Advanced Iridium Catalysis for Efficient Alpha-Borocarbonyl Manufacturing
Advanced Iridium Catalysis for Efficient Alpha-Borocarbonyl Manufacturing
The landscape of organoboron chemistry is undergoing a significant transformation driven by the urgent need for safer and more efficient synthetic methodologies in the pharmaceutical and fine chemical sectors. Patent CN110590821B introduces a groundbreaking iridium-catalyzed method for the synthesis of alpha-boronated carbonyl compounds, utilizing sulfur ylides as stable carbene precursors in a B-H bond insertion reaction with Lewis base borane adducts. This innovation addresses critical limitations in existing technologies by replacing hazardous diazo compounds with benign sulfur ylides, thereby enhancing both operational safety and process reliability. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards sustainable and scalable manufacturing of complex molecular entities that are pivotal in modern drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-boron bonds in alpha-borocarbonyl compounds has relied heavily on transition metal-catalyzed reactions involving alpha-diazocarbonyl compounds. While effective in laboratory settings, these traditional methods suffer from severe drawbacks that hinder their industrial applicability. Diazo compounds are inherently unstable, toxic, and possess a high potential for explosiveness, necessitating stringent safety protocols and specialized equipment that drastically increase capital expenditure and operational complexity. Furthermore, alternative approaches such as gold-catalyzed oxidative coupling of terminal alkynes have demonstrated low atom utilization and rely on commercially difficult-to-obtain catalysts, limiting their widespread adoption. Other methods involving N-heterocyclic carbene (NHC) borane complexes often result in generally low isolated yields, making them economically unviable for large-scale production where efficiency and cost-effectiveness are paramount concerns for supply chain stability.
The Novel Approach
The novel approach detailed in the patent data leverages the unique reactivity of sulfur ylides, which serve as safe and stable alternatives to diazo compounds while maintaining high reactivity under mild conditions. By employing an iridium catalyst, specifically [Ir(COD)Cl]2, in conjunction with Lewis base borane adducts, this method achieves efficient B-H bond insertion without the need for high-energy initiators or hazardous reagents. The process operates at a moderate temperature of 60°C, significantly reducing energy consumption compared to high-temperature alternatives. This methodology not only simplifies the synthetic route by utilizing easily obtainable raw materials derived from corresponding carboxylic acids but also demonstrates exceptional substrate applicability across various aromatic and aliphatic systems. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this route offers a streamlined pathway that minimizes waste generation and maximizes overall process efficiency.
Mechanistic Insights into Iridium-Catalyzed B-H Bond Insertion
The core of this technological advancement lies in the precise mechanistic orchestration of the iridium-catalyzed B-H bond insertion reaction. The catalytic cycle initiates with the activation of the sulfur ylide by the iridium center, generating a reactive metal-carbenoid species in situ. This transient intermediate then undergoes a concerted insertion into the B-H bond of the Lewis base borane adduct, facilitated by the electronic properties of the iridium complex. The presence of specific additives, such as copper difluoride or cuprous cyanide, plays a crucial role in stabilizing the transition state and enhancing the turnover frequency of the catalyst. This synergistic interaction ensures high conversion rates and minimizes side reactions, resulting in the formation of the desired alpha-borocarbonyl product with high fidelity. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrate classes.
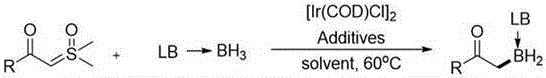
Furthermore, the amphiphilic nature of the resulting alpha-borocarbonyl compounds makes them versatile building blocks for subsequent functionalization, including Suzuki-Miyaura cross-coupling and oxidation reactions. The ability to control the stereochemistry and regioselectivity of the insertion through ligand modulation on the iridium center provides a powerful tool for synthesizing chiral organoboron compounds, which are increasingly demanded in the development of novel therapeutic agents. The robustness of this catalytic system allows it to tolerate a wide range of functional groups, including esters, ethers, and halides, without compromising yield or purity. This broad tolerance is essential for the late-stage functionalization of complex molecules, enabling medicinal chemists to rapidly explore chemical space and identify potent drug candidates with improved pharmacokinetic profiles.
