Advanced Synthesis of 5-Fluoro-4-Imino-Pyrimidinone Intermediates for Scalable Fungicide Production
Advanced Synthesis of 5-Fluoro-4-Imino-Pyrimidinone Intermediates for Scalable Fungicide Production
The global demand for high-efficacy fungicides continues to drive innovation in agrochemical intermediate manufacturing. A pivotal advancement in this sector is detailed in Chinese Patent CN111362881A, which discloses robust methods for preparing 5-fluoro-4-imino-3-(alkyl/substituted alkyl)-1-(arylsulfonyl)-3,4-dihydropyrimidin-2(1H)-ones. These compounds serve as critical scaffolds for next-generation agricultural protection agents. For R&D directors and procurement specialists, understanding the nuances of this synthetic pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality. The patent outlines a sophisticated approach that balances chemical efficiency with practical scalability, addressing long-standing challenges in regioselectivity and purification that have historically plagued the production of N3-substituted-N1-sulfonyl-5-fluoropyrimidinones.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating N-substituted pyrimidinone derivatives often suffer from poor regioselectivity and harsh reaction conditions. Conventional alkylation strategies frequently result in complex mixtures of N1 and N3 isomers, necessitating expensive and time-consuming chromatographic separations that are ill-suited for large-scale commercial production. Furthermore, older methodologies often rely on strong, non-selective bases that can degrade sensitive fluorinated heterocycles or lead to over-alkylation, producing undesired dialkylated by-products that reduce overall yield. The inability to effectively recycle unreacted starting materials in these legacy processes contributes to significant waste generation and inflated manufacturing costs, creating bottlenecks for supply chain heads seeking cost reduction in agrochemical intermediate manufacturing.
The Novel Approach
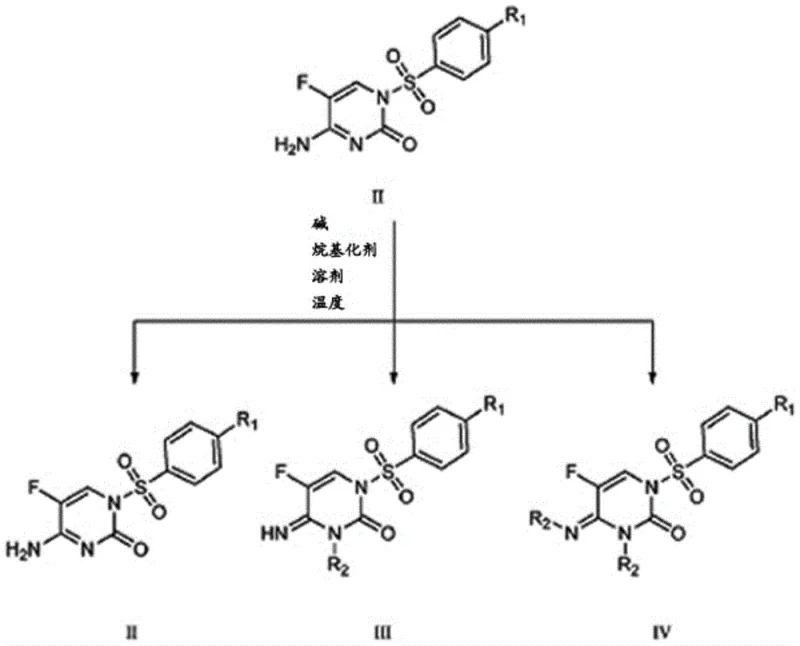
The methodology presented in the patent data introduces a refined, base-mediated alkylation protocol that significantly enhances process control. By utilizing specific alkali metal carbonates or alkoxides in polar aprotic solvents, the reaction selectively targets the N3 position of the pyrimidinone ring while minimizing N1 displacement or dialkylation. This novel approach allows for the formation of product mixtures containing the desired Formula III compound alongside unreacted Formula II intermediate, which can be easily separated through simple precipitation and recrystallization techniques rather than chromatography. This shift from complex purification to physical separation represents a major leap forward in process chemistry, enabling the recovery and recycling of valuable intermediates and drastically simplifying the downstream processing workflow for industrial applications.
