Advanced Biocatalytic Resolution of Chiral Amyl Alcohol for Industrial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-value chiral building blocks, and patent CN100344766C presents a transformative approach to the synthesis of optically active amyl alcohol. This specific intellectual property discloses a novel biocatalytic method that utilizes vinyl acetate as both a substrate and solvent to replace traditional, costly acyl donors like tributyrin. By leveraging porcine pancreatic lipase (PPL) as the biocatalyst, this technology enables the kinetic resolution of racemic 2-methyl-1-butanol with exceptional selectivity and operational simplicity. For R&D directors and procurement specialists alike, this represents a critical shift away from high-energy separation processes toward a more sustainable, cost-effective manufacturing paradigm. The ability to produce (S)-2-methyl-1-butanol and its corresponding chiral esters with high optical purity under mild conditions addresses long-standing bottlenecks in the supply chain for liquid crystal materials and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of chiral amyl alcohol via enzymatic resolution has relied heavily on tributyrin as the acyl donor substrate. While effective in laboratory settings, this conventional approach suffers from severe economic and engineering drawbacks when scaled to commercial production levels. Tributyrin is an expensive raw material that can account for more than 80% of the total raw material cost, creating a fragile cost structure vulnerable to market fluctuations. Furthermore, tributyrin possesses a very high boiling point, which necessitates the use of high-vacuum distillation equipment to separate the reaction mixture after biocatalysis. This requirement for high vacuum not only increases capital expenditure (CAPEX) for specialized equipment but also significantly drives up operational expenditure (OPEX) due to high energy consumption and complex maintenance protocols. These factors combined make the traditional tributyrin-based route less competitive for large-scale manufacturing of fine chemicals.
The Novel Approach
In stark contrast, the methodology outlined in patent CN100344766C introduces vinyl acetate as a superior alternative substrate that fundamentally alters the economic equation of chiral alcohol production. Vinyl acetate is a widely available commodity chemical with a significantly lower cost profile compared to tributyrin, immediately reducing the raw material burden on the manufacturing process. More importantly, its physical properties allow for separation and purification to be conducted under normal atmospheric pressure at temperatures between 60°C and 120°C. This eliminates the need for complex high-vacuum systems, simplifying the plant design and reducing energy consumption drastically. The reaction scheme below illustrates how the lipase selectively acylates one enantiomer while leaving the other intact, driven by the irreversible tautomerization of vinyl alcohol to acetaldehyde.
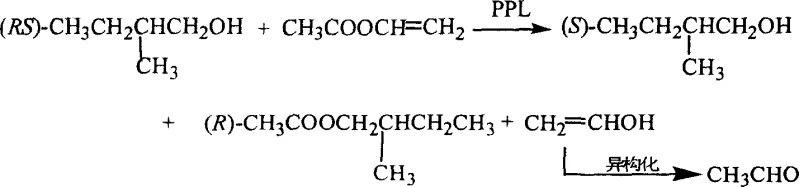
This novel approach not only lowers costs but also enhances the overall yield and purity profile by enabling easier recovery of unreacted vinyl acetate for reuse in subsequent batches, creating a closed-loop system that minimizes waste and maximizes resource efficiency for the reliable chiral amyl alcohol supplier.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core of this technology lies in the stereoselective nature of porcine pancreatic lipase (PPL), which acts as a highly specific biological catalyst to differentiate between the enantiomers of racemic 2-methyl-1-butanol. In this transesterification mechanism, the enzyme preferentially binds to the hydroxyl group of the (S)-enantiomer (or the (R)-enantiomer depending on specific enzyme batch characteristics) to form an acyl-enzyme intermediate with the vinyl acetate. This intermediate then collapses to release the chiral ester product and regenerate the free enzyme. A critical thermodynamic driver in this system is the fate of the leaving group; the vinyl alcohol generated during the reaction is unstable and rapidly undergoes keto-enol tautomerization to form acetaldehyde. This irreversible conversion prevents the reverse hydrolysis reaction, effectively pushing the equilibrium towards the formation of the ester product and ensuring high conversion rates without the need for excessive reagent loading.
Controlling the optical purity of the final product requires precise management of the reaction conversion rate, typically halted between 40% and 55% conversion to maximize the enantiomeric excess (e.e.%). The patent data indicates that stopping the reaction at these specific checkpoints allows for the isolation of (S)-2-methyl-1-butanol with an e.e.% ranging from 80% to 98%. In scenarios where the enzyme exhibits reversed specificity—preferentially acylating the (S)-alcohol to form the (S)-ester—the process includes a robust remediation step involving alkaline hydrolysis. By adjusting the pH to greater than 13 using sodium or potassium hydroxide, the unwanted (S)-ester can be quantitatively converted back into the desired (S)-alcohol. This flexibility in handling enzyme variability ensures consistent product quality, a crucial factor for maintaining stringent purity specifications in pharmaceutical intermediates manufacturing.
