing demand for reliable high-purity pharmaceutical intermediates by offering a synthetic pathway that balances chemical efficiency with economic feasibility. By leveraging the Shapiro reaction mechanism, the process converts readily available ketones into valuable vinyl boronates through a sequence involving tosylhydrazone formation, lithiation, and subsequent boronation. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is essential for securing a stable supply chain of these high-value coupling partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cycloalkenyl boronic esters has relied on methodologies that often suffer from limited substrate scope, expensive reagents, or cumbersome purification protocols that hinder large-scale adoption. Prior art, such as the routes documented in literature like J. Am. Chem. Soc. 2008, typically involves multi-step sequences where the introduction of the boron moiety can be inefficient or prone to side reactions that generate difficult-to-remove impurities. These conventional pathways frequently utilize specialized boronating agents that drive up the cost of goods sold (COGS) and may require harsh reaction conditions that compromise the integrity of sensitive functional groups elsewhere in the molecule. Furthermore, the lack of flexibility in reagent choice often forces manufacturers to rely on single-source suppliers for niche reagents, creating bottlenecks in the supply chain that can delay critical drug development timelines. The reliance on less stable intermediates in older methods also poses risks regarding batch-to-batch consistency, a critical parameter for regulatory compliance in GMP manufacturing environments.
The Novel Approach
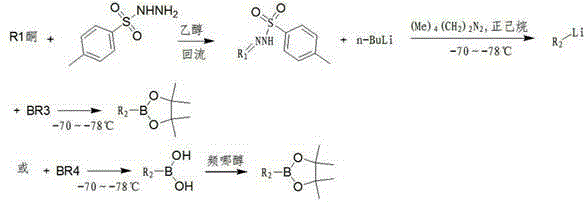
In contrast, the methodology outlined in CN103145746A introduces a streamlined and adaptable strategy that significantly mitigates the drawbacks associated with traditional synthesis routes. The core innovation lies in the generation of a vinyllithium intermediate via the decomposition of a ketone tosylhydrazone, which is then trapped with a variety of boron electrophiles to form the desired pinacol ester. This approach allows for the use of diverse boron sources, ranging from direct pinacol borates to more economical trialkyl borates, providing procurement teams with the flexibility to optimize raw material costs without sacrificing yield. The process operates under controlled cryogenic conditions to ensure high selectivity during the lithiation step, followed by a straightforward workup and purification via vacuum distillation that delivers products with exceptional purity profiles. By decoupling the boron source from the final ester structure until the final steps, this novel approach empowers manufacturers to tailor the synthesis based on real-time market availability of reagents, thereby enhancing supply chain resilience.
Mechanistic Insights into Shapiro Reaction-Mediated Boronation
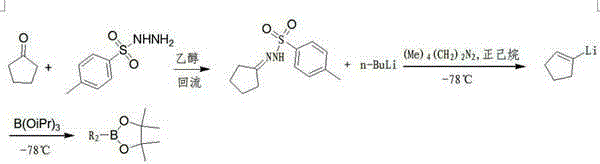
The chemical elegance of this process is rooted in the Shapiro reaction mechanism, where the initial formation of the tosylhydrazone serves as a masked precursor for the generation of a highly reactive vinyllithium species. Upon treatment with a strong base such as n-butyllithium in the presence of a chelating agent like tetramethylethylenediamine (TMEDA), the tosylhydrazone undergoes a double elimination of nitrogen and the sulfonyl group to yield the corresponding alkenyllithium intermediate. This transformation is highly temperature-dependent, requiring precise control between -70°C and -78°C to prevent competing decomposition pathways or protonation by the solvent. The resulting organolithium species acts as a potent nucleophile, capable of attacking the electrophilic boron center of various reagents, including triisopropyl borate or methoxy pinacol borate, to forge the carbon-boron bond with high fidelity. Understanding this mechanistic pathway is crucial for R&D teams aiming to replicate the process, as the stoichiometry of the base and the rate of addition directly influence the conversion efficiency and the profile of side products formed during the reaction.
Impurity control in this synthesis is primarily achieved through the strategic selection of reaction conditions and the final purification step, which leverages the volatility differences between the product and byproducts. The use of inert gas protection throughout the synthesis prevents the oxidation of the sensitive organolithium intermediate, which could otherwise lead to the formation of hydroxylated impurities or homocoupling products. Following the boronation step, the reaction mixture is quenched and extracted, with the organic layer subjected to concentration and subsequent reduced pressure distillation. This physical separation technique is highly effective in removing residual solvents, unreacted boron species, and higher boiling point oligomers, resulting in a final product that meets rigorous purity specifications, often exceeding 99% GC area normalization. For quality assurance professionals, this distillation step provides a robust checkpoint to ensure that the final API intermediate is free from genotoxic impurities or heavy metal contaminants that might be associated with alternative transition-metal catalyzed borylation methods.
