Industrial Scale-Up of Tauroursodeoxycholic Acid Dihydrate via Optimized Mixed Anhydride Technology
The pharmaceutical landscape for bile acid derivatives continues to evolve, driven by the expanding clinical applications of Tauroursodeoxycholic acid (TUDCA) in neuroprotection and metabolic regulation. A significant technological breakthrough in this domain is detailed in patent CN114539342B, which discloses an industrialized preparation method for Tauroursodeoxycholic acid dihydrate. This innovation addresses long-standing challenges in the synthesis of this critical pharmaceutical intermediate, specifically focusing on the optimization of the mixed anhydride pathway. By shifting the reaction medium from traditional aprotic solvents to specific alcohol systems, the patent outlines a route that drastically minimizes impurity formation while enhancing overall process efficiency. For global stakeholders seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this solvent-engineered approach is vital for securing high-quality supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
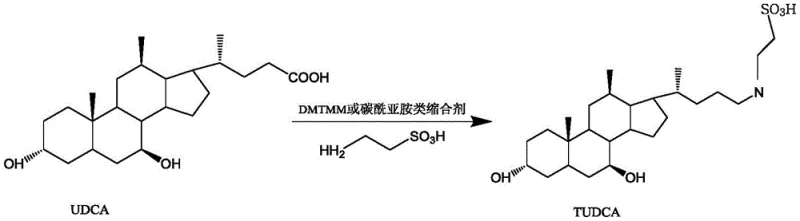
Historically, the synthesis of TUDCA has relied heavily on three primary methodologies, each presenting distinct operational and economic hurdles. The condensing agent method, often utilizing carbodiimides like DCC or peptide agents like DMTMM, suffers from significant drawbacks; DCC is known for its skin irritation and potential carcinogenicity, making it undesirable for late-stage pharmaceutical processing, while DMTMM, though milder, incurs prohibitive costs that hinder large-scale adoption. Alternatively, the active ester method involves reacting UDCA with silane or borate compounds. While effective, this route generates wastewater contaminated with boron or silicon, creating substantial environmental compliance burdens and increasing the cost of waste treatment. Furthermore, the steric hindrance required to prevent side reactions in these systems often necessitates expensive, high-molecular-weight reagents.
The Novel Approach
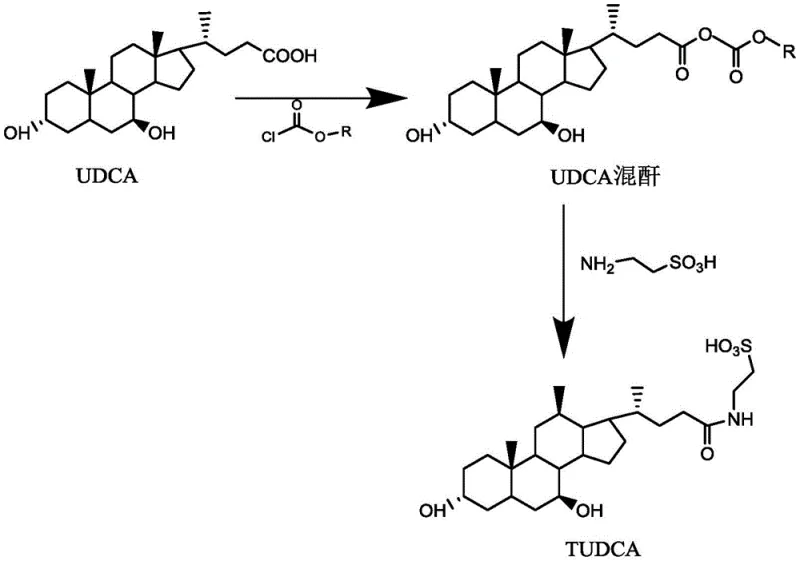
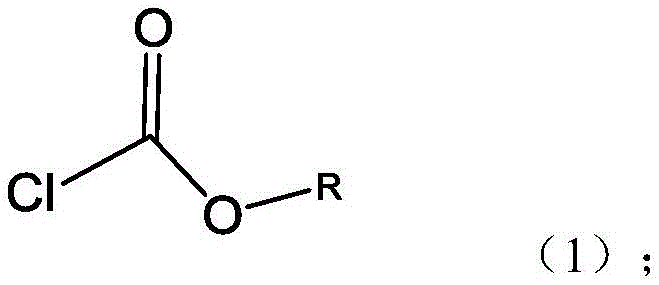
The methodology presented in CN114539342B revolutionizes the mixed anhydride process by introducing a synergistic solvent-reagent system. Unlike prior art that predominantly utilized acetone, ethyl acetate, or dioxane—solvents prone to facilitating side reactions at the 3-hydroxy position of UDCA—this invention employs propanol or isopropanol as the reaction medium. Crucially, the alkyl group of the chloroformate reagent matches the alcohol solvent (e.g., propyl chloroformate in propanol). This strategic alignment not only suppresses the formation of ether-linked impurities but also simplifies the downstream solvent recovery process. The result is a streamlined workflow that achieves high molar yields exceeding 85% and HPLC purities greater than 99.5%, positioning it as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Alcohol-Mediated Mixed Anhydride Formation
The core chemical innovation lies in the counter-intuitive use of alcohols as solvents for chloroformate activation. Conventionally, alcohols are avoided in mixed anhydride preparations due to the risk of nucleophilic substitution, where the solvent alcohol reacts with the chloroformate to form carbonates. However, the patent elucidates that the stability of alkyl chloroformates in alcoholic media is strictly dependent on carbon chain length and temperature. By selecting alcohols with three or more carbon atoms (propanol/isopropanol) and maintaining rigorous low-temperature control between -5°C and -25°C, the reactivity of the chloroformate towards the solvent is significantly dampened. This allows the chloroformate to selectively activate the carboxylic acid of UDCA without compromising the integrity of the 3-hydroxy group, which is highly susceptible to acylation in other solvent systems.
