Scalable Biocatalytic Production of High-Purity (R)-1-(3-Trifluoromethylphenyl) Ethanol for Neuroprotective Drug Synthesis
Scalable Biocatalytic Production of High-Purity (R)-1-(3-Trifluoromethylphenyl) Ethanol for Neuroprotective Drug Synthesis
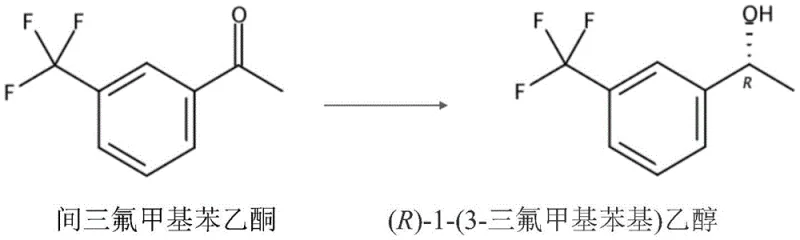
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and high-purity chiral intermediates has led to significant advancements in biocatalytic technologies, as exemplified by the groundbreaking methodology detailed in Chinese Patent CN112176019B. This patent discloses a novel and highly effective method for preparing (R)-1-(3-trifluoromethylphenyl) ethanol, a critical chiral building block used in the synthesis of neuroprotective compounds designed to treat cerebral ischemia and central nervous system injuries. The core innovation lies in the utilization of recombinant Escherichia coli whole cells expressing a specific short-chain dehydrogenase (LXCAR) derived from Leifsonia sp., combined with a unique reaction medium system incorporating deep eutectic solvents. This approach not only addresses the longstanding challenges of low substrate tolerance in traditional biocatalysis but also ensures exceptional stereochemical control, delivering products with an enantiomeric excess (e.e.) value exceeding 99.9%. For global procurement teams and R&D directors seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift towards greener, more cost-effective manufacturing processes that do not compromise on quality or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohols like (R)-1-(3-trifluoromethylphenyl) ethanol has relied heavily on chemical asymmetric reduction methods, which often involve the use of complex chiral ligands coordinated with transition metals such as ruthenium or rhodium. While these chemical routes can be effective, they suffer from significant drawbacks that impact both cost and environmental sustainability, including the requirement for stringent anhydrous conditions, the use of hazardous reducing agents like borohydrides, and the inevitable contamination of the final product with trace heavy metals. Furthermore, earlier attempts at biocatalytic reduction using wild-type microorganisms or less optimized strains were plagued by poor substrate tolerance, typically limiting the substrate concentration to extremely low levels such as 5 mM or 10 mM to avoid enzyme inhibition and cell toxicity. These low concentrations necessitate the processing of vast volumes of aqueous solvent to produce meaningful quantities of product, leading to excessive energy consumption during downstream separation and purification, thereby drastically inflating the overall manufacturing costs and complicating the supply chain logistics for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach described in Patent CN112176019B leverages the power of metabolic engineering and green chemistry principles to overcome previous barriers. By employing recombinant E. coli BL21(DE3)/pET28a(+)-LXCAR as a robust whole-cell biocatalyst, the process achieves high catalytic efficiency under mild physiological conditions, specifically at temperatures between 20°C and 40°C and a neutral pH range. The true breakthrough, however, is the strategic incorporation of deep eutectic solvents (DES), such as Choline Chloride:Lysine, into the reaction medium. These solvents act as permeabilizing agents that modify the microbial cell membrane, facilitating better mass transfer of the hydrophobic substrate into the cell while simultaneously reducing the inhibitory effects of both the substrate and the product on the enzyme. This innovation allows the reaction to operate at substantially higher substrate concentrations, reaching up to 400 mM, which translates to a massive increase in space-time yield and a significant reduction in the volume of waste water generated, making it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Biocatalytic Asymmetric Reduction
The mechanistic foundation of this process rests on the highly specific activity of the LXCAR short-chain dehydrogenase/reductase enzyme expressed within the recombinant bacterial host. This enzyme utilizes NADPH as a cofactor to stereoselectively reduce the carbonyl group of m-trifluoromethylacetophenone, transferring a hydride ion specifically to the re-face of the ketone to generate the desired (R)-alcohol configuration with near-perfect fidelity. To sustain this catalytic cycle without the prohibitive cost of adding stoichiometric amounts of expensive cofactors, the system employs an auxiliary substrate, preferably isopropanol, which serves as a hydrogen donor for the in-situ regeneration of NADPH from NADP+. This cofactor recycling mechanism is crucial for economic viability, as it allows a catalytic amount of the cofactor to drive the conversion of large quantities of substrate, effectively decoupling the cost of the redox equivalent from the scale of the production run and ensuring that the process remains economically competitive against traditional chemical synthesis routes.
Beyond the enzymatic mechanism, the role of the deep eutectic solvent in modulating the reaction environment cannot be overstated in terms of impurity control and process stability. The hydrogen bond network formed between the hydrogen bond acceptor (Choline Chloride) and the hydrogen bond donor (Lysine) creates a unique microenvironment that stabilizes the enzyme structure against denaturation while enhancing the solubility of the fluorinated aromatic substrate which is otherwise poorly soluble in pure aqueous buffers. By improving the homogeneity of the reaction mixture, the DES minimizes the formation of side products that often arise from non-enzymatic background reactions or substrate aggregation, thereby simplifying the downstream purification process. This results in a cleaner crude product profile, reducing the burden on chromatographic separation steps and ensuring that the final active pharmaceutical ingredient (API) intermediate meets the rigorous purity specifications required by regulatory bodies for neuroprotective drug candidates.
