Advanced Manufacturing of 2-Fluoro-5-Formylbenzonitrile for Olaparib Production
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with Poly ADP-ribose polymerase (PARP) inhibitors like Olaparib standing at the forefront of targeted cancer therapy. As demand for these life-saving medications grows, the efficiency and safety of their supply chains become paramount. Patent CN114907234A introduces a groundbreaking preparation method for 2-fluoro-5-formylbenzonitrile, a critical building block in the synthesis of Olaparib. This technical disclosure addresses long-standing industry pain points regarding toxicity, waste generation, and operational complexity associated with legacy synthetic routes. By shifting away from hazardous halogenation and cyanidation protocols, this innovation offers a streamlined pathway that aligns with modern green chemistry principles while maintaining high yield and purity standards essential for active pharmaceutical ingredient (API) manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-5-formylbenzonitrile has been plagued by significant safety and environmental hurdles that complicate large-scale production. One prevalent legacy route involves the bromination of 4-fluorobenzaldehyde followed by cyanidation, a process depicted in earlier patents such as WO201271684A1. This method necessitates the use of liquid bromine or oleum, both of which are highly corrosive, volatile, and pose severe risks to personnel and infrastructure. Furthermore, the subsequent cyanidation step requires cuprous cyanide at elevated temperatures, introducing extreme toxicity concerns and demanding specialized containment equipment. Another existing approach utilizes 2-fluoro-5-methylbenzonitrile as a starting material, relying on bromination in carbon tetrachloride, a solvent known for its ozone-depleting potential and high toxicity. These conventional pathways not only generate substantial amounts of hazardous three wastes but also suffer from poor selectivity and low yields, driving up the overall cost of goods and creating bottlenecks in the supply chain for downstream drug manufacturers.
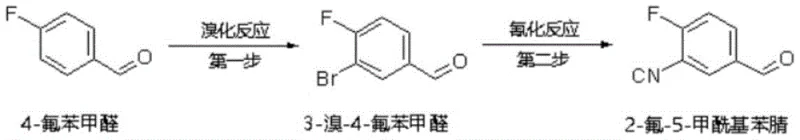
The Novel Approach
In stark contrast to these hazardous legacy methods, the technology disclosed in patent CN114907234A presents a robust and environmentally benign alternative. This novel strategy employs a three-step sequence starting from o-fluorobenzonitrile, a commercially abundant and cost-effective raw material. The process initiates with a chloromethylation reaction under acidic conditions, followed by a hydrolysis step to convert the intermediate into a benzyl alcohol derivative, and concludes with a selective oxidation to yield the target aldehyde. By replacing dangerous reagents like liquid bromine and cuprous cyanide with common industrial chemicals such as paraformaldehyde, zinc chloride, and sodium hypochlorite, the new route drastically reduces the risk profile of the manufacturing operation. The reaction conditions are notably mild, operating effectively within a temperature range of 0 to 100 degrees Celsius, which lowers energy consumption and relaxes the specifications for production reactors. This shift not only enhances operational safety but also simplifies the purification workflow, allowing for crude intermediates to be carried forward without extensive isolation, thereby improving overall process efficiency.
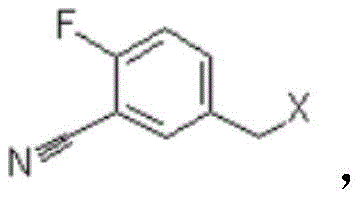
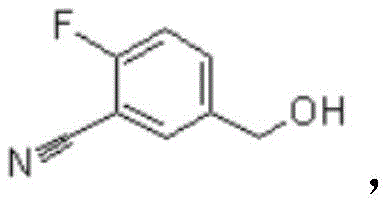
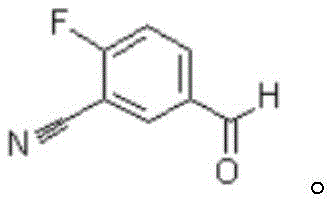
Mechanistic Insights into Chloromethylation and Selective Oxidation
The core of this innovative synthesis lies in the precise control of functional group transformations, beginning with the electrophilic aromatic substitution known as chloromethylation. In the first step, o-fluorobenzonitrile reacts with a source of formaldehyde, such as paraformaldehyde or a methanol solution, in the presence of a halide salt and a Lewis acid catalyst like zinc chloride. The acidic environment facilitates the generation of a chloromethyl cation species, which selectively attacks the aromatic ring at the position para to the nitrile group, driven by the electronic directing effects of the substituents. This step is critical as it installs the necessary carbon backbone for the subsequent aldehyde functionality without introducing heavy metals or complex protecting groups. The resulting Intermediate I, a chloromethyl derivative, is then subjected to hydrolysis in an aqueous alkaline medium. Here, the nucleophilic attack by hydroxide ions displaces the chloride atom, converting the reactive halide into a stable benzyl alcohol moiety (Intermediate II). This transformation is highly efficient and avoids the formation of side products that typically plague radical bromination processes.