How to Synthesize Cyclopentenyl Pinacol Borate Efficiently
The practical execution of this synthesis requires careful attention to thermal management and reagent addition rates to maximize the yield of the vinyllithium intermediate. Operators must ensure that the lithiation step is conducted under strictly anhydrous conditions to prevent premature quenching of the organometallic species, which would drastically reduce the overall throughput of the process. The patent details specific molar ratios for the reactants, suggesting a slight excess of the boronating agent to drive the reaction to completion while maintaining a balance that minimizes waste. Detailed standardized operating procedures for the synthesis, including specific temperature ramps and quenching protocols, are essential for transferring this laboratory-scale success to pilot and commercial production facilities. The following guide outlines the critical operational phases derived from the patent examples to assist technical teams in implementing this route.
- Synthesize the ketone tosylhydrazone by refluxing the corresponding ketone with p-toluenesulfonyl hydrazide in an alcoholic solvent.
- Generate the vinyllithium intermediate by treating the tosylhydrazone with n-butyllithium and TMEDA at cryogenic temperatures between -70°C and -78°C.
- Quench the lithiated species with a boron reagent (such as trialkyl borate or alkoxy pinacol borate) followed by esterification and vacuum distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for organizations focused on cost reduction in pharmaceutical intermediate manufacturing and supply chain reliability. The ability to interchange boron reagents means that procurement managers are not locked into a single supplier for expensive specialty chemicals; instead, they can leverage commodity chemicals like trimethyl borate which are produced on a massive global scale and are subject to less price volatility. This flexibility acts as a hedge against supply disruptions, ensuring that production schedules for critical drug candidates remain uninterrupted even when specific niche reagents face shortages. Furthermore, the high yields reported in the patent examples translate directly into improved atom economy and reduced waste disposal costs, contributing to a more sustainable and economically viable manufacturing process that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts often required in direct C-H borylation methods, thereby removing the costly and time-consuming step of heavy metal scavenging from the downstream processing workflow. By utilizing simple ketones and hydrazines as starting materials, the raw material cost base is significantly lowered compared to routes requiring pre-functionalized halides or organometallic reagents that are hazardous to transport and store. The operational expenditure is further optimized by the use of standard unit operations such as distillation and extraction, which do not require specialized equipment beyond standard cryogenic reactors found in most fine chemical plants. Consequently, the overall cost of goods is drastically simplified, allowing for competitive pricing in the global market for complex building blocks.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals such as cyclopentanone, p-toluenesulfonyl hydrazide, and n-butyllithium ensures a robust supply chain that is less susceptible to the geopolitical or logistical constraints often affecting exotic reagents. Since the synthesis does not depend on a single proprietary catalyst or a rare earth element, the risk of supply interruption due to mining restrictions or export controls is effectively mitigated. This stability is paramount for long-term commercial contracts where consistent delivery of high-quality intermediates is a contractual obligation. Additionally, the process scalability allows for rapid ramp-up of production volumes to meet sudden spikes in demand during late-stage clinical trials or commercial launch phases without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, as demonstrated by the patent's emphasis on its suitability for industrialized production, with reaction parameters that can be safely managed in large-scale stirred tank reactors. The waste stream generated is primarily composed of organic solvents and salt byproducts which can be treated using standard effluent treatment protocols, avoiding the generation of toxic heavy metal sludge that complicates environmental compliance. The high selectivity of the reaction minimizes the formation of complex byproduct mixtures, simplifying the purification train and reducing the volume of solvent required for chromatography or recrystallization. This streamlined approach not only lowers the environmental footprint of the manufacturing process but also accelerates the regulatory approval process by presenting a cleaner and more well-defined impurity profile to health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims presented in the patent documentation. These insights are intended to clarify the operational feasibility and quality attributes of the resulting cycloalkenyl boronates for potential licensees and manufacturing partners. Understanding these details is critical for making informed decisions about integrating this chemistry into existing production portfolios.
Q: What is the expected purity of the cyclopentenyl pinacol borate produced via this method?
A: According to the patent data, the crude product can achieve a GC peak content of approximately 80-85%, and subsequent rectification or vacuum distillation can elevate the purity to 99% GC, meeting stringent pharmaceutical standards.
Q: Can this synthesis route be adapted for different boron reagents to optimize costs?
A: Yes, the process offers significant flexibility by allowing the use of either direct pinacol borates (like methoxy or isopropoxy pinacol borate) or cheaper trialkyl borates (like trimethyl or triisopropyl borate) followed by an esterification step, enabling cost optimization based on raw material availability.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly states that the method features good process stability and high production rates, making it highly suitable for industrialized production, provided that cryogenic conditions (-70°C to -78°C) are properly managed during the lithiation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentenyl Pinacol Borate Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in accelerating the drug discovery and development pipeline. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the identity and potency of every batch before release. Our facility is equipped to handle the cryogenic requirements and inert atmosphere conditions necessary for this specific lithiation chemistry, guaranteeing consistent quality and supply continuity for our global partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your commercial goals and enhance your competitive advantage in the marketplace.
