Revolutionizing LCD Material Production: High-Yield Synthesis of Trans-Trans Cyclohexyl Alcohol Intermediates
Revolutionizing LCD Material Production: High-Yield Synthesis of Trans-Trans Cyclohexyl Alcohol Intermediates
The rapid evolution of display technology, particularly in the realm of Thin Film Transistor Liquid Crystal Displays (TFT-LCD), demands intermediates of exceptional purity and specific stereochemical configurations. Patent CN101704724B introduces a groundbreaking methodology for the preparation of high-proportion trans,trans-4-(4'-alkylcyclohexyl)cyclohexyl alcohol compounds, which serve as vital precursors for low-viscosity liquid crystal monomers. This technical breakthrough addresses the longstanding inefficiencies in synthesizing these critical building blocks, offering a pathway that significantly enhances both yield and stereoselectivity. By shifting away from traditional phenol hydrogenation routes, this innovation provides a robust solution for manufacturers seeking to optimize their supply chains for high-performance electronic chemicals. The method leverages a sophisticated combination of borohydride reduction and Lewis acid-catalyzed isomerization to secure the thermodynamically stable trans,trans configuration.
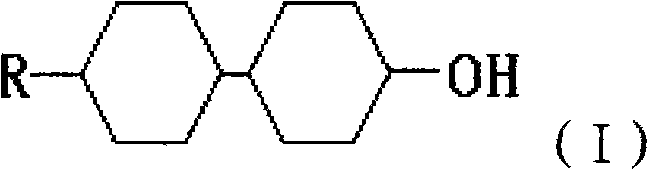
For R&D directors and procurement specialists, understanding the structural integrity of these molecules is paramount. The target compound, characterized by the general formula (I) where R represents a C1-C10 alkyl group, acts as a key viscosity reducer in liquid crystal formulations. The precise control over the alkyl chain length, ranging from methyl to heptyl groups, allows for fine-tuning the physical properties of the final display material. This patent outlines a process that not only simplifies the synthetic route but also ensures that the resulting intermediates meet the rigorous standards required for next-generation displays. As a reliable liquid crystal intermediate supplier, recognizing the value of such patented processes is essential for maintaining a competitive edge in the global market for display and optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans,trans-alkyl bicyclohexyl alcohols relied heavily on the hydrogenation of trans-alkylcyclohexylphenols. This conventional approach, while chemically feasible, suffers from severe stereochemical drawbacks that hinder large-scale efficiency. The hydrogenation process typically yields a mixture of cis and trans isomers, with the cis-isomer being the dominant product, often comprising up to 55% of the mixture. To isolate the desired trans,trans-isomer, manufacturers were forced to employ multiple, labor-intensive recrystallization steps, which drastically reduced the overall yield to approximately 20%. Furthermore, older strategies involved oxidizing these alcohol mixtures back into ketones using hazardous reagents like chromic acid or potassium permanganate, creating significant environmental liabilities and waste disposal challenges. These inefficiencies created a bottleneck in the production of high-quality liquid crystal materials, driving up costs and limiting supply availability for the booming electronics sector.
The Novel Approach
In stark contrast, the methodology described in Patent CN101704724B circumvents these historical bottlenecks by utilizing trans-4-alkylcyclohexylcyclohexyl ketone as the direct starting material. This strategic shift allows for a direct reduction and isomerization sequence that inherently favors the formation of the desired trans,trans configuration. The process involves dissolving the ketone in a solvent system, typically tetrahydrofuran, and reacting it with a borohydride reducing agent in ethanol. Following the reduction, a Lewis acid, such as aluminum trichloride, is introduced to catalyze the isomerization, effectively converting any formed cis-isomers into the more stable trans-form. This one-pot style progression eliminates the need for the polluting oxidation steps associated with previous methods. The result is a streamlined workflow that achieves yields exceeding 80% and a trans-isomer content of over 90%, representing a monumental leap in process efficiency and cost reduction in electronic chemical manufacturing.

Mechanistic Insights into Borohydride Reduction and Lewis Acid Isomerization
The core of this technological advancement lies in the precise orchestration of reduction and isomerization mechanisms. Initially, the borohydride species, whether sodium borohydride or potassium borohydride, acts as a nucleophile attacking the carbonyl carbon of the trans-4-alkylcyclohexylcyclohexyl ketone. This step is conducted at controlled low temperatures, typically between 0°C and 40°C, to manage the exothermic nature of the reaction and prevent side reactions. The choice of solvent system is critical; a mixture of tetrahydrofuran and ethanol provides the optimal polarity to solubilize both the organic ketone and the inorganic borohydride salt, ensuring homogeneous reaction kinetics. This careful control over the reduction environment sets the stage for the subsequent stereochemical correction, ensuring that the initial alcohol formed is primed for isomerization rather than remaining trapped in a kinetic cis-state.
Following the reduction, the introduction of a Lewis acid like aluminum trichloride triggers a pivotal isomerization event. The Lewis acid coordinates with the hydroxyl group of the newly formed alcohol, facilitating a reversible equilibration between the cis and trans conformers. Since the trans,trans-isomer is thermodynamically more stable due to reduced steric hindrance between the axial substituents on the cyclohexane rings, the equilibrium shifts decisively towards the trans product. This mechanism effectively "corrects" any stereochemical errors from the reduction step, driving the trans-content to over 90%. Subsequent acidic hydrolysis quenches the reaction and releases the free alcohol. This dual-step mechanism ensures that impurities, particularly the unwanted cis-isomers, are minimized to levels acceptable for high-end applications, thereby securing the high-purity liquid crystal intermediate status required by discerning customers.