Furthermore, this mechanistic stability translates directly into superior impurity control. In traditional acetone-based systems, the high reactivity of the 3-hydroxy group often leads to the formation of di-acylated byproducts or ether impurities, complicating purification. The alcohol-mediated system effectively protects this position through kinetic control, ensuring that the subsequent coupling with taurine proceeds with high specificity. The resulting mixed anhydride remains stable for up to 24 hours under these low-temperature conditions, providing a robust window for the addition of the alkaline taurine solution. This precise control over reaction kinetics is essential for achieving the stringent purity specifications required for high-purity pharmaceutical intermediates intended for therapeutic use.
How to Synthesize Tauroursodeoxycholic Acid Efficiently
The synthesis protocol described in the patent offers a reproducible framework for manufacturing TUDCA dihydrate at an industrial scale. The process begins with the dissolution of ursodeoxycholic acid in the selected alcohol solvent, followed by the controlled addition of the matching alkyl chloroformate at sub-zero temperatures. Once the mixed anhydride is formed, it reacts directly with an aqueous solution of sodium taurate, eliminating the need for intermediate isolation and reducing material handling losses. The final crude product is then subjected to a specialized crystallization process involving water, alcohol, and acetone to remove residual impurities. For a detailed breakdown of the specific operational parameters, stoichiometry, and purification steps, please refer to the standardized guide below.
- React Ursodeoxycholic acid (UDCA) with propyl or isopropyl chloroformate in a corresponding alcohol solvent (propanol or isopropanol) at low temperatures (-5°C to -25°C) to form the mixed anhydride.
- Add an alkaline aqueous solution of taurine directly to the mixed anhydride reaction mixture to perform the coupling reaction without isolating the intermediate.
- Purify the crude TUDCA product through crystallization using a solvent system comprising water, alcohol, and acetone to achieve high purity (>99.5%).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this alcohol-based mixed anhydride method offers transformative benefits for procurement and supply chain management. The primary advantage stems from the drastic simplification of the solvent system. By utilizing the same alcohol class for both the solvent and the chloroformate reagent, the process eliminates the complexity of separating disparate solvent streams. This alignment facilitates highly efficient solvent recovery and recycling, which directly correlates to substantial cost savings in raw material procurement. Additionally, the avoidance of exotic reagents like borates or silanes removes the need for specialized waste treatment infrastructure, further lowering the total cost of ownership for manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive condensing agents like DMTMM and the removal of heavy metal or boron-based reagents significantly lowers the direct material costs associated with production. Moreover, the ability to recover and reuse the alcohol solvent reduces the volume of fresh solvent required per batch. This closed-loop solvent management system not only cuts purchasing costs but also minimizes the expenses related to hazardous waste disposal, driving down the overall manufacturing overhead without compromising product quality.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process, specifically propanol, isopropanol, and alkyl chloroformates, are commodity chemicals with robust global supply chains. Unlike specialized peptide coupling agents that may face supply bottlenecks or long lead times, these reagents are readily available from multiple vendors. This abundance ensures consistent production scheduling and reduces the risk of supply disruptions, making it easier for partners to maintain continuous inventory levels of this critical pharmaceutical intermediate.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic byproducts make this process inherently scalable from pilot plants to multi-ton commercial reactors. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the regulatory burden on manufacturing sites. The simplified purification process, which relies on standard crystallization techniques rather than complex chromatography, further supports rapid scale-up, ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal technical friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on quality control and operational feasibility. Understanding these details is crucial for technical teams evaluating the viability of this method for their specific production needs.
Q: Why are alcohol solvents preferred over acetone or dioxane in this TUDCA synthesis?
A: Traditional solvents like acetone often lead to side reactions at the 3-hydroxy position of UDCA, generating impurities. The patented method utilizes propanol or isopropanol, which, under controlled low temperatures, stabilizes the alkyl chloroformate and prevents unwanted substitution, significantly improving product purity and yield.
Q: How does this method address environmental concerns compared to active ester methods?
A: Unlike active ester methods that utilize silane or borate reagents resulting in difficult-to-treat wastewater containing boron or silicon, this mixed anhydride approach uses common organic solvents and reagents. Furthermore, the matching of the solvent alcohol with the chloroformate alkyl group facilitates efficient solvent recovery and recycling, reducing environmental impact.
Q: What represents the critical control point for impurity management in this process?
A: The critical control point is the reaction temperature during the formation of the mixed anhydride, specifically maintaining it between -5°C and -25°C. This low-temperature regime ensures the stability of the alkyl chloroformate in the alcohol system, preventing it from reacting with the solvent while allowing it to activate the carboxylic acid of UDCA selectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tauroursodeoxycholic Acid Supplier
The technological advancements detailed in CN114539342B underscore the potential for producing TUDCA with exceptional purity and efficiency. At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to life. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of TUDCA meets the highest international standards. We are committed to translating complex laboratory discoveries into robust, GMP-compliant manufacturing processes that deliver value to our global partners.
We invite you to collaborate with us to optimize your supply chain for bile acid derivatives. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can support your long-term strategic goals.