Mechanistic Insights into Base-Mediated Selective Alkylation
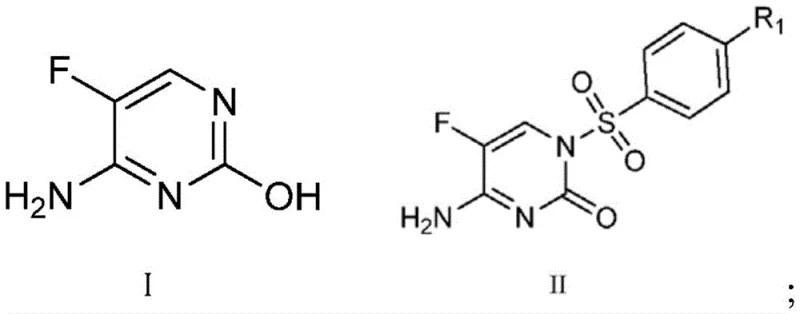
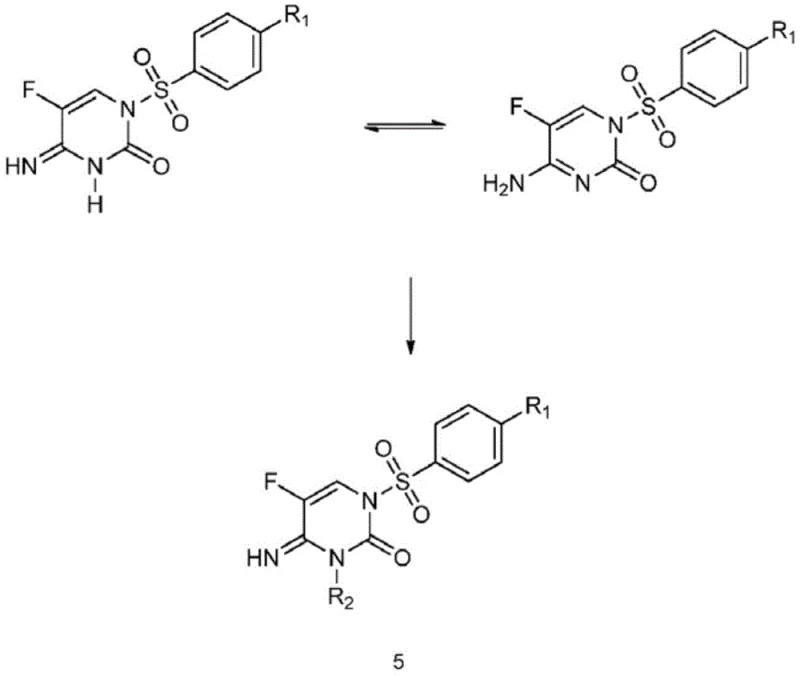
The core of this synthetic strategy lies in the precise manipulation of tautomeric equilibria and nucleophilicity within the pyrimidinone system. The process begins with the conversion of 5-fluorocytosine (Formula I) into the N1-sulfonyl protected intermediate (Formula II) using bis-N,O-trimethylsilylacetamide (BSA). The silyl protection step is critical as it activates the nitrogen center for sulfonylation while preventing O-sulfonylation, ensuring the formation of the correct N1-S bond architecture required for subsequent fungicidal activity. Following this, the introduction of a mild base, such as lithium carbonate or potassium tert-butoxide, generates a specific anionic species at the N3 position. The choice of base and solvent polarity dictates the hardness and softness of this nucleophile, thereby steering the reaction towards the desired N3-alkylation pathway when exposed to alkyl halides like methyl iodide or benzyl bromide.
Impurity control is inherently built into the reaction design through stoichiometric optimization. The patent data indicates that maintaining a molar ratio of Formula II compound to base between 3:1 and 1:1, and to alkylating agent between 1:1 and 3:1, is vital for suppressing the formation of the dialkylated Formula IV by-product. Unlike traditional methods where dialkylation is a dominant side reaction requiring difficult removal, this process tolerates a degree of unreacted starting material. The mechanistic understanding here allows manufacturers to tune the reaction to stop at the mono-alkylated stage, leveraging the differential solubility of the components for purification. This deep control over the reaction landscape ensures that the final high-purity pharmaceutical intermediate meets stringent specifications without the need for resource-intensive cleanup steps.
