Advanced Triphosgene Chlorination Strategy for Commercial Scale-Up of Complex Pyrimidine Intermediates
Introduction to Next-Generation Pyrimidine Chlorination Technology
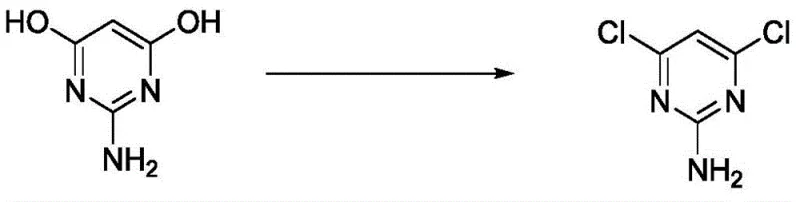
The global demand for high-performance agrochemical and pharmaceutical intermediates necessitates a paradigm shift in synthetic methodology, moving away from legacy processes plagued by inefficiency and environmental burden. Patent CN109053590B introduces a groundbreaking approach for the preparation of 4,6-dichloro-2-aminopyrimidine, a critical building block in the synthesis of various herbicides and medicinal compounds. This technology leverages triphosgene as a superior chlorinating agent within a toluene solvent system, fundamentally altering the reaction kinetics and thermodynamic profile compared to traditional phosphorus oxychloride routes. By operating at mild temperatures between 70°C and 75°C, the process mitigates thermal degradation risks while ensuring complete conversion of the starting material, 4,6-dihydroxy-2-aminopyrimidine. For R&D directors and procurement strategists, this patent represents a viable pathway to securing a reliable agrochemical intermediate supplier capable of delivering consistent quality without the volatility associated with older, waste-intensive chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4,6-dichloro-2-aminopyrimidine has relied heavily on the use of phosphorus oxychloride (POCl3) in conjunction with tertiary amines such as N,N-dimethylaniline acting as acid scavengers. As detailed in prior art like U.S. Pat. No. 3,399,190, these conventional methods require boiling reflux conditions around 107°C, which imposes severe energy demands and increases the risk of side reactions that generate complex impurity profiles. A critical bottleneck in these legacy processes is the post-reaction workup; the acid-binding agent forms salts that are notoriously difficult to recover, leading to substantial waste acid streams that complicate environmental compliance and drive up disposal costs. Furthermore, the yields in these traditional setups often stagnate around 70%, with purity levels struggling to exceed 92%, forcing manufacturers to invest in expensive recrystallization or chromatography steps to meet the stringent specifications required for high-purity OLED material or pharmaceutical applications.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes triphosgene dissolved in toluene to effect the chlorination under significantly milder conditions, eliminating the immediate need for an acid-binding agent during the primary reaction phase. This strategic modification not only simplifies the reaction mixture but also drastically reduces the formation of by-products, allowing for a direct isolation of the target molecule through a simple pH adjustment and phase separation protocol. The elimination of the amine scavenger during the reaction step means that the downstream processing is streamlined, removing the need for complex filtration of amine salts and reducing the overall cycle time. This approach facilitates the commercial scale-up of complex pyrimidine derivatives by providing a robust, reproducible pathway that consistently delivers yields upwards of 92%, thereby maximizing asset utilization and raw material efficiency for large-scale manufacturing facilities.
Mechanistic Insights into Triphosgene-Mediated Chlorination
The efficacy of this synthesis lies in the unique reactivity of triphosgene, which serves as a safe and controllable solid equivalent of phosgene, releasing reactive chlorinating species in situ upon thermal decomposition in the solvent matrix. When introduced to the 4,6-dihydroxy-2-aminopyrimidine substrate in toluene, the triphosgene facilitates a nucleophilic substitution where the hydroxyl groups are sequentially replaced by chlorine atoms, driven by the evolution of carbon dioxide and hydrogen chloride gas which escapes the system or is managed in the scrubber. Unlike the POCl3 mechanism which generates phosphoric acid byproducts that require neutralization, the gaseous byproducts of the triphosgene reaction minimize the accumulation of non-volatile residues in the reaction vessel. This clean reaction profile is essential for maintaining high purity, as it prevents the entrapment of phosphorous-containing impurities that are difficult to remove and can act as catalyst poisons in subsequent downstream coupling reactions used in drug discovery.
