Advanced Synthesis of 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol for Commercial Fragrance Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for high-value terpene derivatives, particularly those serving as critical building blocks for fragrances and active pharmaceutical ingredients. A significant breakthrough in this domain is documented in patent CN103554061A, which details a highly efficient preparation method for 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol. This compound, existing in both cis and trans isomeric forms, is a vital intermediate widely utilized in the flavors, food, and tobacco sectors due to its unique olfactory profile and chemical reactivity. The patented technology addresses long-standing challenges in terpene synthesis by introducing a streamlined, one-step reduction strategy that bypasses the complexities of earlier methodologies. By leveraging the chemoselective properties of sodium borohydride in a biphasic ethanol-water system, this process achieves exceptional control over the reaction pathway, ensuring the integrity of the sensitive epoxide ring while efficiently reducing the aldehyde functionality. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for complex acyclic monoterpene alcohols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol has been plagued by significant technical hurdles that hindered its widespread commercial adoption. Early attempts, such as those by Sharpless and colleagues, relied on the direct epoxidation of Geraniol using metachloroperbenzoic acid. While conceptually straightforward, this approach suffered from catastrophic chemo-selectivity issues, yielding a complex mixture of 6,7-epoxy products alongside unwanted 2,3-epoxy byproducts like 2,3-epoxy-3,7-dimethyl-octa-6-enol. Subsequent improvements using vanadium catalysts with tert-butyl peroxide managed to improve selectivity but often favored the wrong isomer entirely, failing to produce the desired 6,7-epoxy target. Furthermore, alternative routes involving esterification followed by epoxidation and saponification, as described by Mohasci, introduced excessive operational complexity. These multi-step sequences not only resulted in low overall yields ranging merely from 55% to 60% but also required difficult-to-source raw materials and generated substantial chemical waste, making them economically unfeasible for large-scale flavor and fragrance intermediate manufacturing.
The Novel Approach
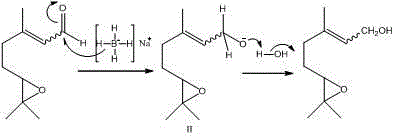
In stark contrast to these cumbersome legacy processes, the method disclosed in CN103554061A offers a paradigm shift by utilizing 6,7-epoxycitral as the direct precursor. This strategic choice of starting material effectively bypasses the selectivity nightmares associated with oxidizing geraniol, as the epoxide motif is already established prior to the reduction step. The novel approach employs sodium borohydride (NaBH4) as a mild yet effective reducing agent, dissolved in a cost-effective solvent system comprising ethanol and distilled water. This single-step transformation proceeds under remarkably mild conditions, typically maintaining a temperature range of 20-25°C after an initial cooling phase. The result is a dramatic improvement in process efficiency, where the reaction completes within 2.5 hours to deliver the target alcohol with high fidelity. By eliminating the need for transition metal catalysts and avoiding harsh oxidative conditions, this new route not only simplifies the operational workflow but also significantly enhances the safety profile and environmental compatibility of the production line, offering a compelling solution for cost reduction in fragrance manufacturing.
Mechanistic Insights into NaBH4-Mediated Chemoselective Reduction
At the heart of this technological advancement lies a precise understanding of hydride transfer mechanisms in the presence of sensitive functional groups. The reaction initiates with the dissolution of sodium borohydride in the ethanol-water mixture under an ice-water bath, creating a stabilized source of nucleophilic hydride ions. Upon the dropwise addition of 6,7-epoxycitral, the hydride species selectively attacks the electrophilic carbonyl carbon of the aldehyde group. Crucially, the reaction conditions are tuned to prevent the nucleophilic attack on the epoxide ring, which is susceptible to ring-opening under acidic or strongly nucleophilic conditions. The presence of water and ethanol acts as a proton source that moderates the reactivity of the borohydride, ensuring that the reduction stops at the alcohol stage without affecting the carbon-carbon double bonds or the oxirane ring. This chemoselectivity is paramount for maintaining the structural integrity required for the molecule's subsequent application in fine fragrance formulations.
Following the hydride attack, the intermediate alkoxide species (labeled as structure II in the reaction scheme) is formed transiently. This anionic intermediate subsequently abstracts a proton from the solvent medium (water or ethanol) to yield the final neutral alcohol product, 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol. The elegance of this mechanism is further reflected in the impurity profile; because the reaction avoids radical pathways or high-energy intermediates, the formation of side products is minimized. The patent data indicates that the crude product obtained after simple extraction already possesses high purity, which can be further elevated to 98% through standard silica gel chromatography using a petroleum ether and ethyl acetate eluent system. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the reaction trajectory is well-defined and robust against minor fluctuations in processing parameters.
