Advanced Synthesis Strategy for High-Purity 6-Amino-2,2-dimethyl-2H-pyridino[3,2-b][1,4]oxazine-3(4H)-one Intermediates
Introduction to Next-Generation Syk Inhibitor Intermediate Synthesis
The pharmaceutical landscape for oncology treatments, particularly for lymphomas and leukemias, relies heavily on the efficient production of Spleen Tyrosine Kinase (Syk) inhibitors. A critical bottleneck in this supply chain has been the synthesis of the core heterocyclic scaffold, 6-amino-2,2-dimethyl-2H-pyridino[3,2-b][1,4]oxazine-3(4H)-one. Patent CN102491985A introduces a transformative synthetic methodology that addresses long-standing issues of low yield and poor selectivity. This technical insight report analyzes the patented three-step sequence—cyclization, selective nitration, and reduction—which collectively deliver a robust pathway for reliable pharmaceutical intermediate supplier operations. By shifting from non-selective electrophilic substitutions to a controlled low-temperature nitration protocol, this technology enables the production of high-purity API precursors essential for downstream drug development.
The strategic value of this patent lies not merely in the chemical transformation but in its operational feasibility for industrial scale-up. Traditional routes often necessitate tedious purification steps like column chromatography, which are economically unviable for multi-kilogram production. In contrast, the disclosed method leverages crystallization and filtration, unit operations that are inherently scalable and cost-effective. For procurement managers and supply chain heads, understanding this shift from laboratory-scale complexity to manufacturing simplicity is crucial for securing long-term supply continuity. The following sections dissect the mechanistic advantages and commercial implications of adopting this synthesis route for your API manufacturing portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in JP2008/013527A, present significant hurdles for commercial adoption due to inherent chemical inefficiencies. One reported pathway involves a multi-step sequence yielding a mere 29% overall efficiency, primarily due to difficult intermediate purification and low conversion rates in key coupling steps. More critically, alternative nitration strategies described in the same literature utilize acetic anhydride and nitric acid, resulting in a non-selective mixture of 6-nitro and 7-nitro isomers. As illustrated in the reaction scheme below, this lack of regioselectivity forces manufacturers to rely on column chromatography for separation, a technique that drastically reduces final yield to approximately 11% and generates substantial solvent waste.
Furthermore, academic literature such as Tetrahedron Letters (2010) proposed using expensive ionic liquids like [omim][BF4] to facilitate the cyclization step. While achieving decent yields in small batches, the high cost of these specialized reagents renders the process economically prohibitive for large-scale industrial application. The reliance on exotic catalysts increases the raw material cost basis and introduces supply chain vulnerabilities, as these reagents are not typically stocked by bulk chemical suppliers. Consequently, existing methods fail to meet the dual requirements of high purity and cost reduction in pharmaceutical intermediate manufacturing, necessitating a more pragmatic approach.
The Novel Approach
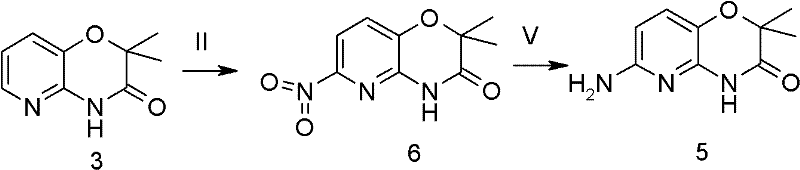
The patented method outlined in CN102491985A overcomes these deficiencies through a streamlined three-step process that prioritizes regioselectivity and operational simplicity. The core innovation is the use of a mixed acid system (concentrated sulfuric acid and fuming nitric acid) at strictly controlled low temperatures (0-5°C). This specific condition set directs the electrophilic attack exclusively to the 6-position of the pyridine ring, avoiding the formation of the 7-nitro impurity entirely. The result is a dramatic improvement in yield, with the nitration step alone achieving up to 97.7% efficiency, thereby eliminating the need for chromatographic purification.
In the final reduction step, the patent offers flexibility by describing both catalytic hydrogenation and a chemical reduction system using hydrazine hydrate and ferric chloride. This chemical reduction pathway is particularly advantageous for facilities lacking high-pressure hydrogenation infrastructure, as it proceeds under atmospheric pressure with excellent conversion rates (up to 80%). By combining inexpensive starting materials like 2-amino-3-hydroxypyridine with robust reaction conditions, this novel approach establishes a new benchmark for the commercial scale-up of complex pharmaceutical intermediates, ensuring both economic viability and consistent product quality.
Mechanistic Insights into Selective Nitration and Catalytic Reduction
The success of this synthesis hinges on the precise control of electronic effects during the nitration phase. The substrate, 2,2-dimethyl-2H-pyrido[3,2-b][1,4]oxazin-3(4H)-one, possesses a fused heterocyclic system where the electron density is unevenly distributed. Under standard nitrating conditions, the pyridine nitrogen exerts a deactivating effect, while the oxygen in the oxazine ring can activate adjacent positions. By maintaining the reaction temperature between 0-5°C using an ice-salt bath, the kinetic energy of the nitronium ions is moderated. This prevents over-nitration and ensures that the electrophile attacks the most activated position (C-6) rather than the sterically or electronically less favorable C-7 position. This kinetic control is the fundamental driver behind the observed 97.7% yield and high regioselectivity.
