Advanced Synthesis of Tricyclodecenyl Alcohol: Technical Breakthroughs for Commercial Manufacturing
Advanced Synthesis of Tricyclodecenyl Alcohol: Technical Breakthroughs for Commercial Manufacturing
The chemical industry is constantly evolving towards more efficient and sustainable synthesis pathways, particularly for high-value intermediates like tricyclodecenyl alcohol. A significant advancement in this domain is detailed in patent CN109912410B, which discloses a novel method for preparing tricyclodecenyl alcohol and its key reaction intermediate. This technology represents a paradigm shift from traditional harsh hydration processes to a milder, two-step sequence involving trifluoroacetic acid addition followed by hydrolysis. For R&D directors and procurement specialists, understanding this transition is critical, as it offers a pathway to higher purity products with significantly reduced operational complexity. The patent highlights a process that not only achieves yields exceeding 94 percent but also ensures product purity reaching 98 percent, addressing long-standing challenges in the fragrance and resin industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tricyclodecenyl alcohol has relied heavily on the direct hydration of dicyclopentadiene using strong mineral acids. As illustrated in the traditional pathway, this typically involves reacting dicyclopentadiene with 20-25 percent sulfuric acid at elevated temperatures around 100 degrees Celsius. While conceptually simple, this legacy approach suffers from severe drawbacks that impact both product quality and operational safety. The use of large quantities of corrosive sulfuric acid generates substantial acidic wastewater, creating significant environmental compliance burdens and waste treatment costs. Furthermore, the harsh reaction conditions often lead to side reactions and polymerization, resulting in lower yields typically ranging from 75 to 85 percent. Perhaps most critically for supply chain efficiency, the crude product obtained from this method requires purification via reduced pressure distillation, an energy-intensive unit operation that poses risks of thermal decomposition for sensitive unsaturated compounds.
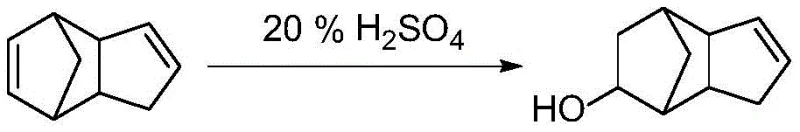
The Novel Approach
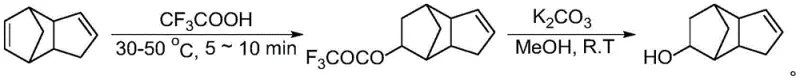
In stark contrast to the legacy hydration routes, the innovative methodology described in the patent utilizes a strategic addition-elimination sequence that fundamentally alters the process landscape. The new route begins with the electrophilic addition of trifluoroacetic acid to dicyclopentadiene under remarkably mild conditions, specifically between 30-50 degrees Celsius, and completes within a mere 5 to 10 minutes. This initial step is performed without any solvent, drastically reducing the volume of reaction mass and eliminating the need for solvent recovery systems. The resulting intermediate, tricyclodecenyl trifluoroacetate, is a stable solid that can be isolated and purified through simple recrystallization rather than distillation. This physical state change is a game-changer for quality control, allowing for the removal of impurities before the final hydrolysis step, thereby ensuring the high purity of the final alcohol product without thermal stress.
Mechanistic Insights into Trifluoroacetic Acid Addition and Hydrolysis
The core of this technological breakthrough lies in the specific reactivity of trifluoroacetic acid (TFA) as both a reactant and a catalyst carrier. In the first stage, the electron-rich double bond of the norbornene moiety in dicyclopentadiene undergoes electrophilic attack by the proton from TFA. This generates a carbocation intermediate which is immediately trapped by the trifluoroacetate anion, forming the ester linkage. The use of TFA is advantageous due to its strong acidity which facilitates rapid reaction kinetics at low temperatures, yet its conjugate base is a good leaving group for the subsequent step. The reaction stoichiometry is carefully controlled with a molar ratio of dicyclopentadiene to TFA between 1:1 and 1:2 to ensure complete conversion while minimizing excess reagent waste. The absence of solvent further drives the reaction equilibrium forward and enhances the collision frequency between reactant molecules, contributing to the exceptionally short reaction time observed.
Impurity control is meticulously managed through the physical properties of the intermediate. Unlike the oily mixtures typical of acid hydration, the tricyclodecenyl trifluoroacetate crystallizes readily, allowing for rigorous purification via recrystallization from anhydrous ethanol. This solid-state purification effectively segregates unreacted starting materials and polymeric byproducts that would otherwise carry through to the final product. The second stage involves a mild base-catalyzed hydrolysis using potassium carbonate in methanol at room temperature. This saponification cleaves the ester bond to release the free alcohol and potassium trifluoroacetate. Because the intermediate was already purified, the hydrolysis proceeds cleanly, and the final workup simply involves removing the methanol and extracting the product, bypassing the need for complex distillation columns and preserving the structural integrity of the sensitive bicyclic framework.
