Advanced FATSFI Manufacturing: Scaling High-Purity Solar and Battery Additives
Advanced FATSFI Manufacturing: Scaling High-Purity Solar and Battery Additives
The rapid evolution of perovskite solar cells and next-generation lithium-ion batteries demands electrolyte additives and charge transport materials that offer superior stability and conductivity without the hygroscopic drawbacks of traditional alkali metal salts. A groundbreaking development detailed in patent CN116332811A introduces a novel class of compounds known as Bistrifluoromethanesulfonylimide Formamidine, or FATSFI, which addresses these critical performance bottlenecks. This technical insight report analyzes the dual synthetic pathways disclosed in the patent, highlighting how the strategic selection of precursors—either acetic acid formamidine or formamidine halides—enables the production of ultra-high purity materials essential for sensitive electronic applications. By replacing unstable cations like lithium or silver with a robust formamidine structure, manufacturers can achieve significant improvements in electrochemical stability and interfacial charge extraction, positioning FATSFI as a cornerstone material for the future of renewable energy storage and conversion technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on alkali metal salts of bistrifluoromethanesulfonimide, such as LiTSFI, NaTSFI, and KTSFI, to serve as conductive additives in electrolytes and hole transport layers. However, these conventional inorganic salts suffer from inherent physicochemical deficiencies that limit device longevity and efficiency, particularly their pronounced tendency to absorb moisture from the environment which leads to rapid degradation of the active layer. Furthermore, the small ionic radius and high charge density of alkali metals often result in uncontrolled migration within the device architecture, causing hysteresis and long-term instability under operational stress. The synthesis of these traditional salts frequently involves harsh conditions or difficult purification steps to remove residual metal ions that can act as recombination centers, thereby reducing the overall photoelectric conversion efficiency. Consequently, there is an urgent industrial need for organic-inorganic hybrid alternatives that retain the high conductivity of the TSFI anion while mitigating the instability associated with simple inorganic cations.
The Novel Approach
The innovative methodology presented in the patent data circumvents these issues by introducing an organic formamidine cation that forms a stable ion pair with the TSFI anion, effectively creating a room-temperature ionic liquid precursor with solid-state crystallinity. This novel approach utilizes mild, room-temperature reactions that proceed to completion within just 20 minutes, drastically reducing energy consumption compared to high-temperature fusion methods used for inorganic salts. The strategy employs two distinct routes: one leveraging the volatility of acetic acid to drive the reaction equilibrium forward through distillation, and another utilizing the insolubility of silver halides to physically separate byproducts via filtration. This dual-pathway flexibility allows manufacturers to select the most cost-effective or logistically feasible route based on local raw material availability, ensuring a robust supply chain for high-purity electronic chemicals. The resulting FATSFI compound exhibits exceptional thermal stability and moisture resistance, directly addressing the failure modes observed in legacy alkali metal-based additives.
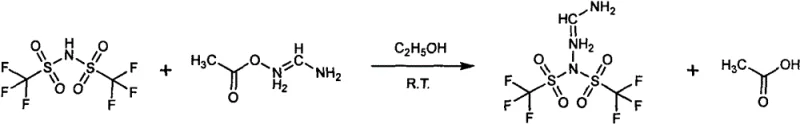
Mechanistic Insights into Acid-Base Neutralization and Metathesis
The first synthetic pathway operates on a classic acid-base neutralization mechanism where the strong sulfonimide acid reacts with the basic formamidine acetate salt in a polar protic solvent such as ethanol. In this process, the proton transfer from the sulfonimide to the acetate anion generates acetic acid as a byproduct, which possesses a significantly lower boiling point than the target ionic product. This thermodynamic disparity is exploited by applying vacuum distillation, which continuously removes the acetic acid and solvent from the reaction mixture, effectively driving the equilibrium towards the formation of the desired FATSFI product according to Le Chatelier's principle. The absence of heavy metal catalysts or complex ligands simplifies the impurity profile, ensuring that the final crystal lattice is free from transition metal contaminants that could detrimentally affect the electronic properties of the solar cell or battery interface. This clean reaction profile is critical for achieving the reported purity levels exceeding 99.9 percent, which is a stringent requirement for semiconductor-grade additives.
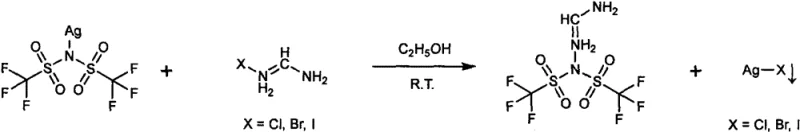
The alternative pathway utilizes a salt metathesis reaction involving silver bistrifluoromethanesulfonimide and various formamidine halides, capitalizing on the extremely low solubility product of silver halides in organic media. When the silver salt solution is mixed with the formamidine halide, an immediate precipitation of silver chloride, bromide, or iodide occurs, effectively sequestering the halide anion and leaving the formamidine cation free to pair with the TSFI anion in the solution phase. This precipitation-driven mechanism provides a powerful purification step inline with the reaction itself, as the solid silver halide can be mechanically removed via simple filtration before the solvent is evaporated. The choice of halide allows for fine-tuning of the reaction kinetics and solubility characteristics, offering process engineers flexibility in optimizing the workup procedure for different batch sizes. Both mechanisms underscore a design philosophy focused on atom economy and ease of separation, which are paramount for scaling fine chemical manufacturing processes.