Regarding the reduction mechanism, the hydrazine/ferric chloride system operates via a catalytic transfer hydrogenation pathway. Ferric chloride acts as a Lewis acid catalyst that facilitates the decomposition of hydrazine hydrate, generating active hydrogen species in situ. These species then reduce the nitro group to the primary amine without affecting the sensitive lactam carbonyl or the ether linkage within the oxazine ring. The addition of activated carbon serves a dual purpose: it acts as a support for the iron catalyst to increase surface area and aids in the adsorption of colored impurities and tars formed during the exothermic reduction. This results in a crude product of such high purity (reported as 100% in Example 1) that simple filtration and drying suffice, bypassing the need for recrystallization in many cases.
How to Synthesize 6-Amino-2,2-dimethyl-2H-pyridino[3,2-b][1,4]oxazine-3(4H)-one Efficiently
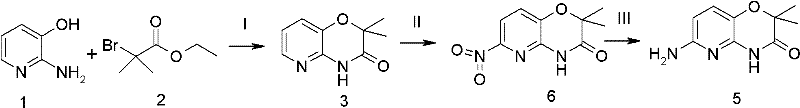
Implementing this synthesis requires strict adherence to the specified stoichiometry and thermal profiles to maximize yield and safety. The process begins with the cyclization of 2-amino-3-hydroxypyridine and ethyl 2-bromo-2-methylpropionate using potassium carbonate in a solvent like acetone or methanol. Following isolation of the intermediate, the critical nitration step must be performed with careful temperature monitoring to prevent runaway exotherms. Finally, the reduction is carried out in THF or DMSO. For detailed operational parameters, stoichiometry, and workup procedures, refer to the standardized guide below.
- Cyclization: React 2-amino-3-hydroxypyridine with ethyl 2-bromo-2-methylpropionate in the presence of potassium carbonate under reflux to form the oxazine ring.
- Selective Nitration: Treat the intermediate with a mixed acid of concentrated sulfuric acid and fuming nitric acid at 0-5°C to selectively introduce the nitro group at the 6-position.
- Reduction: Reduce the nitro compound to the final amine using hydrazine hydrate with ferric chloride and activated carbon, or alternatively via catalytic hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers profound benefits for procurement strategies and supply chain resilience. By replacing low-yielding, chromatography-dependent methods with a high-yield crystallization-based process, manufacturers can significantly reduce the cost of goods sold (COGS). The elimination of expensive ionic liquids and the minimization of solvent usage for purification directly translate to substantial cost savings in raw material procurement. Furthermore, the use of commodity chemicals like potassium carbonate and mineral acids ensures that the supply chain is not dependent on niche vendors, thereby enhancing supply security and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from a 11% yield process to one exceeding 60-80% overall yield represents a massive efficiency gain. By removing the column chromatography step, the process eliminates the high costs associated with silica gel, large volumes of elution solvents, and the labor-intensive nature of batch purification. Additionally, the avoidance of expensive catalysts like palladium on carbon (in the preferred hydrazine method) or ionic liquids further drives down the variable cost per kilogram, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-amino-3-hydroxypyridine and ethyl 2-bromo-2-methylpropionate, are widely available bulk chemicals with stable pricing and multiple global sources. This diversification of supply mitigates the risk of production stoppages due to single-source vendor failures. Moreover, the robustness of the reaction conditions—tolerant of minor variations in mixing and temperature within the specified ranges—ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and uninterrupted delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method is superior to high-pressure hydrogenation routes. The atmospheric pressure reduction using hydrazine reduces the capital expenditure required for specialized pressure vessels and safety systems. The process generates less hazardous waste compared to methods requiring heavy metal catalysts or chlorinated solvents for chromatography. This alignment with green chemistry principles simplifies waste treatment protocols and facilitates easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy for R&D and technical procurement teams evaluating this route for potential licensing or contract manufacturing.
Q: How does this synthesis method improve upon previous literature methods like JP2008/013527A?
A: Unlike previous methods which suffered from non-selective nitration producing difficult-to-separate 6-nitro and 7-nitro mixtures with low yields (11%), this patented process achieves high regioselectivity at the 6-position with yields up to 97.7% in the nitration step, eliminating the need for complex column chromatography.
Q: Is the reduction step suitable for large-scale manufacturing without high-pressure equipment?
A: Yes, the preferred embodiment utilizes a hydrazine hydrate and ferric chloride catalytic system which operates under atmospheric pressure and mild heating, avoiding the safety risks and capital expenditure associated with high-pressure hydrogenation reactors required by traditional Pd/C methods.
Q: What are the cost advantages regarding raw materials and solvents?
A: The process avoids expensive ionic liquids like [omim][BF4] used in other literature methods. Instead, it utilizes commodity chemicals such as potassium carbonate, acetone, and standard mineral acids, significantly reducing the raw material cost basis for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Amino-2,2-dimethyl-2H-pyridino[3,2-b][1,4]oxazine-3(4H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields reported in the laboratory are faithfully reproduced in our manufacturing plants. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of regioisomers and residual solvents, guaranteeing that every batch meets the exacting standards required for oncology drug synthesis.
We invite you to collaborate with us to optimize your supply chain for Syk inhibitor intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our mastery of this advanced synthesis method can drive value and reliability for your pharmaceutical projects.
