Advanced Manufacturing of High-Purity LiFSI Electrolyte Salts via Novel Lithium Alloy Lithiation Technology
The escalating demand for high-energy-density lithium-ion batteries in electric vehicles and grid storage applications has intensified the search for superior electrolyte salts, specifically Lithium Bis(fluorosulfonyl)imide (LiFSI). Patent CN116040592A introduces a groundbreaking preparation method that addresses critical bottlenecks in conventional manufacturing, offering a pathway to ultra-high purity and low-moisture content essential for next-generation battery performance. This technical disclosure outlines a robust two-step synthesis strategy that begins with the catalytic fluorination of chlorosulfonyl isocyanate to form bis(fluorosulfonyl)imide (HFSI), followed by a unique lithiation process utilizing lithium alloys rather than traditional aqueous-compatible bases. For global stakeholders seeking a reliable battery electrolyte additive supplier, this technology represents a significant leap forward in process efficiency and product quality, effectively mitigating the risks associated with hydrolysis and thermal instability often found in legacy production routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for synthesizing LiFSI predominantly rely on the neutralization of bis(fluorosulfonyl)imide with inorganic lithium bases such as lithium hydroxide, lithium carbonate, or lithium acetate. While chemically straightforward, these aqueous or semi-aqueous pathways inherently generate water as a stoichiometric byproduct during the salt formation reaction. The presence of water is catastrophic for LiFSI stability, as it promotes hydrolysis leading to the formation of corrosive acidic impurities and degrades the electrochemical performance of the final battery cell. Consequently, manufacturers employing these legacy routes face substantial downstream challenges, necessitating energy-intensive drying processes, complex solvent exchanges, and rigorous purification steps to reduce water content to acceptable parts-per-million levels. Furthermore, the thermal sensitivity of intermediates in these conventional routes often results in lower overall yields and inconsistent impurity profiles, creating significant supply chain vulnerabilities for high-volume battery material procurement.
The Novel Approach
In stark contrast, the methodology disclosed in CN116040592A circumvents the fundamental issue of water generation by employing a lithium alloy, such as zinc-lithium or tin-lithium, as the lithiating agent. This innovative approach allows the acid-base reaction to proceed in anhydrous organic solvents without producing water, thereby preserving the structural integrity of the sensitive imide anion. The process operates under mild conditions, typically between 15°C and 120°C, and remarkably does not require the continuous blanket of inert gas often mandated for handling reactive lithium species, as the alloy reacts at a controlled and moderate rate. This simplification of the reaction environment not only enhances operational safety but also drastically reduces the complexity of the reactor setup and post-reaction workup. By eliminating the need for extensive dehydration and avoiding the formation of hydrolysis byproducts, this novel route delivers a product with exceptional purity exceeding 99.9% and water content below 10 ppm, directly addressing the critical quality parameters demanded by top-tier battery manufacturers.
Mechanistic Insights into Lewis Acid Catalyzed Fluorination and Alloy Lithiation
The core of this synthesis lies in the efficient construction of the bis(fluorosulfonyl)imide backbone through a Lewis acid-catalyzed fluorination mechanism. In the first stage, chlorosulfonyl isocyanate reacts with fluorosulfonic acid in the presence of anhydrous hydrogen fluoride and a catalytic amount of a strong Lewis acid, such as antimony pentachloride (SbCl5), tin tetrachloride (SnCl4), or molybdenum pentachloride (MoCl5). The Lewis acid activates the sulfur-chlorine bonds, facilitating the nucleophilic attack by fluoride ions derived from the hydrogen fluoride and fluorosulfonic acid mixture. This transformation effectively replaces the chlorosulfonyl groups with fluorosulfonyl groups while maintaining the central imide linkage, releasing hydrogen chloride and carbon dioxide as gaseous byproducts which are easily separated from the reaction mixture. The use of specific molar ratios, such as a chlorosulfonyl isocyanate to fluorosulfonic acid ratio of 1:1.1 to 1.2, ensures complete conversion while minimizing the formation of polymeric side products, resulting in a crude intermediate that is already of high quality prior to distillation.
Following the isolation of high-purity bis(fluorosulfonyl)imide, the second mechanistic phase involves the deprotonation of the imide nitrogen by the lithium alloy. Unlike monometallic lithium which can react violently, the alloying with zinc or tin modulates the reactivity of the lithium atoms, allowing for a smooth substitution reaction in solvents like petroleum ether or dichloromethane. The lithium atom displaces the acidic proton on the nitrogen, forming the stable lithium imide salt while the remaining alloy components precipitate or remain soluble depending on the specific solvent system chosen. This solid-liquid or liquid-liquid heterogeneous reaction is kinetically favorable at temperatures as low as 25°C to 35°C, preventing thermal degradation of the product. The absence of water generation in this step is the key differentiator, ensuring that the final crystal lattice of the LiFSI is free from hydrate formations, which is critical for achieving the low impedance and high cycle life required in modern lithium-ion cells.
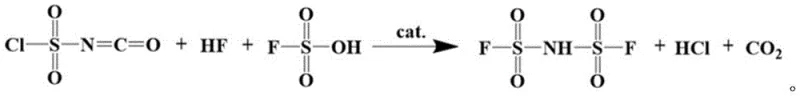
How to Synthesize Lithium Bis(fluorosulfonyl)imide Efficiently
To implement this advanced manufacturing protocol effectively, precise control over reaction stoichiometry and temperature profiles is essential to maximize yield and minimize impurity carryover. The process begins with the careful mixing of chlorosulfonyl isocyanate and fluorosulfonic acid under stirring, followed by the continuous introduction of anhydrous hydrogen fluoride gas while maintaining the reaction temperature between 65°C and 85°C for a duration of 6 to 12 hours. Once the fluorination is complete, the reaction mixture undergoes cooling crystallization and reduced pressure distillation to isolate the bis(fluorosulfonyl)imide intermediate with purity levels approaching 99.9%. The subsequent lithiation step requires dissolving this intermediate in an anhydrous organic solvent and adding the lithium alloy at a molar ratio of 1:1.0 to 1.25 relative to the lithium content in the alloy. Detailed standardized operating procedures for scaling this pathway from laboratory to commercial production are outlined below.
