Industrial Biocatalytic Synthesis of (S)-3-(Pyrrolidin-2-yl)pyridine Using Engineered Imine Reductase Mutants
Industrial Biocatalytic Synthesis of (S)-3-(Pyrrolidin-2-yl)pyridine Using Engineered Imine Reductase Mutants
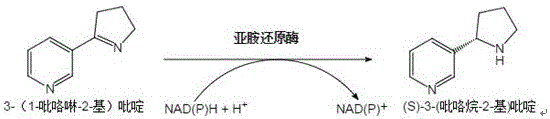
The global demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors continues to drive innovation in biocatalytic manufacturing. A significant breakthrough in this domain is detailed in patent CN113774036A, which discloses a novel imine reductase mutant specifically engineered for the asymmetric reduction of 3-(1-pyrroline-2-yl)pyridine. This compound serves as a critical chiral precursor in the synthesis of nicotine and related alkaloids, representing a high-value target for fine chemical producers. The patent describes a protein engineering strategy that modifies the amino acid sequence of the wild-type enzyme derived from Nocardiopsis alba, resulting in a biocatalyst with dramatically enhanced activity and stereoselectivity. For R&D directors and process chemists, this development offers a compelling alternative to traditional chemical reduction methods, promising superior optical purity and operational efficiency. As a reliable pharmaceutical intermediate supplier, understanding these enzymatic advancements is crucial for optimizing supply chains and reducing the environmental footprint of complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the biocatalytic production of (S)-3-(pyrrolidin-2-yl)pyridine faced significant hurdles regarding substrate tolerance and reaction kinetics. Existing technologies, such as those disclosed in WO 2020098978A1, utilized imine reductases like IRED-C which demonstrated adequate performance only at relatively low substrate concentrations. Specifically, while IRED-C could achieve high conversion at 58 g/L, its efficiency plummeted when the substrate concentration was increased to industrially relevant levels, such as 1 mol/L (approximately 146 g/L), where conversion dropped to merely 52.4% even after 24 hours. This limitation necessitates large reactor volumes and extensive downstream processing to recover the product, thereby inflating capital expenditure and operational costs. Furthermore, the prolonged reaction times associated with these older enzymes create bottlenecks in production schedules, limiting the overall throughput of manufacturing facilities. For procurement managers, these inefficiencies translate into higher costs per kilogram and potential supply chain vulnerabilities due to the inability to rapidly scale production to meet market surges.
The Novel Approach
The technology presented in CN113774036A overcomes these historical barriers through the strategic mutation of the imine reductase gene. By introducing specific point mutations, namely A246V and D285V, either individually or in combination, the inventors have created an enzyme variant that thrives under high-substrate conditions. The double mutant, designated as SEQ ID NO: 4, exhibits exceptional catalytic prowess, achieving a conversion rate of over 99.9% at a substrate loading of 100 g/L within a mere 4 hours. This represents a paradigm shift in process intensity, allowing manufacturers to process significantly more material in less time without compromising on yield. The robustness of this mutant enzyme simplifies the reaction setup, as it maintains high activity and stereoselectivity (e.e. > 99.3%) even under the rigorous demands of industrial batch processing. This advancement directly addresses the need for cost reduction in pharmaceutical intermediate manufacturing by maximizing reactor utilization and minimizing the energy and time inputs required per unit of product generated.
Mechanistic Insights into Imine Reductase-Catalyzed Asymmetric Reduction
The core of this technological leap lies in the precise modification of the enzyme's active site architecture to better accommodate the bulky pyridine substrate. The wild-type imine reductase, while functional, possesses steric or electronic constraints that limit its turnover number (kcat) and binding affinity (Km) for 3-(1-pyrroline-2-yl)pyridine. The substitution of Alanine with Valine at position 246 and Aspartic Acid with Valine at position 285 alters the hydrophobic environment and spatial configuration of the catalytic pocket. These changes facilitate a more efficient hydride transfer from the cofactor NAD(P)H to the imine bond of the substrate. The reaction mechanism involves the oxidation of the reduced cofactor NAD(P)H to NAD(P)+, which is subsequently regenerated in situ by a coupled glucose dehydrogenase (GDH) system using glucose as the sacrificial electron donor. This cofactor recycling loop is essential for economic viability, as it eliminates the need for stoichiometric amounts of expensive nicotinamide cofactors.
Furthermore, the mutant enzyme demonstrates remarkable stability and specificity within a narrow physiological window, which is critical for maintaining product quality. The patent data indicates that the reaction must be strictly controlled within a pH range of 6.8 to 7.8, with an optimum around 7.0 to 7.2. Deviations from this range lead to rapid denaturation or loss of catalytic activity, highlighting the importance of automated pH control systems in the manufacturing process. Similarly, the temperature profile is optimized between 25°C and 30°C; operating outside this range reduces reaction velocity. From an impurity control perspective, the high enantioselectivity (e.e. > 99.3%) ensures that the formation of the unwanted (R)-enantiomer is negligible. This minimizes the burden on downstream purification steps, such as crystallization or chromatography, which are often required to upgrade optical purity in less selective chemical processes. For quality assurance teams, this inherent selectivity provides a robust safety margin for meeting stringent regulatory specifications for chiral drugs.
