Advanced Biocatalytic Production of Chiral Alcohol Intermediates for Commercial Scale-Up
Advanced Biocatalytic Production of Chiral Alcohol Intermediates for Commercial Scale-Up
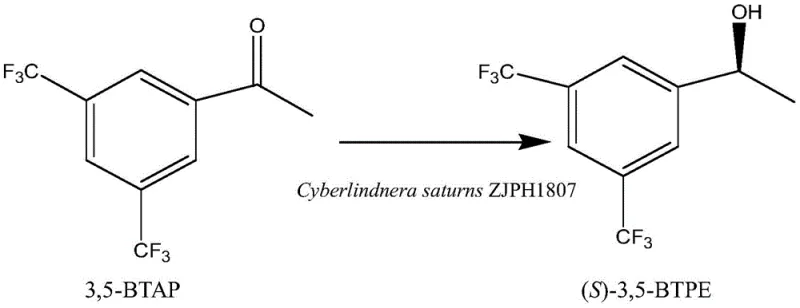
The pharmaceutical industry continuously seeks robust, sustainable, and cost-effective methodologies for synthesizing chiral building blocks, particularly for high-value Active Pharmaceutical Ingredients (APIs). A significant breakthrough in this domain is documented in patent CN112048538B, which details a novel biocatalytic method for preparing (S)-[3,5-bis(trifluoromethyl)phenyl]ethanol, a critical chiral intermediate for the synthesis of Aprepitant, a potent NK-1 receptor antagonist. This technology leverages the resting cells of the yeast strain Cyberlindnera saturns ZJPH1807 to achieve asymmetric reduction with exceptional stereo-selectivity. For R&D directors and procurement specialists, this patent represents a paradigm shift from traditional chemical synthesis to a greener, more efficient biological route that promises substantial improvements in both product quality and manufacturing economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-[3,5-bis(trifluoromethyl)phenyl]ethanol has relied heavily on chemical catalysis or less optimized biological routes. Conventional chemical methods often necessitate the use of expensive transition metal catalysts, such as ruthenium or rhodium complexes, which not only drive up raw material costs but also introduce significant environmental liabilities due to heavy metal waste. Furthermore, removing trace metal residues to meet stringent pharmaceutical purity standards requires complex and costly downstream purification steps. Existing biological methods reported in prior art have struggled with low substrate tolerance, often failing to maintain high conversion rates when substrate concentrations exceed 50 mM, thereby limiting volumetric productivity and increasing solvent usage per unit of product.
The Novel Approach
The methodology disclosed in CN112048538B overcomes these bottlenecks by utilizing a specialized whole-cell biocatalyst system. Unlike isolated enzyme systems that require expensive cofactor regeneration schemes, this approach employs resting cells of Cyberlindnera saturns ZJPH1807, which possess intrinsic metabolic pathways for in-situ cofactor regeneration using cheap auxiliary substrates like glucose. This innovation allows the reaction to proceed at significantly higher substrate concentrations, up to 128.07 g/L (500 mM), while maintaining an enantiomeric excess (e.e.) value of greater than 99.9%. The integration of Natural Deep Eutectic Solvents (NADES) and non-ionic surfactants further enhances mass transfer and enzyme stability, creating a highly efficient conversion system that is both economically and environmentally superior.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this technology lies in the stereoselective reduction of the ketone group in 3,5-bis(trifluoromethyl)acetophenone (3,5-BTAP) to the corresponding chiral alcohol. The Cyberlindnera saturns ZJPH1807 strain expresses specific alcohol dehydrogenases (ADHs) that exhibit high specificity for the pro-S face of the carbonyl group. In the presence of a co-substrate such as glucose, the cellular metabolism oxidizes the glucose to regenerate NADPH from NADP+, which serves as the essential hydride donor for the reduction reaction. This internal recycling mechanism eliminates the need for external addition of costly cofactors, a common economic barrier in enzymatic processes. The reaction environment is carefully buffered, typically using a phosphate system at pH 7.5, to maintain optimal enzyme activity and cell integrity throughout the conversion period.
