Advanced Biocatalytic Route for High-Purity (S)-5-Methyl-2-Pyrrolidone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for the production of chiral building blocks. Patent CN114686451A represents a significant technological breakthrough in this domain, specifically addressing the synthesis of (S)-5-methyl-2-pyrrolidone, a critical intermediate for various bioactive compounds. This patent discloses a novel class of amine dehydrogenase mutants derived from Thermoanaerobacter thermohydrosulfuricus that exhibit dramatically improved catalytic performance compared to wild-type enzymes. By leveraging protein engineering and directed evolution, the inventors have created biocatalysts capable of operating under mild conditions while maintaining exceptional stereoselectivity. For R&D directors and procurement specialists, this technology offers a compelling alternative to traditional metal-catalyzed hydrogenation, promising enhanced purity profiles and simplified downstream processing. The ability to utilize renewable biomass-derived levulinic acid as a starting material further aligns this process with modern green chemistry principles, making it a highly attractive option for sustainable manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-methyl-2-pyrrolidone has relied heavily on chemical catalysis, which presents several inherent drawbacks that complicate large-scale manufacturing and quality control. Conventional methods often utilize hydrogen gas as a reducing agent and ammonia or organic amines as nitrogen sources in the presence of metal catalysts such as nickel supported on diatomite. These processes typically require harsh reaction conditions, including temperatures as high as 200°C and elevated pressures, which pose significant safety risks and energy consumption challenges. Furthermore, chemical hydrogenation frequently suffers from poor stereoselectivity, with some reported methods yielding products with enantiomeric excess values lower than 10%, necessitating costly and wasteful resolution steps to isolate the desired optical isomer. Even more recent chemical approaches using ammonium formate as a hydrogen source have struggled to achieve high stereoselectivity, often resulting in moderate ee values that are insufficient for high-value pharmaceutical applications. The reliance on transition metals also introduces the risk of heavy metal contamination in the final product, requiring rigorous and expensive purification protocols to meet stringent regulatory standards for pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the biocatalytic route described in the patent utilizes engineered amine dehydrogenase mutants to catalyze the asymmetric reductive amination of levulinic acid with remarkable efficiency and precision. This novel approach operates under mild physiological conditions, typically around 40°C and neutral pH, which significantly reduces energy requirements and eliminates the need for high-pressure equipment. The core of this innovation lies in the specific mutations introduced into the TtherAmDH enzyme sequence, such as E298G, V82T, and others, which collectively enhance the enzyme's affinity for the substrate and its stability in industrial reaction environments. By coupling this mutant enzyme with formate dehydrogenase, the process achieves efficient in-situ regeneration of the NADH cofactor, minimizing the consumption of expensive reagents. The result is a streamlined one-pot process that converts levulinic acid directly into the chiral amino acid intermediate, which subsequently undergoes spontaneous or chemically induced cyclization to form the target lactam. This method not only simplifies the synthetic route but also ensures product purity levels exceeding 99% with enantiomeric excess values greater than 99% (S), effectively eliminating the need for complex chiral separation techniques.
