Revolutionizing Penicillin Sulfoxide Ester Production via Continuous Flow Catalysis for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical antibiotic intermediates, and patent CN114437110A presents a groundbreaking advancement in this domain. This intellectual property details a novel application of supported catalysts specifically designed for the continuous preparation of penicillin sulfoxide ester, a pivotal precursor in the manufacture of cephalosporin antibiotics. The core innovation lies in the utilization of ordered mesoporous material SBA-15 as a carrier for active metal salt components, which fundamentally transforms the oxidation process of penicillin G potassium esters. By shifting from traditional batch operations to a continuous flow regime, this technology addresses long-standing challenges regarding selectivity, catalyst recovery, and environmental impact. The reported conversion rates reaching 99.5% and selectivity hitting 100% demonstrate a level of precision that is rarely achieved in conventional oxidation methodologies. For global supply chain leaders, this represents not just a chemical improvement but a strategic opportunity to secure a more reliable source of high-purity pharmaceutical intermediates.
Furthermore, the integration of hydrogen peroxide as the oxidant in conjunction with this heterogeneous catalyst system eliminates the reliance on hazardous peracids that have historically plagued this synthesis route. The ability to recycle the catalyst after simple activation processes underscores the economic viability of this approach, reducing the overall consumption of precious metal resources. As we delve deeper into the technical specifics, it becomes evident that this patent offers a robust framework for modernizing the production infrastructure of beta-lactam intermediates. The implications for cost reduction and process safety are profound, positioning this technology as a cornerstone for next-generation antibiotic manufacturing facilities aiming to meet stringent regulatory standards while optimizing operational expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of penicillin sulfoxide ester has relied heavily on methods involving peracetic acid or homogeneous metal catalysts, both of which present significant drawbacks for large-scale industrial application. The peracetic acid oxidation method, while effective in converting the penicillin skeleton, necessitates the use of large quantities of alkali to neutralize the acetic acid byproduct during post-treatment. This neutralization step generates substantial volumes of industrial wastewater and waste salts, creating a heavy burden on environmental compliance systems and driving up disposal costs. Moreover, the quenching of excessive peracetic acid typically requires sodium bisulfite, further complicating the waste stream management. On the other hand, methods utilizing vanadium pentoxide or sodium tungstate as homogeneous catalysts suffer from the inability to easily separate the metal residues from the final product. These residual metals can adversely affect product quality and pose toxicity risks, requiring additional purification steps that lower overall yield and increase processing time. The inability to recycle these homogeneous catalysts also leads to resource wastage and higher raw material costs, making the traditional approaches less sustainable in the long run.
The Novel Approach
In stark contrast, the novel approach outlined in the patent leverages a supported catalyst system that fundamentally resolves the separation and recycling issues inherent in previous methodologies. By immobilizing active metal salts onto the structured pores of SBA-15 mesoporous material, the catalyst becomes a heterogeneous entity that can be physically separated from the reaction mixture through simple filtration. This structural design ensures that the active sites remain accessible for the oxidation of the thioether group while preventing metal leaching into the product stream. The continuous flow configuration allows for precise control over reaction parameters such as temperature and residence time, which is critical for maintaining the high selectivity observed in the experimental data. Unlike batch processes where heat transfer limitations can lead to hot spots and side reactions, the continuous flow reactor ensures uniform reaction conditions throughout the process. This results in a dramatic reduction in over-oxidation products and impurities, simplifying the downstream purification process and enhancing the overall purity of the penicillin sulfoxide ester. The combination of heterogeneous catalysis and continuous processing creates a synergistic effect that boosts efficiency and sustainability simultaneously.