How to Synthesize (S)-2-Methyl-1-butanol Efficiently
The synthesis protocol described in the patent offers a streamlined pathway that balances reaction kinetics with downstream processing ease. The process begins by mixing racemic 2-methyl-1-butanol with porcine pancreatic lipase and vinyl acetate in a molar ratio of at least 1.2:1. The reaction is maintained at a mild temperature range of 30°C to 45°C, which preserves enzyme activity while providing sufficient thermal energy for the transformation. Monitoring the reaction progress is critical, and once the conversion reaches the optimal window of 40-55%, the biocatalyst is removed via filtration. The detailed standardized synthesis steps for implementing this route in a pilot or commercial plant are provided in the guide below.
- Mix racemic 2-methyl-1-butanol with porcine pancreatic lipase (PPL) and vinyl acetate (molar ratio ≥ 1.2) at 30-45°C.
- Monitor the reaction progress until conversion reaches 40-55%, then stop the reaction to maximize optical purity.
- Filter off the enzyme and perform rectification at 60-120°C under normal pressure to separate the product from unreacted substrates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from tributyrin to vinyl acetate represents a strategic opportunity to de-risk the supply of critical chiral intermediates. The primary advantage lies in the substantial reduction of raw material costs, as vinyl acetate is a bulk chemical with a stable global supply chain, unlike the more niche and expensive tributyrin. Additionally, the ability to perform distillation under normal pressure removes the dependency on specialized high-vacuum infrastructure, which reduces both the initial capital investment required for plant setup and the ongoing maintenance costs associated with vacuum pumps and seals. This simplification of the unit operations directly translates to lower manufacturing overheads and improved margin potential for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The replacement of high-cost tributyrin with inexpensive vinyl acetate dramatically lowers the direct material cost per kilogram of product. Furthermore, the unreacted vinyl acetate can be easily recovered and recycled due to its volatility, minimizing raw material waste. The elimination of high-vacuum distillation also results in significant energy savings, as normal pressure rectification requires less intensive utility support. These combined factors create a leaner cost structure that is more resilient to market volatility and provides a competitive pricing advantage.
- Enhanced Supply Chain Reliability: Vinyl acetate is a commodity chemical produced on a massive scale globally, ensuring a consistent and reliable supply source that is less prone to shortages compared to specialty esters. The simplified process conditions (30-45°C, normal pressure) reduce the risk of equipment failure and unplanned downtime, leading to more predictable production schedules. This reliability is essential for reducing lead time for high-purity chiral compounds, allowing downstream customers to maintain tighter inventory controls and respond faster to market demands.
- Scalability and Environmental Compliance: The process generates acetaldehyde as a byproduct, which is volatile and can be captured or treated using standard abatement technologies, simplifying waste management compared to heavy high-boiling residues. The mild reaction conditions and aqueous workup options for enzyme removal align well with green chemistry principles, reducing the environmental footprint of the manufacturing site. This scalability ensures that the commercial scale-up of complex chiral intermediates can be achieved smoothly from pilot batches to multi-ton annual production without encountering significant engineering barriers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is vital for technical teams assessing the integration of this method into existing production lines.
Q: Why is vinyl acetate preferred over tributyrin for this resolution?
A: Vinyl acetate is significantly cheaper and has a lower boiling point than tributyrin. This allows for product separation under normal pressure distillation rather than requiring energy-intensive high vacuum systems, drastically reducing operational costs.
Q: What optical purity can be achieved with this method?
A: According to the patent data, the process can achieve an optical purity (e.e.%) of 80-98% for (S)-2-methyl-1-butanol and 85-97% for the corresponding chiral ester, depending on reaction control and separation efficiency.
Q: How is the byproduct acetaldehyde handled?
A: The vinyl alcohol produced during transesterification spontaneously isomerizes into acetaldehyde. This volatile byproduct can be easily collected and removed during the distillation process, simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-Methyl-1-butanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality chiral building blocks for the next generation of pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-2-methyl-1-butanol meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to offer customized solutions that optimize both performance and cost for our partners.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic resolution technology can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this vinyl acetate-based route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your supply chain is built on a foundation of reliability and innovation.