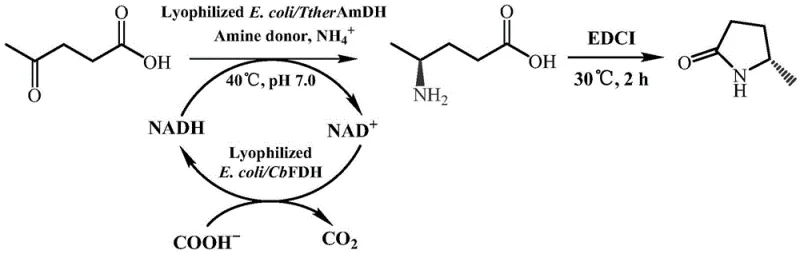
Mechanistic Insights into Amine Dehydrogenase Catalyzed Asymmetric Reductive Amination
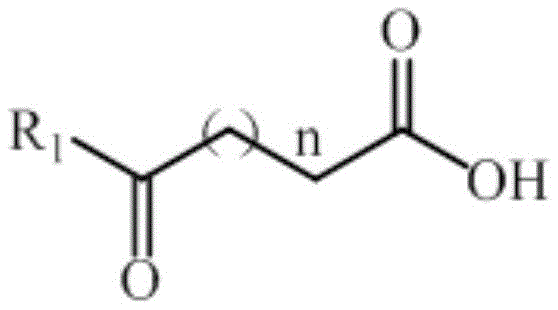
The mechanistic foundation of this technology rests on the precise molecular interactions between the engineered amine dehydrogenase and the prochiral carbonyl substrate. The patent details how specific amino acid residues within the enzyme's active site, such as those at positions 11, 24, 41, 82, 102, 198, 226, 268, and 298, have been mutated to optimize the binding pocket for levulinic acid and its derivatives. These modifications facilitate a highly stereospecific hydride transfer from the NADH cofactor to the carbonyl carbon, ensuring the formation of the (S)-configured amino acid intermediate with high fidelity. The general structural formula of the substrates accepted by this biocatalyst includes various alkyl chains, demonstrating the enzyme's versatility beyond just levulinic acid. This broad substrate scope is crucial for process flexibility, allowing manufacturers to produce a range of chiral lactams from different keto-acid precursors using the same enzymatic platform. The enzyme's ability to function effectively at high substrate concentrations, up to 500 mM, indicates a robust tolerance to potential inhibitory effects, which is a common bottleneck in biocatalytic processes.
Furthermore, the integration of a cofactor regeneration system is a critical component of the mechanism that drives the economic viability of this process. The reaction utilizes ammonium formate as both a nitrogen source and a sacrificial electron donor for the formate dehydrogenase enzyme. This auxiliary enzyme oxidizes formate to carbon dioxide while reducing NAD+ back to NADH, creating a closed loop that sustains the primary reductive amination reaction without the need for stoichiometric amounts of expensive cofactors. This dual-enzyme cascade ensures that the reaction proceeds to completion with minimal waste generation, as the only by-products are water and carbon dioxide. The subsequent cyclization step, often facilitated by reagents like EDCI or occurring spontaneously under acidic conditions, closes the ring to form the stable lactam structure. Understanding this mechanistic interplay allows process engineers to fine-tune parameters such as pH, temperature, and enzyme loading to maximize space-time yields, which the patent reports can reach impressive levels of 75.3 g/L/day.
How to Synthesize (S)-5-Methyl-2-Pyrrolidone Efficiently
Implementing this biocatalytic route requires a systematic approach to enzyme preparation and reaction engineering to fully realize its potential benefits. The process begins with the construction of recombinant expression vectors containing the gene sequences for the optimized TtherAmDH mutants, which are then transformed into host cells like E. coli BL21(DE3) for high-level protein production. Following fermentation and cell harvesting, the biocatalyst can be used in various forms, including whole cells, crude lysates, or lyophilized powders, depending on the specific manufacturing setup and stability requirements. The reaction is typically conducted in an aqueous buffer system, such as ammonium formate/ammonia at pH 6.5, where the substrate levulinic acid is introduced along with the necessary cofactors and auxiliary enzymes. Careful control of reaction conditions, including temperature maintenance at 40°C and agitation to ensure proper mass transfer, is essential to achieve the reported high conversion rates. Once the biocatalytic reduction is complete, the reaction mixture is treated to induce cyclization, followed by standard extraction and purification techniques to isolate the final high-purity product. The detailed standardized synthesis steps for this process are outlined below.
- Prepare recombinant E. coli BL21(DE3) expressing the engineered TtherAmDH mutant (e.g., V9) and lyophilize the cells or crude enzyme powder.
- Conduct asymmetric reductive amination in ammonium formate buffer (pH 6.5) at 40°C, adding levulinic acid substrate, NAD+, and formate dehydrogenase for cofactor recycling.