How to Synthesize Alpha-Boronated Carbonyl Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it accessible for both laboratory research and industrial production. The standard procedure involves the sequential addition of the sulfur ylide, Lewis base borane adduct, iridium catalyst, and selected additive into a reaction vessel containing an appropriate solvent such as chlorobenzene or 1,2-dichloroethane. The mixture is then heated to 60°C and stirred for approximately three hours, after which the reaction progress is monitored via thin-layer chromatography (TLC). Upon completion, the workup procedure is straightforward, involving dilution with ethyl acetate, solvent removal under reduced pressure, and purification via silica gel column chromatography. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to assist technical teams in replicating these results.
- Sequentially add the sulfur ylide compound, Lewis base borane adduct, iridium catalyst ([Ir(COD)Cl]2), additive (e.g., CuF2), and solvent (e.g., chlorobenzene) into a reaction vessel.
- Heat the reaction mixture to 60°C and stir continuously for approximately 3 hours to ensure complete conversion monitored by TLC.
- Upon completion, dilute with ethyl acetate, remove solvent under reduced pressure, and purify the residue via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed sulfur ylide methodology offers profound advantages for procurement and supply chain management within the fine chemical industry. The substitution of hazardous diazo compounds with stable sulfur ylides fundamentally alters the risk profile of the manufacturing process, leading to significant reductions in insurance costs, safety compliance expenditures, and waste disposal fees. This shift not only enhances the overall safety of the production facility but also ensures a more consistent and reliable supply of raw materials, as sulfur ylides can be synthesized from abundant carboxylic acids without the need for specialized storage conditions. For supply chain heads, this translates to reduced lead times and minimized risk of production stoppages due to regulatory scrutiny or raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and hazardous reagents, coupled with the use of earth-abundant iridium catalysts that operate at low loadings. By avoiding the complex safety infrastructure required for handling explosive diazo compounds, manufacturers can achieve substantial cost savings in facility maintenance and operational overhead. Additionally, the high yields reported in the patent examples indicate excellent atom economy, reducing the amount of raw material wasted and lowering the cost of goods sold (COGS). This efficiency is critical for maintaining competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable and easily accessible starting materials ensures a robust supply chain that is less susceptible to disruptions. Unlike specialized alkynes or unstable diazo precursors that may have limited suppliers, carboxylic acids and borane adducts are commodity chemicals available from multiple sources worldwide. This diversification of the supply base mitigates the risk of single-source dependency and price volatility. Furthermore, the mild reaction conditions allow for flexible production scheduling and easier integration into existing manufacturing lines, enhancing the overall agility of the supply network to respond to market demands.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the absence of extreme conditions make this process highly amenable to scale-up from kilogram to multi-ton quantities. The reduced generation of hazardous byproducts aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes and reducing the environmental footprint of chemical manufacturing. This sustainability aspect is becoming a key differentiator for suppliers seeking to partner with major pharmaceutical companies that prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium-catalyzed synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of adopting this novel methodology for the production of alpha-boronated carbonyl compounds. Understanding these details is essential for stakeholders evaluating the potential integration of this process into their existing portfolios.
Q: Why are sulfur ylides preferred over diazo compounds for this synthesis?
A: Sulfur ylides are significantly safer, stable, and easier to prepare from corresponding carboxylic acids compared to diazo compounds, which are toxic, unstable, and potentially explosive. This switch enhances operational safety and simplifies raw material sourcing.
Q: What are the key advantages of the iridium-catalyzed method described in CN110590821B?
A: The method offers mild reaction conditions (60°C), high yields (up to 97% in examples), and excellent substrate applicability. It avoids the harsh conditions and low atom utilization associated with previous gold-catalyzed or NHC-borane methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of stable raw materials, simple operational steps, and mild temperatures makes this process highly scalable. The elimination of hazardous diazo reagents further reduces regulatory and safety barriers for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Boronated Carbonyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iridium-catalyzed sulfur ylide methodology in advancing the synthesis of high-value organoboron intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality and volume regardless of their project stage. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-boronated carbonyl compounds delivered meets the highest industry standards for pharmaceutical and fine chemical applications.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cutting-edge technology for their next-generation projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecular targets, along with specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern chemical synthesis and secure a competitive advantage through superior process efficiency and supply chain resilience.
