Advanced Two-Step Synthesis of Trans-Bicyclohexane Derivatives for High-Performance Liquid Crystal Applications
Advanced Two-Step Synthesis of Trans-Bicyclohexane Derivatives for High-Performance Liquid Crystal Applications
The rapid evolution of display technologies, particularly STN and TFT liquid crystal displays, demands monomers with exceptional electro-optic properties, including low viscosity, high resistivity, and superior low-temperature compatibility. Central to achieving these performance metrics is the incorporation of rigid bicyclohexyl structures, specifically (trans)-4-alkyl-4'-alkyl-1,1'-bicyclohexane, which serve as critical building blocks in modern liquid crystal formulations. A groundbreaking approach detailed in patent CN111233605A introduces a highly efficient, two-step synthetic pathway that overcomes the historical limitations of yield, purity, and safety associated with traditional manufacturing methods. This innovation leverages a strategic combination of catalytic hydrogenation and Lewis acid-mediated isomerization to deliver high-purity products from readily available alkylcyclohexyl phenyl alkyl ketone precursors.
For R&D directors and procurement specialists seeking a reliable liquid crystal intermediate supplier, this patent represents a pivotal shift towards more sustainable and economically viable production models. By streamlining the synthesis into just two distinct chemical transformations, the method drastically reduces the cumulative loss of material often seen in multi-step sequences. The ability to control stereochemistry effectively ensures that the final product meets the rigorous purity specifications required for high-end electronic applications, thereby minimizing the need for costly downstream purification processes that typically erode profit margins in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
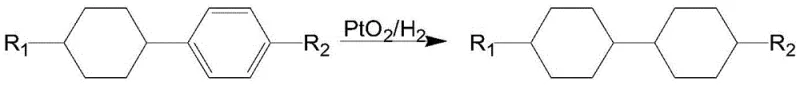
Historical approaches to synthesizing 4,4'-alkyl-substituted bicyclohexanes have been plagued by significant technical and economic hurdles that hinder efficient mass production. For instance, early methods described in patent DE19528085 relied on expensive platinum catalysts and necessitated extremely high temperatures and pressures, imposing severe demands on reactor equipment and driving up capital expenditure. Other routes, such as those disclosed in US4868341 and CN101580714, utilized Grignard reagents or lithium substitution reactions which inevitably generated substantial amounts of cis-configuration impurities, drastically reducing the yield of the desired trans-isomer.
Furthermore, methods involving long synthetic chains, such as those starting from alkyl bicyclohexyl formic acid (DE332173, CN104447171), suffer from poor overall yields due to the accumulation of losses at each step. These traditional pathways often involve hazardous reagents, strict anaerobic conditions, and difficult purification steps to remove self-coupling impurities, making them unsuitable for the cost-sensitive and safety-conscious environment of modern industrial chemistry. The cumulative effect of these drawbacks is a high production cost and a supply chain vulnerable to operational disruptions, failing to meet the growing demand for high-purity OLED material and liquid crystal precursors.
The Novel Approach
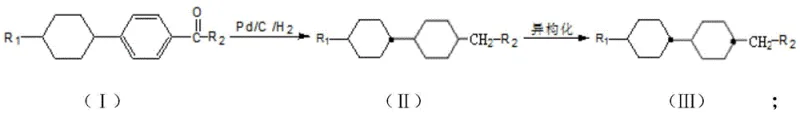
In stark contrast, the novel methodology presented in CN111233605A offers a streamlined solution that directly addresses these inefficiencies by utilizing a simple two-step reaction sequence starting from alkyl cyclohexyl phenyl alkyl ketone. The first step involves a robust catalytic hydrogenation using accessible catalysts like palladium on carbon or rhodium on carbon, operating under moderate conditions of 90-160°C and 2-6 MPa, which are far less demanding than the extreme parameters of prior art. This initial transformation efficiently converts the aromatic ring into a cyclohexane ring, generating a mixture of cis and trans isomers that serves as the direct precursor for the subsequent refinement step.
The second step employs a sophisticated Lewis acid-catalyzed isomerization in the presence of an alcohol, which selectively drives the equilibrium toward the thermodynamically stable trans-configuration. This clever chemical maneuver effectively resolves the stereochemical issues that plagued earlier Grignard-based routes, achieving trans-isomer contents exceeding 92% without the need for complex separation techniques. By eliminating the need for dangerous organometallic reagents and reducing the total number of synthetic operations, this approach not only enhances safety but also delivers cost reduction in electronic chemical manufacturing through improved atom economy and reduced waste generation.
Mechanistic Insights into Catalytic Hydrogenation and Lewis Acid Isomerization
The core of this synthesis lies in the precise control of reaction kinetics and thermodynamics during the hydrogenation and isomerization phases. In the first stage, the aromatic ring of the alkylcyclohexyl phenyl alkyl ketone undergoes heterogeneous catalytic hydrogenation. The choice of catalyst, such as Pd/C or Rh/C, is critical as it facilitates the addition of hydrogen across the double bonds of the benzene ring while preserving the integrity of the existing cyclohexane ring and the alkyl side chains. The reaction conditions, specifically the temperature range of 90-160°C and pressure of 2-6 MPa, are optimized to ensure complete conversion of the starting material while minimizing side reactions that could lead to ring opening or dealkylation.