The final stage involves the oxidation of the benzyl alcohol Intermediate II to the desired aldehyde, a transformation that requires careful selection of oxidants to prevent over-oxidation to the carboxylic acid. The patent highlights the efficacy of using mild oxidizing agents such as sodium hypochlorite, potentially catalyzed by TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl), or alternatively, pyridinium chlorochromate (PCC). When using the hypochlorite system, the reaction proceeds under biphasic conditions with rigorous temperature control, typically between 0 and 5 degrees Celsius during the addition phase, to ensure selectivity. This mechanistic pathway ensures that the sensitive nitrile group remains intact while the primary alcohol is cleanly converted to the formyl group. The ability to monitor reaction progress via HPLC and achieve conversion rates where starting material content drops below detectable limits underscores the robustness of this chemical design. By optimizing these mechanistic steps, the process achieves high purity levels exceeding 98 percent, meeting the stringent quality requirements for pharmaceutical intermediates used in the synthesis of potent anticancer agents.
How to Synthesize 2-Fluoro-5-Formylbenzonitrile Efficiently
Implementing this synthesis route requires adherence to specific procedural guidelines to maximize yield and safety. The process begins with the careful mixing of o-fluorobenzonitrile, paraformaldehyde, and a halide salt in concentrated sulfuric acid, ensuring thorough stirring to manage the exothermic nature of the chloromethylation. Following the formation of Intermediate I, the reaction mixture is quenched and extracted, after which the crude product is directly subjected to alkaline hydrolysis to form the alcohol. Finally, the oxidation step is performed under controlled cooling to maintain selectivity. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React o-fluorobenzonitrile with formaldehyde and a halide salt under acidic catalysis to form the chloromethyl intermediate.
- Hydrolyze the chloromethyl intermediate in an aqueous alkaline solution to generate the corresponding benzyl alcohol derivative.
- Oxidize the benzyl alcohol intermediate using a mild oxidizing agent like sodium hypochlorite or PCC to yield the final aldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing commodity chemicals like paraformaldehyde, sodium chloride, and sodium hypochlorite, manufacturers can mitigate the risks associated with sourcing specialized, high-cost, or regulated precursors such as cuprous cyanide or sulfuryl fluoride. This shift significantly enhances supply chain resilience, as these common reagents are widely available from multiple global suppliers, reducing the likelihood of production stoppages due to raw material shortages. Furthermore, the elimination of highly toxic substances simplifies regulatory compliance and reduces the overhead costs associated with hazardous waste disposal and personnel safety monitoring.
- Cost Reduction in Manufacturing: The economic impact of this new route is profound, primarily driven by the removal of expensive and dangerous reagents that require specialized handling and disposal protocols. Traditional methods involving liquid bromine and cuprous cyanide incur high costs for safety infrastructure, personal protective equipment, and waste treatment facilities. By replacing these with benign alternatives, the operational expenditure is substantially lowered. Additionally, the ability to use crude intermediates directly in subsequent steps without intermediate purification reduces solvent consumption and processing time, leading to further savings in utility and labor costs. The overall process efficiency is improved, allowing for higher throughput within existing facility footprints.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by the volatility of specialty chemical markets. This synthesis route relies on basic industrial chemicals that are produced in massive volumes globally, ensuring a stable and continuous supply. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities without the need for exotic high-pressure or high-temperature reactors. This flexibility allows for easier technology transfer and multi-site production strategies, safeguarding against regional disruptions. The reduced hazard profile also simplifies logistics and storage requirements, minimizing delays related to hazardous material transport regulations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this method is inherently designed for scalability. The absence of ozone-depleting solvents like carbon tetrachloride and toxic gases like sulfuryl fluoride ensures that the process aligns with increasingly stringent global environmental regulations. The reduction in three wastes—waste water, waste gas, and solid waste—lowers the environmental footprint of the manufacturing site. This compliance not only avoids potential fines and shutdowns but also enhances the corporate social responsibility profile of the manufacturer, which is increasingly important for partnerships with major multinational pharmaceutical companies committed to sustainable sourcing.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common inquiries regarding safety, scalability, and quality control based on the patent data. These insights are derived directly from the experimental examples and comparative analysis provided in the intellectual property documentation, offering a clear picture of the process capabilities.
Q: Why is the new synthesis route for 2-fluoro-5-formylbenzonitrile considered safer than traditional methods?
A: Traditional methods rely on highly toxic reagents such as liquid bromine, cuprous cyanide, carbon tetrachloride, and sulfuryl fluoride. The new patented route eliminates these hazardous substances, utilizing common chemicals like paraformaldehyde and sodium hypochlorite under mild conditions.
Q: What are the key cost drivers reduced in this manufacturing process?
A: Cost reduction is achieved by avoiding expensive and dangerous specialty reagents, simplifying equipment requirements due to mild reaction conditions (0-100°C), and enabling crude-to-crude processing which minimizes purification steps and solvent consumption.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses readily available industrial raw materials and avoids high-pressure or extreme temperature conditions, making it highly scalable and compliant with modern environmental and safety regulations for bulk pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-Formylbenzonitrile Supplier
As the demand for Olaparib and similar PARP inhibitors continues to rise, securing a stable supply of high-quality intermediates is critical for drug developers. NINGBO INNO PHARMCHEM stands ready to support this need with our advanced manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-fluoro-5-formylbenzonitrile meets the exacting standards required for API synthesis. We are committed to delivering consistency and reliability in every shipment.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our optimized routes can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency and innovation in your pharmaceutical manufacturing operations.