- Mix chlorosulfonyl isocyanate and fluorosulfonic acid, then introduce anhydrous hydrogen fluoride gas with a Lewis acid catalyst at 65-85°C to synthesize bis(fluorosulfonyl)imide.
- Purify the resulting bis(fluorosulfonyl)imide intermediate through cooling crystallization and reduced pressure distillation to achieve >99.8% purity.
- React the purified intermediate with a zinc-lithium or tin-lithium alloy in an organic solvent at 25-35°C, followed by filtration and vacuum drying to obtain final LiFSI crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this alloy-based synthesis route offers profound economic and logistical benefits that extend beyond simple yield improvements. By fundamentally altering the chemistry to avoid water generation, the process eliminates the need for expensive and time-consuming dehydration units, molecular sieve beds, and extended vacuum drying cycles that characterize traditional manufacturing lines. This reduction in unit operations translates directly into lower capital expenditure for new facilities and reduced operational expenditures for existing plants, as energy consumption for heating and vacuum generation is significantly curtailed. Furthermore, the ability to operate the lithiation step without strict inert gas protection simplifies the reactor design and reduces the consumption of costly noble gases like argon or nitrogen, contributing to a leaner and more cost-effective production model that enhances overall margin potential in a competitive market.
- Cost Reduction in Manufacturing: The elimination of water as a byproduct removes the necessity for complex purification trains designed to separate azeotropic mixtures or remove trace moisture, which are traditionally the most expensive stages in LiFSI production. By utilizing lithium alloys that react cleanly to form the salt, the process avoids the yield losses associated with hydrolysis and the subsequent reprocessing of off-spec material. Additionally, the catalyst loading is extremely low, ranging from 0.1 per mille to 2 percent, which minimizes the cost of precious metal or specialized chemical inputs while maintaining high turnover numbers. These factors combine to create a substantially lower cost of goods sold, allowing suppliers to offer more competitive pricing structures to downstream battery cell manufacturers without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including chlorosulfonyl isocyanate, fluorosulfonic acid, and zinc or tin lithium alloys, are commercially available from established chemical suppliers with robust global distribution networks. Unlike some exotic precursors that may suffer from geopolitical supply constraints, these feedstocks benefit from mature production scales in the broader fine chemical industry. The simplified process flow also reduces the risk of unplanned downtime caused by equipment fouling or purification bottlenecks, ensuring a more consistent and predictable output volume. This reliability is crucial for automotive OEMs and battery giants who require guaranteed continuity of supply to meet their aggressive production schedules for electric vehicles and energy storage systems.
- Scalability and Environmental Compliance: The gaseous byproducts generated in the first step, specifically hydrogen chloride and carbon dioxide, are easily separated from the liquid reaction mass and can be captured or neutralized using standard scrubbing technologies, minimizing the environmental footprint of the facility. The absence of aqueous waste streams significantly reduces the burden on wastewater treatment plants, aligning the manufacturing process with increasingly stringent environmental regulations regarding industrial effluent. Moreover, the moderate reaction temperatures and pressures facilitate safe scale-up from pilot plants to multi-ton commercial reactors, as the thermal hazards associated with highly exothermic lithiation reactions are mitigated by the use of the alloy system. This inherent safety and scalability make the technology ideally suited for rapid capacity expansion to meet the surging global demand for high-performance electrolyte salts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific LiFSI preparation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of purity, yield, and operational simplicity. Understanding these nuances is vital for technical teams evaluating potential licensing opportunities or procurement partners who utilize this specific synthetic route to ensure alignment with their quality assurance protocols.
Q: Why is lithium alloy preferred over lithium hydroxide for LiFSI production?
A: Using lithium alloys eliminates water generation during the salt formation step, which is a critical advantage over traditional lithium hydroxide methods that require complex dehydration and purification to remove trace moisture.
Q: What is the expected purity level of LiFSI produced via this patent method?
A: The described process consistently achieves product purity exceeding 99.9%, with water content controlled below 10 ppm and metal ion impurities also maintained below 10 ppm, meeting stringent battery-grade specifications.
Q: Does this synthesis route require inert gas protection during lithiation?
A: No, the reaction between bis(fluorosulfonyl)imide and the lithium alloy proceeds at a moderate rate without the need for inert gas protection, significantly simplifying the operational requirements and equipment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lithium Bis(fluorosulfonyl)imide Supplier
As the global transition to electrification accelerates, the need for high-performance electrolyte components like LiFSI has never been more critical, requiring partners who possess both the technical expertise and the manufacturing capacity to deliver consistent quality. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous demands of the battery sector. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of LiFSI delivered meets the exacting standards required for high-voltage and high-temperature battery applications. We understand that in the fast-paced world of energy storage, reliability and quality are non-negotiable, and our commitment to continuous process improvement ensures we remain a trusted partner for long-term supply agreements.
We invite procurement leaders and technical directors to engage with our team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments that demonstrate the tangible benefits of our production methods. Whether you are developing next-generation cylindrical cells or large-format prismatic modules, our dedicated support ensures that your supply chain is optimized for performance, cost, and continuity, positioning your organization for success in the competitive landscape of electric mobility and renewable energy storage.
