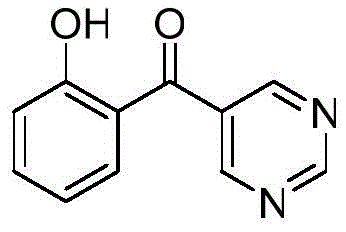
Advanced One-Step Synthesis of 2-Hydroxyphenyl-5-Pyrazinyl Ketone for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex heterocyclic scaffolds, which serve as critical building blocks for next-generation therapeutics. A recent technological breakthrough documented in patent CN112047891B introduces a highly streamlined synthesis method for 2-hydroxyphenyl-5-pyrazinyl ketone, a versatile pyrazine derivative with dual functionality as both a specialized intermediate and a homogeneous catalyst. This innovation addresses the longstanding challenges associated with constructing nitrogen-containing heterocycles, offering a robust alternative to traditional multi-step methodologies. By leveraging a specific palladium-complex catalytic system, the process transforms readily available chromone-3-formaldehyde into the target pyrazine structure in a single operational step. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains for pharmaceutical intermediates while reducing the overall environmental footprint of chemical manufacturing. The ability to generate high-value pyrazine cores with such atomic economy positions this technology as a cornerstone for modern API production strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyrazines and related aroyl-pyrazine structures has been fraught with synthetic inefficiencies that hinder large-scale commercial adoption. Traditional routes often rely on the condensation of alpha-dicarbonyl compounds with 1,2-diamines, which frequently suffer from poor regioselectivity and the formation of difficult-to-separate isomeric byproducts. Furthermore, existing literature, such as the methods cited in references regarding the transformation of substituted-3-formylchromones, often necessitates harsh reaction conditions or multiple protection-deprotection sequences to achieve the desired substitution pattern. These legacy processes not only inflate the cost of goods sold due to excessive solvent and reagent consumption but also introduce significant variability in batch-to-batch purity. For supply chain managers, the reliance on unstable precursors or sensitive reaction environments translates directly into increased risk of production delays and compromised quality control metrics, making the search for a more direct synthetic route a critical priority for maintaining competitive advantage in the fine chemical intermediates market.
The Novel Approach
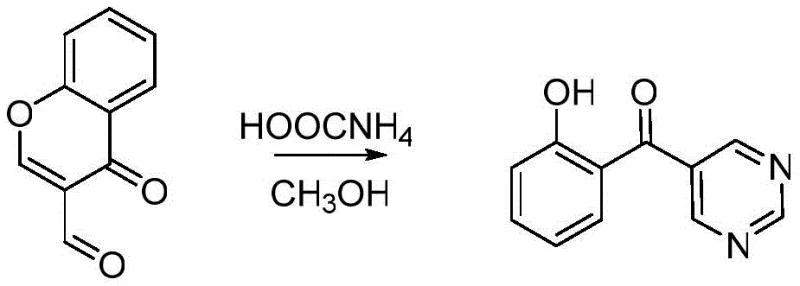
The methodology disclosed in the patent data revolutionizes this landscape by employing a direct cyclization strategy that bypasses the need for pre-functionalized diamine precursors. By utilizing chromone-3-formaldehyde as the sole carbon and oxygen source alongside ammonium formate as the nitrogen donor, the process achieves the construction of the pyrazine ring through an elegant cascade sequence. The reaction is mediated by a chiral palladium complex, which facilitates the ring-opening of the chromone scaffold followed by immediate cyclization, effectively stitching together the heterocyclic core in one pot. This approach not only simplifies the operational workflow but also significantly enhances the overall yield profile compared to stepwise alternatives. The simplicity of the workup, involving standard column chromatography with common solvent systems, further underscores the practical viability of this method for industrial scale-up, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Pyrazine Formation
At the heart of this synthetic advancement lies a sophisticated catalytic cycle driven by a bis-oxazolinyl palladium complex. The mechanism is presumed to initiate with the coordination of the palladium center to the carbonyl oxygen of the chromone-3-formaldehyde, activating the cyclic ether towards nucleophilic attack. Under the thermal energy provided by refluxing methanol, the strained pyrone ring undergoes cleavage, generating a reactive acyclic intermediate that retains the ortho-hydroxy phenyl motif. Simultaneously, the ammonium formate serves a dual role: acting as a source of ammonia for imine formation and providing the necessary reducing equivalents or dehydration driving force to aromatize the newly formed ring. This concerted transformation ensures that the nitrogen atoms are incorporated precisely at the 1 and 4 positions of the resulting pyrazine ring, avoiding the scrambling often seen in non-catalyzed thermal condensations. The ligand environment of the palladium catalyst is crucial in stabilizing these transient species, preventing polymerization or decomposition pathways that typically plague high-temperature heterocycle syntheses.