How to Synthesize (R)-1-(3-Trifluoromethylphenyl) Ethanol Efficiently
The implementation of this biocatalytic route requires precise control over fermentation parameters and reaction conditions to maximize the expression of the LXCAR enzyme and its subsequent activity in the conversion step. The process begins with the cultivation of the recombinant strain in a defined medium supplemented with kanamycin to maintain plasmid stability, followed by induction with IPTG to trigger high-level protein expression. Once the wet cells are harvested, they are suspended in a phosphate buffer system optimized for pH and ionic strength, to which the substrate, co-substrate, and the critical deep eutectic solvent additive are introduced. The detailed standard operating procedures, including specific incubation times, agitation speeds, and work-up protocols necessary to achieve the reported yields of over 74% at high substrate loading, are outlined in the comprehensive guide below for technical teams looking to replicate or license this technology.
- Prepare the biocatalyst by fermenting recombinant Escherichia coli BL21(DE3)/pET28a(+)-LXCAR, inducing expression with IPTG, and harvesting the wet cells via centrifugation.
- Construct the conversion system by mixing the wet cells, m-trifluoromethylacetophenone substrate, isopropanol co-substrate, and Choline Chloride: Lysine deep eutectic solvent in a phosphate buffer.
- Incubate the reaction mixture at 30°C with shaking, then perform extraction and purification to isolate the final (R)-1-(3-trifluoromethylphenyl) ethanol product with >99.9% e.e.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers profound strategic advantages that extend far beyond simple yield metrics, fundamentally altering the cost structure and risk profile of the supply chain. By shifting from a chemical synthesis reliant on precious metals and hazardous reagents to a fermentation-based process using renewable biological catalysts, manufacturers can eliminate the volatile cost fluctuations associated with rare earth metals and the substantial expenses linked to hazardous waste disposal and regulatory compliance. The ability to run reactions at high substrate concentrations means that less water and solvent are required per kilogram of product, which directly correlates to reduced utility costs for heating, cooling, and distillation, thereby driving down the overall cost of goods sold (COGS) and enhancing the margin potential for the final neuroprotective drug product.
- Cost Reduction in Manufacturing: The elimination of expensive chiral chemical catalysts and the removal of heavy metal scavenging steps significantly lower the raw material and processing costs. Furthermore, the in-situ cofactor regeneration system removes the need for purchasing stoichiometric amounts of NADPH, which is traditionally one of the most expensive components in enzymatic synthesis, resulting in substantial long-term savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Utilizing recombinant E. coli as the catalyst ensures a consistent and renewable source of biocatalytic activity that is not subject to the geopolitical supply risks often associated with mined transition metals. The robustness of the whole-cell system allows for the production of the catalyst on-demand through standard fermentation infrastructure, guaranteeing supply continuity and reducing the lead time for high-purity pharmaceutical intermediates even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The process operates under mild, aqueous conditions that are inherently safer and easier to scale from laboratory benchtop to multi-ton industrial reactors without the need for specialized high-pressure or cryogenic equipment. Additionally, the use of biodegradable deep eutectic solvents and the reduction of organic solvent waste align perfectly with modern green chemistry mandates, simplifying environmental permitting and reducing the carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process, derived directly from the experimental data and beneficial effects reported in the patent documentation. These insights are intended to clarify the operational feasibility and quality advantages of the method for stakeholders evaluating its integration into their existing manufacturing portfolios. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: What is the primary advantage of using deep eutectic solvents in this biocatalytic process?
A: The integration of deep eutectic solvents, specifically Choline Chloride:Lysine, significantly enhances the solubility of the hydrophobic substrate and improves microbial cell membrane permeability. This dual action allows the reaction to tolerate much higher substrate concentrations (up to 400 mM) while maintaining excellent catalytic efficiency and yield, overcoming the mass transfer limitations often seen in traditional aqueous biocatalysis.
Q: What level of optical purity can be achieved with this recombinant E. coli method?
A: This biocatalytic process demonstrates exceptional stereoselectivity, consistently achieving an enantiomeric excess (e.e.) value of greater than 99.9%. This high level of chiral purity is critical for pharmaceutical applications, particularly for neuroprotective agents where the biological activity is strictly dependent on the (R)-configuration of the molecule.
Q: How does this biological method compare to traditional chemical reduction in terms of scalability?
A: Unlike traditional chemical reductions that often require expensive chiral ligands, harsh reducing agents, and complex heavy metal removal steps, this biological method operates under mild conditions (30°C, atmospheric pressure) using renewable whole-cell catalysts. The elimination of toxic reagents and the robustness of the recombinant E. coli strain make the process inherently safer and significantly easier to scale up for commercial manufacturing without compromising environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-(3-Trifluoromethylphenyl) Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation neuroprotective therapies, and we possess the technical expertise to bring this advanced biocatalytic process to commercial reality. Our state-of-the-art R&D facilities and pilot plants are equipped to handle complex enzymatic transformations, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications. Our rigorous QC labs utilize advanced chiral chromatography and spectroscopic methods to ensure that every batch of (R)-1-(3-trifluoromethylphenyl) ethanol meets the >99.9% e.e. standard required for clinical and commercial applications, providing our partners with the confidence they need to advance their drug development pipelines.
We invite global pharmaceutical companies and contract research organizations to collaborate with us to leverage this innovative technology for their specific project needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that compares this biocatalytic route against your current supply chain methods, as well as obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us partner with you to optimize your supply chain, reduce costs, and accelerate the delivery of life-saving medicines to patients worldwide through superior chemical manufacturing excellence.