How to Synthesize Tricyclodecenyl Alcohol Efficiently
Implementing this synthesis route requires precise control over the addition rate and temperature to manage the exothermic nature of the acid addition, although the overall thermal load is much lower than traditional methods. The process flow dictates that after the rapid addition reaction, the mixture is quenched with water and extracted, followed by the critical recrystallization step which defines the purity profile. Once the pure intermediate is secured, the hydrolysis is straightforward, requiring only stirring at ambient conditions. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the optimal conditions derived from the patent examples.
- Perform an addition reaction between dicyclopentadiene and trifluoroacetic acid at 30-50°C for 5-10 minutes without solvent to form tricyclodecenyl trifluoroacetate.
- Purify the resulting solid intermediate through extraction, washing, drying, and recrystallization from anhydrous ethanol.
- Hydrolyze the purified intermediate using potassium carbonate in methanol at room temperature to obtain high-purity tricyclodecenyl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of the vacuum distillation step represents a massive reduction in capital expenditure (CAPEX) and operating expenditure (OPEX), as distillation units are among the most energy-consuming assets in a chemical plant. By replacing thermal separation with crystallization, the process significantly lowers utility consumption, particularly steam and cooling water, leading to substantial cost savings in manufacturing. Furthermore, the solvent-free nature of the primary reaction step reduces the inventory of hazardous organic solvents required on-site, simplifying regulatory compliance and lowering insurance premiums associated with flammable storage. These factors combine to create a leaner, more cost-effective production model that enhances margin potential in competitive markets.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the simplification of the downstream processing train. Traditional methods require expensive distillation equipment to separate the product from tarry byproducts and water, whereas this new method relies on crystallization, a far cheaper unit operation. Additionally, the high selectivity of the trifluoroacetic acid addition minimizes the formation of heavy ends and polymers, increasing the effective yield of saleable product per ton of raw material input. The ability to operate at near-ambient temperatures also reduces the demand for high-grade heating media, further driving down variable production costs without compromising output quality.
- Enhanced Supply Chain Reliability: From a logistics perspective, the generation of a stable solid intermediate provides a strategic buffer for production scheduling. Unlike unstable reaction mixtures that must be processed immediately, the solid tricyclodecenyl trifluoroacetate can be stored, quality-checked, and accumulated before hydrolysis. This decoupling of the synthesis steps allows for greater flexibility in batch planning and reduces the risk of total batch loss due to equipment failure in a continuous line. Moreover, the use of common reagents like potassium carbonate and methanol ensures that raw material sourcing remains robust and unaffected by the supply volatility often seen with specialized catalysts or exotic reagents.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer due to the mild reaction conditions. The low temperature range of 30-50 degrees Celsius mitigates the risk of thermal runaway, a common concern in large-scale acid-catalyzed reactions. Environmentally, the process aligns with green chemistry principles by reducing waste generation; the absence of sulfuric acid eliminates the production of sulfate-containing wastewater, and the solvent-free step minimizes VOC emissions. These attributes facilitate easier permitting for new capacity expansions and ensure long-term operational continuity in regions with increasingly stringent environmental regulations regarding industrial effluent and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tricyclodecenyl alcohol synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity mechanisms, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or for procurement professionals assessing supplier capabilities.
Q: How does the new method improve purity compared to traditional sulfuric acid hydration?
A: The new method utilizes a solid intermediate, tricyclodecenyl trifluoroacetate, which can be purified via recrystallization. This eliminates the need for reduced pressure distillation required in traditional methods, preventing thermal degradation and achieving purities up to 98%.
Q: What are the reaction conditions for the addition step?
A: The addition reaction occurs under mild conditions, specifically at temperatures between 30-50°C for a short duration of 5-10 minutes. Crucially, this step is performed without any solvent, reducing waste and simplifying the process.
Q: Why is this process considered more environmentally friendly?
A: This process avoids the use of large amounts of concentrated sulfuric acid and high temperatures (100°C) associated with older methods. Additionally, the solvent-free nature of the first step and the elimination of energy-intensive vacuum distillation significantly reduce the environmental footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricyclodecenyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global fine chemicals market. Our technical team has thoroughly analyzed the methodology described in CN109912410B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of tricyclodecenyl alcohol meets the highest standards required for fragrance and resin applications. Our commitment to process excellence allows us to deliver high-purity intermediates consistently, leveraging our expertise in crystallization and mild hydrolysis techniques to maximize yield and minimize impurities.
We invite you to collaborate with us to optimize your supply chain for this valuable intermediate. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your production needs are met with the most efficient and economically viable solutions available in the industry today.