Furthermore, the control of impurities is inherently built into the solvent selection and temperature profile, as toluene provides an ideal medium for solubilizing the organic intermediates while allowing for easy phase separation upon aqueous workup. The absence of a bulky amine scavenger during the chlorination step ensures that the reaction mixture remains homogeneous and free from precipitating salts that could occlude product or catalyze decomposition pathways. By adjusting the pH to a neutral range of 7-8 post-reaction using sodium hydroxide, any residual acidic components are neutralized without inducing hydrolysis of the sensitive chloro-pyrimidine ring, a common failure mode in overly basic or acidic environments. This precise control over the chemical environment ensures that the final product meets the rigorous quality standards expected of a high-purity electronic chemical or advanced pharmaceutical intermediate.
How to Synthesize 4,6-Dichloro-2-Aminopyrimidine Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to strict stoichiometric controls and temperature gradients to maximize the benefits of the triphosgene chemistry. The process begins with the suspension of the dihydroxy precursor in toluene, followed by the controlled addition of the chlorinating agent to manage exotherms and gas evolution safely. Detailed operational parameters regarding addition rates, stirring speeds, and specific quenching protocols are critical for reproducing the high yields reported in the patent literature.
- Charge 4,6-dihydroxy-2-aminopyrimidine and toluene into a reactor, heat to 70-75°C, and add triphosgene solution dropwise.
- Maintain reaction temperature for approximately 6 hours to ensure full conversion to the dichloro intermediate.
- Cool the mixture, adjust pH to 7-8 using sodium hydroxide solution, separate layers, and dry the organic phase to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this triphosgene-based route offers transformative advantages that directly impact the bottom line and operational resilience. The most significant benefit is the drastic simplification of the production workflow, which removes the dependency on volatile and often supply-constrained acid scavengers like N,N-dimethylaniline, thereby stabilizing the raw material basket and reducing exposure to price fluctuations in the amine market. By eliminating the generation of difficult-to-treat waste acids and amine salts, the process significantly lowers the environmental compliance burden, translating into reduced operational expenditures related to wastewater treatment and hazardous waste disposal. This lean manufacturing approach not only enhances the sustainability profile of the supply chain but also shortens the overall production lead time, enabling faster response to market demands for critical agrochemical intermediates.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the elimination of the acid-binding agent which traditionally accounts for a significant portion of raw material costs and waste processing fees. Without the need to purchase, recover, or dispose of large quantities of tertiary amines, the variable cost per kilogram of the final product is substantially decreased. Additionally, the higher reaction yield of over 90% means that less starting material is wasted, improving the overall atom economy and reducing the cost of goods sold. These efficiencies compound over large production runs, offering a distinct competitive advantage in pricing strategies for bulk chemical contracts.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of triphosgene and toluene relies on commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The simplified workup procedure, which avoids complex filtration and salt removal steps, decreases the equipment turnaround time and increases the throughput capacity of existing reactor trains. This operational agility ensures a more consistent and reliable supply of 4,6-dichloro-2-aminopyrimidine, safeguarding downstream production schedules for finished pesticides or pharmaceuticals against raw material shortages.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method make it highly scalable, as the reduction in waste volume simplifies the engineering requirements for plant expansion. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory tightening. This compliance advantage is crucial for maintaining uninterrupted operations in regions with strict environmental oversight, ensuring long-term business continuity for partners relying on this key intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology, derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or process optimization projects.
Q: Why is triphosgene preferred over phosphorus oxychloride for this synthesis?
A: Triphosgene allows for milder reaction conditions (70-75°C) compared to the harsh reflux required for phosphorus oxychloride. Crucially, it eliminates the need for an acid scavenger like N,N-dimethylaniline during the reaction, simplifying downstream purification and reducing waste generation.
Q: What is the expected purity and yield of this novel process?
A: According to patent data, this method achieves yields up to 92% with a content purity of approximately 95.5%. This represents a significant improvement over conventional methods which typically yield around 70-75% with lower purity profiles.
Q: How does this process impact environmental compliance?
A: The process is designed to be green and environmentally friendly by avoiding the generation of 'three wastes' commonly associated with traditional chlorination. The absence of difficult-to-recover acid-binding agents significantly reduces the burden on wastewater treatment systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dichloro-2-Aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in the competitiveness of the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to adapt the triphosgene chlorination process allows us to offer a superior product profile that meets the exacting needs of R&D directors and formulation scientists alike.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic efficiencies available through our manufacturing platform. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the confidence of a secure and high-quality supply foundation.