How to Synthesize 5-Fluoro-4-Imino-Pyrimidinone Efficiently
The synthesis of these valuable heterocyclic compounds follows a logical two-stage progression designed for reproducibility and scale-up. Initially, the 5-fluorocytosine starting material undergoes silylation and sulfonylation to establish the core N1-aryl sulfonyl framework. Subsequently, the N3-position is functionalized through a controlled alkylation event. The detailed operational parameters, including specific temperature ranges from 22°C to 60°C and solvent systems involving DMF or acetonitrile, are optimized to maximize yield while maintaining safety profiles suitable for pilot and commercial plants. For a comprehensive breakdown of the exact reagent quantities, addition rates, and workup procedures required to execute this synthesis in a GMP environment, please refer to the standardized protocol below.
- Protect 5-fluorocytosine with BSA and react with arylsulfonyl chloride to form the N1-sulfonyl intermediate.
- Treat the intermediate with an alkali metal carbonate or alkoxide base in a polar aprotic solvent.
- Add an alkylating agent such as methyl iodide at controlled temperatures (22°C to 60°C) to achieve selective N3-alkylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthetic route offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the substantial cost savings achieved through simplified purification. By eliminating the reliance on preparative chromatography and replacing it with filtration and crystallization, the process drastically reduces solvent consumption and processing time. This efficiency translates directly into a lower cost of goods sold (COGS), making the final agrochemical active ingredient more competitive in the global market. Furthermore, the ability to recycle unreacted Formula II intermediate from the reaction mixture means that raw material utilization is maximized, reducing waste disposal costs and enhancing the overall sustainability profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps is a game-changer for cost structures. Traditional methods often require large volumes of expensive eluents and specialized equipment to separate isomers, which this new method avoids entirely by leveraging precipitation techniques. The use of readily available and inexpensive bases like lithium carbonate or potassium carbonate further drives down reagent costs compared to exotic catalysts. Additionally, the mild reaction temperatures (often near ambient or slightly elevated) reduce energy consumption for heating and cooling, contributing to a leaner and more economically viable production model that supports long-term margin stability.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines to downstream formulators. Because the reaction tolerates a range of conditions and allows for the recycling of intermediates, the risk of batch failure due to minor deviations is significantly mitigated. The starting materials, such as 5-fluorocytosine and common arylsulfonyl chlorides, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. This reliability allows supply chain heads to plan inventory with greater confidence and reducing lead time for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to multi-ton commercial production is facilitated by the use of standard unit operations like stirred tank reactors and filter dryers. The avoidance of hazardous reagents and the minimization of organic solvent waste align with increasingly strict environmental regulations worldwide. The aqueous workup procedures described, utilizing sodium thiosulfate and water/acetonitrile mixtures, simplify wastewater treatment requirements. This environmental compatibility not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the manufacturing site, making it a preferred partner for multinational agrochemical corporations seeking green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their own production lines or for sourcing partners who utilize these methods.
Q: How does this patent address the issue of dialkylation by-products?
A: The patent describes controlling the molar ratios of the base and alkylating agent to favor the formation of the mono-alkylated product (Formula III) over the dialkylated by-product (Formula IV). Additionally, it highlights that mixtures of unreacted starting material (Formula II) and product (Formula III) can be separated via precipitation, allowing for the recycling of starting materials.
Q: What are the preferred solvents for this alkylation reaction?
A: The process utilizes polar aprotic solvents such as N,N-dimethylformamide (DMF), dimethylsulfoxide (DMSO), or acetonitrile (CH3CN). DMF is specifically highlighted in examples for achieving high yields with lithium carbonate bases.
Q: Why is BSA used in the initial step of the synthesis?
A: Bis-N,O-trimethylsilylacetamide (BSA) is used to protect the hydroxyl group of the pyrimidine ring, converting it into a silyl ether. This activation facilitates the subsequent nucleophilic attack on the arylsulfonyl chloride, ensuring clean formation of the N1-sulfonyl bond without side reactions at the oxygen atom.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Fluoro-4-Imino-Pyrimidinone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of effective crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-fluoro-4-imino-pyrimidinone derivative meets the exacting standards required by the global agrochemical industry. Our commitment to process excellence means we can deliver the consistency and volume necessary to support your product lifecycle from clinical trials to market launch.
We invite you to collaborate with us to optimize your supply chain for these essential building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your project's success and drive value for your organization.