Impurity control is inherently managed through the high selectivity of the biocatalyst. Chemical reduction methods often produce racemic mixtures or over-reduced byproducts, requiring difficult chiral separations. In contrast, the biological system described achieves an e.e. value of >99.9% directly from the reaction broth. The addition of additives like L-carnitine:lysine (1:2 molar ratio) and Tween-80 plays a crucial mechanistic role by improving the solubility of the hydrophobic fluorinated substrate in the aqueous phase and reducing interfacial tension, thereby facilitating better contact between the substrate and the intracellular enzymes without compromising cell viability. This results in a cleaner reaction profile with minimal side products, simplifying the final isolation steps.
How to Synthesize (S)-3,5-BTPE Efficiently
Implementing this biocatalytic route requires precise control over fermentation and conversion parameters to maximize yield and optical purity. The process begins with the cultivation of the ZJPH1807 strain under optimized conditions to generate high-activity wet biomass. This biomass is then suspended in a specifically formulated buffer system containing the necessary co-substrates and performance-enhancing additives. The following guide outlines the critical operational phases derived from the patent data, ensuring reproducibility and high efficiency for laboratory and pilot-scale operations.
- Cultivate Cyberlindnera saturns ZJPH1807 in a optimized fermentation medium containing glucose and yeast extract at 30°C to obtain wet biomass.
- Prepare a conversion system using Na2HPO4-NaH2PO4 buffer (pH 7.5) supplemented with glucose, L-carnitine: lysine NADES, and Tween-80 surfactant.
- Add 3,5-bis(trifluoromethyl)acetophenone substrate to the system and react at 30°C for 24 hours to achieve over 80% yield with >99.9% e.e.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic process offers compelling strategic advantages beyond mere technical feasibility. The elimination of precious metal catalysts fundamentally alters the cost structure of the synthesis, removing a volatile and expensive input from the bill of materials. Additionally, the ability to operate at high substrate concentrations significantly improves space-time yield, meaning that existing reactor infrastructure can produce more product in less time, effectively increasing capacity without capital expenditure. The mild reaction conditions (30°C, atmospheric pressure) also reduce energy consumption compared to high-temperature or high-pressure chemical alternatives.
- Cost Reduction in Manufacturing: The use of whole resting cells circumvents the expensive and complex purification steps associated with isolated enzymes, drastically lowering the cost of the biocatalyst itself. Furthermore, the in-situ regeneration of cofactors via glucose oxidation removes the need for purchasing external NADPH, representing a significant recurring cost saving. The high selectivity reduces the burden on downstream purification, lowering solvent consumption and waste disposal costs associated with removing impurities and metal residues.
- Enhanced Supply Chain Reliability: By relying on a fermentable microbial strain rather than scarce chemical catalysts or animal-derived enzymes, the supply chain becomes more robust and less susceptible to geopolitical or resource shortages. The strain can be preserved and propagated indefinitely, ensuring a consistent and renewable source of catalytic activity. This biological consistency translates to reliable batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification raw materials.
- Scalability and Environmental Compliance: The process operates in an aqueous buffer system with biodegradable additives, aligning perfectly with modern green chemistry principles and strict environmental regulations. The absence of heavy metals simplifies wastewater treatment and regulatory compliance, facilitating faster approval for commercial manufacturing sites. The demonstrated scalability of the fermentation and conversion steps ensures that the process can be seamlessly transferred from laboratory benchtop to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process robustness and product quality assurance for potential partners and licensees.
Q: What are the advantages of using whole-cell biocatalysis over chemical synthesis for this intermediate?
A: Whole-cell biocatalysis eliminates the need for expensive transition metal catalysts and harsh reaction conditions, significantly reducing environmental pollution and downstream purification costs while maintaining >99.9% enantiomeric excess.
Q: How does the addition of Natural Deep Eutectic Solvents (NADES) impact the reaction yield?
A: The patent demonstrates that adding specific NADES, such as L-carnitine to lysine (1:2 molar ratio), alongside surfactants like Tween-80, enhances substrate solubility and enzyme stability, boosting yields from roughly 60% to over 80% at high substrate concentrations.
Q: Is this process scalable for industrial production of Aprepitant intermediates?
A: Yes, the process utilizes robust resting cells that function effectively at high substrate loadings (up to 128.07 g/L) and moderate temperatures (30°C), making it highly suitable for commercial scale-up without the complexity of isolated enzyme systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3,5-BTPE Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN112048538B are successfully translated into reliable industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the >99.9% e.e. required for chiral alcohol intermediates used in oncology and antiemetic therapies.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient synthesis route for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this biocatalytic method can optimize your overall manufacturing budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments for your next project.