How to Synthesize (S)-3-(Pyrrolidin-2-yl)pyridine Efficiently
Implementing this biocatalytic route requires a systematic approach to strain cultivation and reaction engineering to fully realize the kinetic benefits of the A246V-D285V mutant. The process begins with the transformation of the mutant gene into a suitable host, typically E. coli BL21(DE3), followed by fermentation to produce the intracellular enzyme. Once the biomass is harvested and lysed to obtain the crude enzyme liquid, the biotransformation can be initiated. The protocol emphasizes the use of a phosphate buffer system to maintain the critical pH balance throughout the reaction, alongside a glucose-driven cofactor regeneration system to sustain catalytic turnover. Detailed standard operating procedures regarding induction times, cell lysis methods, and specific reagent concentrations are vital for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples to ensure successful technology transfer.
- Preparation of the biocatalyst by expressing the A246V-D285V mutant gene in E. coli BL21(DE3) and harvesting the crude enzyme liquid via centrifugation.
- Establishment of the reaction system containing 100 g/L substrate, phosphate buffer (pH 7.0-7.2), glucose dehydrogenase for cofactor regeneration, and the mutant enzyme.
- Incubation at 25-30°C for 4 hours with pH control, followed by workup involving activated carbon treatment and filtration to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this mutant imine reductase technology offers transformative economic benefits. The primary driver of value is the drastic increase in space-time yield; by compressing a reaction that previously took 24 hours into a 4-hour window, manufacturers can effectively sextuple their production capacity using existing infrastructure. This intensification reduces fixed costs per unit and accelerates cash flow cycles. Additionally, the ability to run reactions at higher substrate concentrations (100 g/L vs. the previous ~58 g/L limit) means less solvent and water are required per kilogram of product, leading to substantial savings in raw material procurement and waste disposal costs. The elimination of harsh chemical reagents and heavy metal catalysts further simplifies the environmental compliance landscape, reducing the overhead associated with hazardous waste management and worker safety protocols.
- Cost Reduction in Manufacturing: The enhanced catalytic efficiency of the A246V-D285V mutant allows for significantly lower enzyme loading to achieve complete conversion. The patent data highlights that only 0.3wt of crude enzyme liquid (relative to wet cell weight) is required to transform 1 gram of substrate, a figure that is markedly lower than typical industry standards for similar biotransformations. This reduction in biocatalyst consumption directly lowers the variable cost of goods sold (COGS). Furthermore, the high conversion rate (>99.9%) minimizes the loss of valuable starting material, ensuring that nearly every gram of input is converted to saleable product. By avoiding the need for complex recycling loops or extensive purification to remove unreacted starting material, the overall process economics are optimized, delivering a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of the recombinant enzyme system ensures consistent batch-to-batch performance, which is a critical factor for long-term supply agreements. Unlike wild-type enzymes that may exhibit variability or sensitivity to minor process fluctuations, this engineered mutant maintains high activity across a defined operational window, reducing the risk of batch failures. The use of a common host organism like E. coli and standard fermentation techniques means that the biocatalyst itself can be produced at scale with high reliability, securing the upstream supply of the enzyme. For supply chain heads, this translates to predictable lead times and the confidence to commit to large-volume contracts without fear of biological instability disrupting the flow of critical nicotine intermediates to downstream customers.
- Scalability and Environmental Compliance: Biocatalysis is inherently greener than traditional synthetic chemistry, and this specific process exemplifies those advantages. The reaction proceeds under mild aqueous conditions (near neutral pH and moderate temperatures), eliminating the need for high-pressure hydrogenation equipment or toxic organic solvents often used in chemical reduction. This simplifies the engineering requirements for scale-up, as standard stainless steel reactors can be utilized without specialized lining or pressure ratings. Moreover, the byproduct of the cofactor regeneration system is gluconic acid, which is benign and easily treated in standard wastewater facilities. This alignment with green chemistry principles not only reduces the environmental impact but also future-proofs the supply chain against tightening global regulations on industrial emissions and chemical usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imine reductase technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this biocatalytic route into existing production portfolios. We encourage technical teams to review these details closely to assess the alignment with their specific process requirements and quality standards.
Q: How does the A246V-D285V mutant compare to wild-type imine reductase in terms of catalytic efficiency?
A: The A246V-D285V double mutant exhibits approximately 12 times higher catalytic efficiency compared to the wild-type parent enzyme. It achieves over 99.9% conversion at a high substrate loading of 100 g/L within just 4 hours, whereas previous methods struggled with lower concentrations and longer reaction times.
Q: What are the optimal reaction conditions for this biocatalytic process?
A: The process operates optimally at a controlled pH range of 7.0 to 7.2 using a phosphate buffer and a temperature between 25°C and 30°C. Deviations outside the pH range of 6.8 to 7.8 significantly reduce reaction speed and yield, making precise pH stat control essential for industrial success.
Q: Is this method suitable for large-scale manufacturing of nicotine intermediates?
A: Yes, the method is specifically designed for industrial application. It utilizes a robust recombinant E. coli expression system and achieves high space-time yields with minimal enzyme loading (0.3wt), addressing the scalability limitations of prior art enzymes which showed poor performance at high substrate concentrations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-(Pyrrolidin-2-yl)pyridine Supplier
The technological potential of the A246V-D285V imine reductase mutant represents a significant opportunity for the efficient production of high-value chiral intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory findings into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of (S)-3-(pyrrolidin-2-yl)pyridine meets the highest international standards for optical purity and chemical identity.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized biocatalytic platforms, we can help you reduce total landed costs while securing a stable supply of critical materials. Please contact our technical procurement team to request specific COA data and route feasibility assessments. We are committed to partnering with you to drive innovation and efficiency in your supply chain, ensuring that you have access to the highest quality intermediates for your pharmaceutical and agrochemical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