How to Synthesize Bistrifluoromethanesulfonylimide Formamidine Efficiently
Implementing this synthesis requires precise control over stoichiometry and solvent selection to maximize yield and minimize residual impurities. The patent specifies that maintaining a strict 1:1 molar ratio between the sulfonimide source and the formamidine source is essential to prevent the presence of unreacted starting materials which could complicate the downstream purification. Solvents such as anhydrous ethanol are preferred due to their ability to dissolve both ionic precursors while remaining volatile enough for easy removal, though other polar solvents like ketones or ethers may also be employed depending on specific solubility requirements. Following the reaction, a recrystallization step using dichloromethane as an anti-solvent is recommended to further refine the crystal morphology and remove trace organic impurities, yielding white needle-shaped crystals suitable for high-end electronic applications.
- Dissolve bistrifluoromethanesulfonimide and formamidine acetate in anhydrous ethanol at room temperature, maintaining a 1: 1 molar ratio for optimal conversion.
- Remove the solvent and acetic acid byproduct via vacuum distillation, followed by recrystallization using dichloromethane as an anti-solvent to isolate pure crystals.
- Alternatively, react silver bistrifluoromethanesulfonimide with formamidine halides, filtering off the silver halide precipitate before evaporating the solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FATSFI synthesis route offers compelling strategic advantages rooted in process simplicity and raw material accessibility. The reliance on commodity chemicals such as acetic acid formamidine and ethanol eliminates the dependency on scarce or geopolitically sensitive reagents, thereby stabilizing the cost structure and ensuring consistent supply continuity even during market fluctuations. The elimination of expensive transition metal catalysts or complex chromatographic purification steps translates directly into reduced operational expenditures, as the process relies on standard unit operations like distillation and filtration that are easily implemented in existing multipurpose chemical plants. Furthermore, the room-temperature operating conditions significantly lower the energy footprint of the manufacturing process, aligning with global sustainability goals and reducing utility costs associated with heating or cryogenic cooling systems.
- Cost Reduction in Manufacturing: The process design inherently minimizes waste generation and energy consumption by utilizing volatile byproducts that are easily separated without intensive workup procedures. By avoiding the use of precious metal catalysts that require recovery and recycling infrastructure, the overall cost of goods sold is substantially decreased, allowing for more competitive pricing in the high-volume electronic chemical market. The high reaction efficiency and rapid completion time also increase reactor throughput, enabling manufacturers to produce larger quantities of material within the same timeframe compared to slower, multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key precursors are widely available bulk chemicals with established global supply chains, reducing the risk of bottlenecks associated with specialty reagents. The robustness of the synthesis against minor variations in reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with Tier-1 solar and battery manufacturers who demand rigorous specification compliance. This reliability fosters stronger partnerships and reduces the administrative burden of quality disputes or supply interruptions.
- Scalability and Environmental Compliance: The straightforward nature of the reaction workup, involving simple distillation or filtration, facilitates seamless scale-up from laboratory grams to metric ton production without requiring specialized equipment or hazardous high-pressure conditions. The use of environmentally benign solvents like ethanol and the generation of non-toxic byproducts like acetic acid simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations in major chemical manufacturing hubs. This green chemistry profile enhances the brand reputation of suppliers and meets the sustainability criteria often mandated by end-users in the renewable energy sector.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating FATSFI into their formulations, we have compiled answers to common inquiries regarding the material's properties and handling. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for R&D decision-making. Understanding these technical nuances is essential for optimizing device performance and troubleshooting potential integration challenges during the pilot testing phase.
Q: What are the primary advantages of FATSFI over traditional alkali metal TSFI salts?
A: Unlike hygroscopic and unstable lithium or sodium TSFI salts, FATSFI utilizes a formamidine cation that significantly improves moisture resistance, thermal stability, and interfacial charge extraction in photovoltaic layers.
Q: How is the 99.9% purity specification achieved in this synthesis?
A: The process leverages volatile byproducts like acetic acid which are removed via distillation, or insoluble silver halides removed by filtration, followed by a rigorous recrystallization step using anti-solvents.
Q: Is this synthesis method scalable for industrial battery additive production?
A: Yes, the reaction proceeds rapidly at room temperature within 20 minutes using common solvents like ethanol, eliminating the need for extreme heating or cooling, which facilitates easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FATSFI Supplier
As the global demand for high-efficiency perovskite solar cells and advanced lithium batteries accelerates, securing a dependable source of high-purity charge transport additives is critical for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your development efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling via NMR and MS analysis to guarantee that every batch meets the exacting standards required for electronic grade materials. We understand the critical nature of your timelines and are committed to delivering materials that enable your team to focus on innovation rather than supply chain uncertainties.
We invite you to engage with our technical procurement team to discuss how our optimized FATSFI manufacturing process can drive value for your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient synthesis routes can lower your overall material costs while enhancing device performance. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique formulation requirements, ensuring a seamless transition from laboratory research to full-scale commercial deployment.