Following hydrogenation, the resulting mixture of cis and trans isomers is subjected to a Lewis acid-catalyzed equilibration. Catalysts such as aluminum trichloride (AlCl3) or boron trifluoride (BF3) act as electron pair acceptors, coordinating with the cyclohexane rings to lower the energy barrier for conformational interconversion. In the presence of a proton source like tert-butanol or isopropanol, this system allows the less stable cis-isomers to reversibly open and reclose or flip into the more energetically favorable trans-configuration. Operating this step at low temperatures (-70 to -10°C) is essential to prevent degradation and to kinetically trap the product in the desired trans-state, ensuring high stereochemical purity essential for commercial scale-up of complex liquid crystal monomers.
How to Synthesize Trans-4-alkyl-4'-alkyl-1,1'-bicyclohexane Efficiently
The practical implementation of this synthesis requires careful attention to solvent selection and stoichiometric ratios to maximize yield and purity. The process begins by dissolving the ketone precursor in a solvent such as ethyl acetate or isopropanol, followed by hydrogenation in a stainless steel reactor. Once the intermediate is isolated, it is redissolved in a halogenated solvent like dichloromethane and treated with the Lewis acid catalyst under an inert nitrogen atmosphere to prevent moisture interference. Detailed standard operating procedures regarding exact dosing rates, quenching protocols, and workup strategies are critical for reproducibility and are outlined in the comprehensive guide below.
- Dissolve alkylcyclohexyl phenyl alkyl ketone in an organic solvent and perform catalytic hydrogenation using Pd/C or Rh/C at 90-160°C and 2-6 MPa to generate the mixed cis/trans bicyclohexane intermediate.
- Dissolve the hydrogenated intermediate in a halogenated solvent and treat with a Lewis acid (such as AlCl3 or BF3) and an alcohol at low temperatures (-70 to -10°C) to induce isomerization.
- Quench the reaction with aqueous acid, separate the organic phase, wash with bicarbonate solution, and remove the solvent under negative pressure to isolate the high-purity trans-configured product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience. The elimination of hazardous Grignard reagents and the avoidance of cryogenic lithium chemistry significantly reduce the safety risks associated with production, lowering insurance costs and minimizing the potential for costly shutdowns due to safety incidents. Moreover, the use of common, commercially available solvents and catalysts ensures that the supply chain remains robust, avoiding the bottlenecks often associated with specialized or proprietary reagents required by older methodologies.
- Cost Reduction in Manufacturing: The streamlined two-step process inherently reduces manufacturing costs by minimizing unit operations, which translates to lower labor, energy, and equipment utilization expenses. By avoiding the long synthetic routes of prior art that suffered from cumulative yield losses, this method maximizes the output per kilogram of raw material input. The ability to use cheaper hydrogenation catalysts like Pd/C instead of expensive platinum systems further drives down the variable cost of goods sold, enabling more competitive pricing strategies in the global market for display materials.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable raw materials, such as alkyl cyclohexyl phenyl alkyl ketone, mitigates the risk of supply disruptions that can occur with niche intermediates. The moderate reaction conditions (2-6 MPa) mean that the process can be executed in standard industrial hydrogenation reactors without requiring bespoke high-pressure vessels, facilitating faster technology transfer and scale-up. This flexibility allows manufacturers to respond more agilely to fluctuations in market demand, ensuring reducing lead time for high-purity display materials and maintaining consistent delivery schedules to downstream clients.
- Scalability and Environmental Compliance: From an environmental perspective, the shorter route generates less chemical waste and solvent consumption per unit of product, aligning with increasingly stringent global environmental regulations. The absence of heavy metal contaminants often associated with complex organometallic couplings simplifies wastewater treatment and product purification, reducing the burden on environmental compliance teams. This green chemistry profile not only future-proofs the production facility against regulatory changes but also enhances the brand value of the final product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of the operational advantages. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional Grignard-based routes?
A: Unlike traditional methods that rely on hazardous Grignard reagents and low-temperature lithiation which pose significant safety risks and operational difficulties, this patented method utilizes a safer two-step sequence of hydrogenation and isomerization. This eliminates the need for strict anaerobic conditions and dangerous organometallic reagents, significantly simplifying the production process and enhancing workplace safety while maintaining high product yields.
Q: How does the new process improve the stereochemical purity of the final liquid crystal monomer?
A: The process specifically addresses the issue of cis-configuration impurities common in prior art by incorporating a dedicated Lewis acid-catalyzed isomerization step. By utilizing catalysts like aluminum trichloride or boron trifluoride at controlled low temperatures, the reaction drives the equilibrium towards the thermodynamically more stable trans-configuration, achieving trans-isomer content exceeding 92% without the need for complex and yield-reducing purification steps associated with older methods.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for batch production and industrial scalability. It avoids the extreme high-pressure and high-temperature requirements of platinum-catalyzed methods found in older patents, operating instead at moderate pressures (2-6 MPa) and temperatures. Furthermore, the use of readily available raw materials and common solvents like ethyl acetate and dichloromethane ensures that the supply chain remains robust and cost-effective for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-alkyl-4'-alkyl-1,1'-bicyclohexane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance liquid crystal intermediates play in the next generation of display technologies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trans-bicyclohexane derivative performs flawlessly in your final electro-optic formulations.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating exactly how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the rapidly evolving electronic materials market.