How to Synthesize 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and temperature controls to maximize yield and safety. The process begins by preparing Solution A, where sodium borohydride is fully dissolved in a mixture of ethanol and distilled water, typically in a molar ratio of 1mol NaBH4 to roughly 2.22L of each solvent component. Into this cooled solution, the 6,7-epoxycitral substrate is introduced slowly over a 5-minute window to manage the exotherm and ensure uniform mixing. The reaction mixture is then allowed to stir at ambient temperature (20-25°C) for approximately 2.5 hours, a duration sufficient to drive the conversion to completion as monitored by TLC or GC. Upon completion, the reaction is carefully quenched with ice water, and the pH is adjusted to neutrality using dilute hydrochloric acid before extraction. For a detailed breakdown of the standardized operating procedures and safety precautions, please refer to the technical guide below.
- Dissolve sodium borohydride in a mixture of ethanol and distilled water under ice-water bath cooling to prepare the reducing solution.
- Add the ethanolic solution of 6,7-epoxycitral dropwise over 5 minutes, then maintain stirring at 20-25°C for 2.5 hours to complete the reduction.
- Quench the reaction with ice water, adjust pH to neutral, extract with ether, dry over magnesium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits that extend far beyond mere chemical yield. The primary economic driver is the drastic simplification of the supply chain for raw materials. By utilizing 6,7-epoxycitral, which is readily available and inexpensive compared to the specialized catalysts required for Sharpless epoxidation, companies can secure a more stable and cost-effective supply base. Furthermore, the elimination of transition metals such as vanadium or titanium removes the necessity for expensive metal scavenging resins and complex wastewater treatment protocols associated with heavy metal removal. This reduction in downstream processing requirements translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with commodity chemicals like sodium borohydride results in substantial cost savings on reagent procurement. Additionally, the use of ethanol and water as the primary solvent system avoids the high costs and disposal fees associated with chlorinated or aromatic solvents often used in older methodologies. The simplified workup procedure, which relies on standard extraction and evaporation techniques rather than complex crystallization or distillation trains, further reduces energy consumption and labor hours per batch, leading to a significantly improved margin structure for the final high-purity epoxy alcohol.
- Enhanced Supply Chain Reliability: Dependence on exotic catalysts often introduces supply chain vulnerabilities due to geopolitical factors or limited manufacturer capacity. This new method relies on universally available bulk chemicals, ensuring that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions—operating effectively at near-room temperature—also means that the process is less sensitive to utility fluctuations, such as cooling water temperature variations, thereby enhancing the overall reliability and predictability of manufacturing lead times for critical fragrance intermediates.
- Scalability and Environmental Compliance: The inherent safety of the sodium borohydride reduction in aqueous ethanol makes this process highly scalable from kilogram to multi-ton scales without requiring specialized high-pressure reactors. The absence of toxic heavy metals simplifies regulatory compliance and reduces the burden on environmental health and safety (EHS) departments. Waste streams are easier to treat, and the high atom economy of the reduction step minimizes the generation of hazardous byproducts, facilitating smoother permitting processes for commercial scale-up of complex terpenes in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what stakeholders can expect regarding performance and scalability. Understanding these nuances is essential for making informed decisions about integrating this technology into existing production portfolios.
Q: What is the primary advantage of this NaBH4 reduction method over traditional epoxidation routes?
A: Unlike traditional methods starting from Geraniol which suffer from poor chemo-selectivity and generate mixed epoxy isomers (2,3-epoxy vs 6,7-epoxy), this method starts with pre-formed 6,7-epoxycitral. This ensures exclusive formation of the desired 6,7-epoxy alcohol structure with significantly higher purity (98%) and yield (approx. 70%).
Q: How does this process impact production costs for fragrance manufacturers?
A: The process eliminates the need for expensive transition metal catalysts (such as vanadium or titanium complexes) and complex multi-step sequences. By utilizing inexpensive sodium borohydride and common solvents like ethanol and water, the raw material costs are drastically reduced, and the downstream purification burden is minimized.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates under mild conditions (20-25°C) without requiring cryogenic temperatures or high-pressure equipment. The simple workup procedure involving extraction and standard column chromatography makes it highly adaptable for commercial scale-up from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol Supplier
As the global demand for high-quality fragrance and pharmaceutical intermediates continues to rise, partnering with a technically proficient manufacturer is crucial for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the NaBH4 reduction process to deliver superior value to our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-(3,3-dimethyloxiranyl)-3-methyl-2-penten-1-ol meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us help you secure a reliable supply of this critical intermediate while driving innovation and efficiency in your product development pipeline.
