Revolutionizing N-Alkyl Phthalimide Production: A Solvent-Free Route for High-Purity Pharmaceutical Intermediates
Revolutionizing N-Alkyl Phthalimide Production: A Solvent-Free Route for High-Purity Pharmaceutical Intermediates
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. In this landscape, Patent CN112479975A emerges as a pivotal technological breakthrough, introducing a novel, solvent-free preparation method for N-alkyl phthalimide that fundamentally alters the economic and environmental calculus of production. Unlike traditional methodologies that rely heavily on toxic organic solvents such as xylene or toluene to facilitate dehydration, this innovation utilizes a direct reaction between phthalic anhydride and aqueous alkylamine solutions. By eliminating the need for auxiliary organic solvents, the process achieves exceptional yields exceeding 96% and purity levels surpassing 99%, while simultaneously simplifying the downstream purification workflow. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediates supplier, this technology represents a paradigm shift towards greener, more cost-effective manufacturing that aligns perfectly with modern sustainability mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-alkyl phthalimides has been plagued by significant inefficiencies inherent to solvent-dependent processes. The conventional approach typically involves the condensation of phthalimide salts with halogenated alkanes or the reaction of phthalic anhydride with amines in the presence of polar protic solvents like xylene or toluene. These traditional routes necessitate rigorous dehydration steps where organic solvents act as water carriers to drive the equilibrium toward the imide product. However, this reliance introduces severe drawbacks: the subsequent removal of these high-boiling organic solvents requires energy-intensive distillation, leading to substantial operational costs and potential product loss during separation. Furthermore, the inability to completely eliminate solvent residues often compromises the final purity, necessitating additional recrystallization steps that further erode overall yield. From a safety and environmental perspective, the handling and disposal of large volumes of flammable, toxic solvents create significant regulatory burdens and increase the risk profile of the manufacturing facility, making these legacy processes increasingly untenable for modern cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN112479975A offers a streamlined, solvent-free alternative that addresses these chronic pain points with elegant simplicity. By utilizing an aqueous solution of alkylamine directly with phthalic anhydride, the process leverages the reactivity of the amine in water to initiate ring-opening, followed by a controlled thermal dehydration that requires no external organic carrier. This approach not only simplifies the reaction setup by removing the need for solvent recovery systems but also drastically enhances the safety profile by eliminating flammable organic vapors from the reactor headspace. The absence of organic solvents means that the final product is free from solvent-related impurities, achieving purity specifications above 99% directly from the reactor discharge. This direct synthesis route significantly reduces the number of unit operations required, translating to lower capital expenditure for equipment and reduced energy consumption for heating and cooling cycles. For supply chain managers, this translates to a more robust and predictable production timeline, as the complexities associated with solvent management and recycling are entirely obviated, ensuring a steady flow of high-purity N-alkyl phthalimide.
Mechanistic Insights into Solvent-Free Imidization
The core of this technological advancement lies in the precise control of the two-step reaction mechanism: ring-opening followed by dehydration ring-closure. Initially, phthalic anhydride reacts with the alkylamine in the aqueous phase to form the corresponding amic acid intermediate. This step is exothermic and proceeds rapidly even at moderate temperatures, facilitated by the nucleophilic attack of the amine nitrogen on the carbonyl carbon of the anhydride. The use of an aqueous medium is critical here, as it helps dissolve the reactants and manage the heat of reaction without the need for dilution by organic solvents. Following the formation of the amic acid, the system undergoes a critical transformation where water is eliminated to close the imide ring. In traditional methods, this equilibrium is pushed forward by azeotropic distillation with solvents like xylene; however, in this novel process, the water is removed simply by raising the temperature to between 100°C and 200°C, often under reduced pressure or inert gas flow. This thermal dehydration is highly efficient because the reaction mixture itself becomes the medium, and as water evaporates, the concentration of reactants increases, driving the reaction to completion without the thermodynamic barriers imposed by solvent interactions.
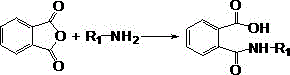
The mechanistic pathway ensures minimal formation of byproducts, which is crucial for maintaining the high purity required in pharmaceutical applications. The patent highlights the importance of excluding oxygen during the reaction, typically by purging with nitrogen, to prevent oxidative degradation which can lead to colored impurities. The dehydration step is particularly sensitive; if the temperature is too low, the ring closure is incomplete, but if too high, sublimation of the product can occur, reducing yield. The optimized protocol balances these factors, often employing a temperature ramp from ambient conditions up to 200°C. This controlled thermal profile ensures that the amic acid intermediate converts quantitatively to the desired N-alkyl phthalimide. The ability to recover excess amine from the distillate further enhances the atom economy of the process, allowing for the recycling of unreacted starting materials back into the batch, thereby minimizing waste and maximizing resource utilization in the commercial scale-up of complex heterocyclic intermediates.
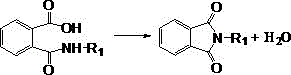
How to Synthesize N-Alkyl Phthalimide Efficiently
Implementing this solvent-free synthesis route requires careful attention to reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the charging of phthalic anhydride into a reactor equipped with heating and distillation capabilities, followed by the replacement of atmospheric air with an inert gas such as nitrogen to prevent oxidation. An aqueous solution of the desired alkylamine is then added, with the molar ratio carefully controlled between 1:1 and 1:10 depending on the specific amine reactivity and volatility. The mixture is heated to facilitate the initial ring-opening reaction, typically maintaining temperatures between 50°C and 120°C until a clear solution is obtained. Subsequently, the temperature is raised to the dehydration range of 100°C to 200°C, where water and excess amine are distilled off until no further condensate is observed, indicating the completion of the imidization. Detailed standardized synthetic steps and specific parameter optimizations for various alkyl chains are provided in the technical guide below.