How to Synthesize Trans-Trans-4-(4'-Alkylcyclohexyl)Cyclohexyl Alcohol Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters outlined in the patent to guarantee reproducibility and quality. The process begins with the preparation of two distinct solutions: the ketone substrate dissolved in an organic solvent A, and the borohydride reducing agent dissolved in organic solvent B. These solutions are mixed under inert conditions with precise temperature control to initiate the reduction. Once the reduction is complete, the Lewis acid is added rapidly to induce isomerization, followed by a controlled acidic hydrolysis step to finalize the transformation. The workup involves standard extraction techniques using solvents like toluene or ethyl acetate, followed by washing to neutral pH and drying. Finally, a crucial recrystallization step using alcohols such as ethanol or methanol polishes the crude product to pharmaceutical-grade purity. For detailed operational parameters and safety protocols, please refer to the standardized guide below.
- Dissolve trans-4-alkylcyclohexylcyclohexyl ketone in an organic solvent such as tetrahydrofuran and prepare a separate solution of sodium borohydride in ethanol.
- Mix the solutions at controlled low temperatures (0°C to 40°C) to effect reduction, followed by the addition of a Lewis acid like aluminum trichloride for isomerization.
- Perform acidic hydrolysis, extract the organic phase, wash to neutrality, dry, and recrystallize to obtain the final product with purity exceeding 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain heads looking to stabilize their sourcing of critical display materials. The most immediate benefit is the dramatic improvement in process yield, which translates directly into better resource utilization and lower raw material consumption per kilogram of finished product. By eliminating the need for multiple recrystallizations that plagued the old phenol-based routes, the new method significantly reduces processing time and energy consumption. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a steady flow of materials for LCD panel production lines without the risk of bottlenecks caused by low-yield purification steps. Furthermore, the simplified workflow reduces the operational complexity, making the process easier to scale from pilot batches to multi-ton commercial production.
- Cost Reduction in Manufacturing: The elimination of hazardous oxidation reagents and the reduction in solvent usage for recrystallization lead to substantial cost savings. By avoiding the use of expensive and toxic heavy metal oxidants like chromium VI, facilities can lower their waste treatment costs and reduce the regulatory burden associated with hazardous material handling. The higher yield means less starting material is wasted, optimizing the cost of goods sold (COGS) for every batch produced. Additionally, the ability to use common, inexpensive solvents like ethanol and tetrahydrofuran further drives down the variable costs of production, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the dependency on complex, multi-step purification sequences that are prone to failure. With a simpler process flow and higher tolerance for variation, manufacturers can maintain consistent output levels even during periods of high demand. The use of readily available starting materials, such as trans-4-alkylcyclohexylcyclohexyl ketone, ensures that raw material sourcing remains stable and unaffected by niche supply constraints. This reliability is crucial for long-term contracts with major display manufacturers who require guaranteed delivery schedules to keep their assembly lines running smoothly without interruption.
- Scalability and Environmental Compliance: This method is inherently designed for scalability, with reaction conditions that are easily managed in large-scale reactors. The avoidance of cryogenic temperatures (the process operates comfortably between 0°C and 40°C) reduces the energy load on cooling systems, facilitating easier commercial scale-up of complex liquid crystal intermediates. Moreover, the green chemistry aspects of the process, specifically the removal of heavy metal oxidants, align perfectly with modern environmental regulations and corporate sustainability goals. This compliance reduces the risk of regulatory shutdowns and enhances the brand reputation of suppliers who adopt this cleaner, more sustainable manufacturing technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in Patent CN101704724B, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains. The answers highlight the balance between high purity, yield, and operational simplicity that defines this new method.
Q: What is the primary advantage of this new synthesis method over traditional phenol hydrogenation?
A: The new method utilizes trans-4-alkylcyclohexylcyclohexyl ketone as a starting material, bypassing the low-yield hydrogenation of phenols which typically produces a cis-dominant mixture. This novel route achieves a trans-isomer proportion of over 90% and overall yields exceeding 80%, compared to roughly 20% in older methods.
Q: How does this process impact the environmental footprint of liquid crystal manufacturing?
A: This process eliminates the need for hazardous oxidation steps involving chromic acid or potassium permanganate, which were previously required to convert cis-alcohol mixtures into ketones. By starting directly with the ketone and using cleaner reduction agents like borohydrides, the method significantly reduces toxic waste generation.
Q: What purity levels can be achieved for TFT-LCD applications?
A: Through optimized reaction conditions and a final recrystallization step using solvents like ethanol or methanol, the process consistently delivers product purity of 99.8% or higher. This high purity is critical for ensuring the low viscosity and high stability required in advanced TFT-LCD panels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Trans-4-(4'-Alkylcyclohexyl)Cyclohexyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global electronics manufacturers. We are committed to delivering products with stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to verify that every batch meets the >99.8% purity standard required for TFT-LCD applications. Our infrastructure is designed to handle the specific solvent systems and temperature controls necessary for this borohydride reduction and isomerization process, guaranteeing consistency and quality in every shipment.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-yield method for your specific production volumes. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to drive the next generation of liquid crystal materials forward with superior chemistry and reliable supply.