Mechanistic Insights into SBA-15 Supported Catalyst Oxidation
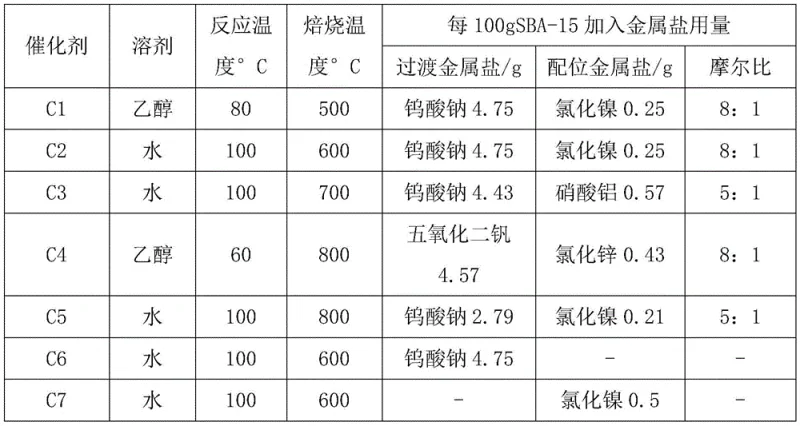
The mechanistic superiority of this system stems from the unique physicochemical properties of the SBA-15 support and its interaction with the transition metal salts. SBA-15 possesses a two-dimensional hexagonal through-hole structure with a large specific surface area and uniform pore diameter distribution, which facilitates the high dispersion of active metal components. When impregnated with metal salts such as sodium tungstate or nickel chloride, the resulting catalyst exhibits enhanced thermal stability and hydrothermal resistance, crucial for maintaining activity under oxidative conditions. The contact catalytic oxidation mechanism involves the activation of hydrogen peroxide by the supported metal ions, generating reactive oxygen species that selectively oxidize the sulfur atom in the penicillin thioether moiety. This activation process is highly efficient due to the confinement effects within the mesopores, which concentrate the reactants near the active sites. After the oxidation is complete, the metal ions are reactivated by hydrogen peroxide for the next cycle, ensuring sustained catalytic performance over multiple runs. This regenerative capability is a key factor in achieving the reported stability over ten recycling cycles without significant degradation in conversion or selectivity.
Impurity control is another critical aspect where this mechanistic design excels, particularly in preventing the formation of sulfone byproducts which are common in uncontrolled oxidation reactions. The precise tuning of the molar ratio between the transition metal salt and the coordination metal salt allows for fine adjustment of the catalyst's electronic properties, optimizing its redox potential for the specific transformation. By maintaining the reaction temperature within a narrow window of 40-60°C and controlling the residence time to mere minutes, the system minimizes the exposure of the sensitive beta-lactam ring to harsh oxidative conditions. This gentle yet effective oxidation environment preserves the structural integrity of the penicillin nucleus while ensuring complete conversion of the sulfide to the sulfoxide. The absence of transitional oxidation products means that the resulting intermediate meets rigorous pharmaceutical standards with minimal need for extensive chromatographic purification. Such high levels of purity are essential for downstream coupling reactions in cephalosporin synthesis, where impurities can propagate and compromise the efficacy of the final drug product.
How to Synthesize Penicillin Sulfoxide Ester Efficiently
Implementing this synthesis route requires a systematic approach to catalyst preparation and reactor configuration to fully realize the benefits of the continuous flow technology. The process begins with the meticulous preparation of the supported catalyst, where metal salts are dissolved in a solvent under nitrogen protection before impregnation onto the SBA-15 carrier. Following impregnation, the material undergoes a controlled roasting process to stabilize the active phases, ensuring optimal dispersion and adhesion to the support structure. Once the catalyst is ready, it is introduced into the continuous flow system as a slurry alongside the penicillin G potassium esterified substrate. The detailed standardized synthesis steps involve precise metering of hydrogen peroxide and quenching agents to maintain the stoichiometric balance required for high selectivity. Operators must monitor temperature and pressure sensors closely to ensure the reaction stays within the safe and effective operating envelope defined by the patent specifications. Adhering to these protocols guarantees consistent product quality and maximizes the lifespan of the catalyst through proper recovery and regeneration cycles.
- Prepare the supported catalyst by impregnating SBA-15 mesoporous material with transition metal salts and coordination metal salts, followed by calcination.
- Feed penicillin G potassium ester and the catalyst slurry into the first reactor while introducing hydrogen peroxide for continuous flow oxidation.