- Charge phthalic anhydride into a reactor and replace air with inert gas (nitrogen) to prevent oxidation and discoloration.
- Add aqueous alkylamine solution (molar ratio 1: 1 to 1:10) and heat to 50-120°C to facilitate ring-opening and dissolution.
- Raise temperature to 100-200°C to distill off water and excess amine, driving the dehydration ring-closure to completion until no water evolves.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology offers compelling strategic advantages that extend far beyond simple chemical yield improvements. The elimination of organic solvents like xylene and toluene removes a significant cost center associated with solvent purchase, storage, recovery, and disposal. Traditional processes require extensive infrastructure for solvent recycling, including specialized distillation columns and storage tanks, which represent substantial capital investment and ongoing maintenance costs. By removing these requirements, the new process drastically simplifies the plant footprint and reduces the working capital tied up in solvent inventory. Furthermore, the regulatory burden associated with VOC (Volatile Organic Compound) emissions is significantly lowered, reducing compliance costs and minimizing the risk of environmental fines. This streamlined approach allows for faster batch turnover times, as the lengthy solvent stripping and drying phases are replaced by a more direct thermal dehydration, effectively increasing the overall throughput of the manufacturing facility without the need for additional hardware.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the total eradication of organic solvent costs. In traditional synthesis, solvents can account for a significant portion of the raw material bill, and their recovery is never 100% efficient, leading to continuous make-up costs. By switching to an aqueous system where water is the only liquid medium and is easily removed, the variable cost per kilogram of product drops precipitously. Additionally, the energy demand is lower because there is no need to heat and vaporize large volumes of high-boiling organic solvents; only water and excess amine need to be distilled. This energy efficiency translates directly into lower utility bills. The high yield of over 96% also means less raw material waste, further optimizing the cost structure and providing a competitive pricing advantage in the global market for high-purity N-alkyl phthalimide.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the new process. Traditional methods are susceptible to disruptions in the supply of specialty solvents or fluctuations in their prices, which can impact production schedules. The solvent-free method relies on commodity chemicals—phthalic anhydride and alkylamines—which are widely available and have stable supply chains. The reduced complexity of the process also means fewer potential points of failure; there are no solvent recovery units to break down or clog, leading to higher equipment availability and uptime. This reliability ensures consistent delivery schedules for downstream customers, a critical factor for pharmaceutical companies that operate on tight Just-In-Time manufacturing schedules. The ability to produce material with consistent quality batch after batch reduces the need for safety stock, allowing for leaner inventory management across the value chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat transfer and mass transfer in solvent systems. The solvent-free nature of this reaction mitigates many of these scale-up risks, as the viscosity and thermal properties of the reaction mixture are more manageable without the presence of viscous organic solvents. The process is inherently safer, with a lower risk of fire and explosion due to the absence of flammable organic vapors, which simplifies the permitting process for new or expanded production lines. From an environmental standpoint, the process is nearly zero-discharge regarding organic waste; the only effluent is water, and even the off-gases containing excess amine can be condensed and recycled. This green chemistry profile aligns with the increasingly stringent environmental regulations globally, future-proofing the production asset against tightening emission standards and enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this solvent-free synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs. The answers below clarify the operational advantages and quality benchmarks associated with this innovative manufacturing method.
Q: How does the solvent-free method improve product purity compared to traditional xylene reflux?
A: Traditional methods using xylene or toluene often leave trace organic solvent residues that are difficult to remove completely. The patented solvent-free route eliminates organic solvents entirely, relying only on water which is easily distilled off, resulting in purity levels consistently above 99% without complex purification steps.
Q: Can this process be scaled for industrial production of N-methylphthalimide?
A: Yes, the process is specifically designed for industrial scalability. It utilizes simple equipment (standard reactors), operates under manageable pressure and temperature conditions (up to 200°C), and avoids the safety hazards associated with large volumes of flammable organic solvents, making it ideal for commercial scale-up.
Q: What are the environmental benefits of this synthesis route?
A: The primary environmental benefit is the elimination of VOC (Volatile Organic Compound) emissions associated with organic solvents like xylene. The only byproduct is water, and excess amine can be recovered and recycled from the tail gas, creating a closed-loop, green manufacturing process with minimal waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl Phthalimide Supplier
The transition to solvent-free manufacturing represents a significant opportunity for the chemical industry to enhance both profitability and sustainability. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced technology to the global market. Our state-of-the-art facilities are equipped to handle the specific thermal and pressure requirements of this process, ensuring that every batch meets stringent purity specifications of >99%. With our rigorous QC labs and commitment to green chemistry principles, we are uniquely positioned to supply high-purity N-alkyl phthalimide that satisfies the most demanding regulatory requirements of the pharmaceutical and agrochemical sectors.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our solvent-free N-alkyl phthalimide can become a cornerstone of your production strategy, delivering value through superior quality and operational excellence.