- After reaction completion, add EDCI aqueous solution to induce intramolecular cyclization of the amino acid intermediate, followed by extraction and purification to obtain the final lactam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology translates into tangible strategic advantages that extend beyond simple technical metrics. The shift from harsh chemical catalysis to mild biocatalysis fundamentally alters the cost structure of manufacturing chiral intermediates by eliminating the need for expensive noble metal catalysts and high-energy input systems. This transition significantly reduces the capital expenditure associated with pressure-rated reactors and hydrogen handling infrastructure, thereby lowering the barrier to entry for scaling production. Moreover, the high stereoselectivity of the enzyme means that the process generates virtually no unwanted enantiomers, which drastically simplifies the purification workflow and reduces the volume of solvents and consumables required for chromatography or crystallization. This efficiency gain leads to substantial cost savings in waste treatment and raw material utilization, making the overall process more economically resilient against fluctuations in commodity prices. The robustness of the mutant enzymes also ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification products.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for costly metal scavenging steps and reduces the risk of heavy metal contamination in the final API, leading to significant downstream processing savings. Additionally, the mild reaction conditions operate at atmospheric pressure and moderate temperatures, which drastically lowers energy consumption compared to traditional high-temperature hydrogenation methods. The efficient cofactor recycling system minimizes the requirement for expensive NADH, further driving down the variable costs per kilogram of product. These factors combined create a leaner manufacturing process that is less sensitive to the volatility of energy and precious metal markets.
- Enhanced Supply Chain Reliability: The use of recombinant enzymes produced in standard microbial hosts like E. coli ensures a stable and scalable supply of the biocatalyst, independent of geopolitical constraints often associated with mining rare earth metals. The high substrate tolerance of the mutants allows for concentrated reaction mixtures, which reduces the volume of water and buffers needed, thereby shrinking the logistical footprint of raw material transport and storage. This compact process design enhances facility throughput and allows for more flexible production scheduling to meet fluctuating market demands. Furthermore, the simplicity of the aqueous-based system reduces dependency on hazardous organic solvents, mitigating supply chain risks related to solvent availability and regulatory compliance.
- Scalability and Environmental Compliance: The biocatalytic process generates benign by-products such as water and carbon dioxide, aligning perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The absence of toxic heavy metals simplifies wastewater treatment protocols and reduces the environmental liability associated with chemical manufacturing. High space-time yields demonstrated in the patent indicate that the process is readily scalable from laboratory to commercial tonnage without significant loss of efficiency. This scalability ensures that supply chain heads can confidently plan for long-term capacity expansion to support growing pipeline assets without encountering the technical bottlenecks typical of complex chemical syntheses.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the operational parameters and performance metrics of the amine dehydrogenase mutants. These questions address critical aspects such as stereoselectivity, substrate scope, and cofactor management, which are pivotal for process development decisions. The responses are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for industrial implementation. Understanding these details helps stakeholders assess the compatibility of this biocatalytic route with existing infrastructure and quality standards.
Q: How does the enzymatic method compare to traditional chemical hydrogenation in terms of stereoselectivity?
A: Traditional chemical methods often suffer from poor diastereoselectivity, sometimes yielding less than 10% ee, whereas the patented amine dehydrogenase mutants achieve greater than 99% enantiomeric excess (ee) under mild conditions.
Q: What specific improvements do the TtherAmDH mutants offer over the wild-type enzyme?
A: The engineered mutants exhibit significantly higher catalytic activity and tolerance to high substrate concentrations up to 500 mM, overcoming the low activity and substrate inhibition issues of the wild-type TtherAmDH.
Q: How is the expensive cofactor NADH managed in this process to ensure cost efficiency?
A: The process employs a coupled enzyme system where formate dehydrogenase regenerates NADH in situ using ammonium formate as a sacrificial donor, drastically reducing the required amount of external cofactor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-5-Methyl-2-Pyrrolidone Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced biocatalytic technology to deliver high-quality chiral intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of (S)-5-methyl-2-pyrrolidone meets the highest industry standards. Our commitment to technical excellence allows us to optimize the enzymatic process for maximum yield and cost-effectiveness, providing our clients with a competitive edge in their own supply chains. By partnering with us, you gain access to a robust and sustainable manufacturing platform that is ready to support your long-term commercial needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this enzymatic process for your manufacturing operations. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about your supply chain strategy. Contact us now to explore the possibilities of collaborating on the production of high-purity pharmaceutical intermediates using cutting-edge biocatalysis.