- Quench the oxidation reaction liquid with sodium bisulfite solution in a second reactor, then filter to recover the catalyst and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this continuous flow technology offers transformative advantages that directly impact the bottom line and operational resilience. The shift to a heterogeneous catalyst system eliminates the need for complex and costly metal scavenging processes that are typically required when using homogeneous catalysts. This simplification of the downstream processing workflow translates into significant cost savings by reducing the consumption of auxiliary chemicals and minimizing waste treatment expenses. Furthermore, the ability to recycle the catalyst multiple times drastically reduces the frequency of catalyst replenishment, stabilizing raw material costs and insulating the supply chain from volatility in metal prices. The continuous nature of the process also enhances production throughput, allowing manufacturers to respond more agilely to market demand fluctuations without the batch-to-batch variability that often plagues traditional methods. These efficiencies collectively contribute to a more robust and cost-effective supply chain for critical antibiotic intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal removal steps and the reduction in waste salt generation lead to substantial operational cost reductions. By avoiding the use of peracetic acid, the process removes the need for large quantities of neutralizing alkalis, further lowering chemical procurement costs. The high selectivity of the reaction minimizes product loss during purification, effectively increasing the overall yield per unit of raw material input. Additionally, the energy efficiency of the continuous flow system, with its rapid reaction times and moderate temperature requirements, reduces utility consumption compared to prolonged batch heating and cooling cycles. These factors combine to create a leaner manufacturing process that delivers high-quality intermediates at a competitive cost structure.
- Enhanced Supply Chain Reliability: The robustness of the SBA-15 supported catalyst ensures consistent performance over extended periods, reducing the risk of production delays caused by catalyst deactivation or failure. The continuous flow setup allows for steady-state operation, which simplifies inventory management and enables just-in-time production strategies. Since the catalyst can be regenerated on-site, dependencies on external suppliers for fresh catalyst batches are minimized, enhancing supply security. The simplified post-treatment process also shortens the overall production cycle time, enabling faster turnaround from raw material intake to finished goods. This agility is crucial for maintaining continuity in the supply of essential pharmaceutical intermediates, especially during periods of high global demand or logistical disruptions.
- Scalability and Environmental Compliance: The modular nature of continuous flow reactors facilitates straightforward scale-up from pilot to commercial production without the engineering challenges associated with enlarging batch vessels. The process inherently generates less hazardous waste, aligning with increasingly stringent environmental regulations and corporate sustainability goals. By reducing the volume of wastewater and solid waste, facilities can lower their environmental footprint and avoid potential regulatory penalties. The use of hydrogen peroxide as a green oxidant further supports eco-friendly manufacturing practices, appealing to stakeholders who prioritize sustainability. This alignment with environmental standards not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible supplier in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oxidation technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on feasibility and performance. Understanding these aspects is vital for technical teams evaluating the integration of this process into existing manufacturing lines. The answers highlight the practical benefits and operational considerations that define the value proposition of this innovative synthetic route.
Q: What are the advantages of using SBA-15 supported catalysts over traditional homogeneous catalysts?
A: SBA-15 supported catalysts offer superior selectivity reaching 100% and allow for easy separation and recycling up to ten times without significant loss of activity, unlike homogeneous catalysts which generate difficult-to-remove metal residues.
Q: How does the continuous flow process impact environmental compliance?
A: The continuous flow method significantly reduces wastewater and waste salt generation compared to batch processes using peracetic acid, as it eliminates the need for large amounts of alkali neutralization and simplifies quenching procedures.
Q: Can this synthesis route be scaled for commercial API intermediate production?
A: Yes, the process is designed for scalability with reaction times as short as 0.5 to 3 minutes and stable operation under moderate pressure, making it highly suitable for large-scale commercial manufacturing of cephalosporin intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Penicillin Sulfoxide Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of experts has thoroughly analyzed the potential of the SBA-15 supported catalyst system and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and efficient. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying every batch against the highest industry standards. By leveraging our CDMO capabilities, clients can access this advanced technology without the need for significant capital investment in new infrastructure, accelerating their time to market for next-generation cephalosporins.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this continuous flow process can optimize your supply chain. Partnering with us means gaining access to a reliable supply of high-purity intermediates produced through sustainable and efficient methods. Let us help you navigate the complexities of modern chemical manufacturing and secure a competitive advantage through technological innovation. Contact us today to discuss how we can support your strategic goals with our advanced synthesis capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