From an impurity control perspective, the specificity of this catalytic system is paramount for meeting the rigorous standards required for high-purity pharmaceutical intermediates. The reaction conditions are tuned to favor the thermodynamic product, minimizing the formation of open-chain oligomers or alternative cyclization isomers. The use of anhydrous methanol as the solvent medium plays a critical role in solubilizing the polar ammonium formate while maintaining the stability of the organometallic catalyst. Post-reaction analysis indicates that the primary impurities are largely unreacted starting materials, which are easily separated due to their distinct polarity differences from the target ketone. This clean reaction profile reduces the burden on downstream purification units, allowing for higher recovery rates of the final crystalline product. For quality assurance teams, this predictability in the impurity spectrum simplifies the validation process and ensures consistent compliance with regulatory specifications for drug substance precursors.
How to Synthesize 2-Hydroxyphenyl-5-Pyrazinyl Ketone Efficiently
Implementing this synthesis requires precise attention to stoichiometry and thermal management to maximize the efficiency of the palladium catalyst. The process begins with the careful weighing of the palladium complex, chromone-3-formaldehyde, and ammonium formate, which are then suspended in anhydrous methanol within a standard round-bottom flask. The mixture is subjected to prolonged heating under reflux, a condition essential for driving the equilibrium towards the cyclized product and ensuring complete consumption of the aldehyde precursor. Following the reaction period, the crude material is isolated and subjected to flash column chromatography, utilizing a binary solvent system of petroleum ether and dichloromethane to resolve the target compound from minor side products. The detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are outlined below for technical reference.
- Charge a reactor with palladium complex catalyst, chromone-3-formaldehyde, and ammonium formate in anhydrous methanol.
- Heat the mixture under reflux conditions for approximately 36 hours to ensure complete cyclization and conversion.
- Purify the crude light yellow crystals via column chromatography using a petroleum ether and dichloromethane solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical novelty. The consolidation of multiple synthetic steps into a single operation drastically reduces the cumulative processing time and the associated labor costs, leading to a leaner manufacturing footprint. By eliminating the need for isolating unstable intermediates, the process minimizes material handling risks and potential yield losses that occur during transfer between reaction vessels. Furthermore, the reliance on commodity chemicals like ammonium formate and methanol ensures that raw material sourcing remains stable and cost-effective, shielding production budgets from the volatility often associated with specialized reagents. This robustness in the supply base is critical for maintaining continuous operations in a global market where reliability is as valuable as price.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final product is not the only saving; the process avoids the use of expensive, custom-synthesized diamine precursors typically required for pyrazine construction. By utilizing chromone-3-formaldehyde, a commercially accessible feedstock, the raw material cost basis is significantly lowered. Additionally, the simplified workup procedure reduces the volume of solvents required for purification, directly impacting waste disposal costs and solvent recovery overheads. These cumulative efficiencies translate into a more competitive cost structure for the final intermediate, allowing downstream partners to optimize their own margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup, requiring only standard glassware and heating mantles, means that the technology can be rapidly deployed across multiple manufacturing sites without the need for specialized high-pressure equipment. This flexibility enhances supply security by allowing for distributed production capabilities, reducing the risk of single-point failures in the supply network. Moreover, the stability of the reagents allows for long-term storage and bulk purchasing, enabling companies to hedge against market fluctuations and ensure uninterrupted availability of this critical building block for time-sensitive drug development programs.
- Scalability and Environmental Compliance: The use of methanol as the primary solvent aligns well with green chemistry principles, as it is a relatively low-toxicity alcohol that is easily recycled. The absence of heavy metal waste streams, aside from the trace palladium which can be recovered, simplifies the environmental permitting process for new production lines. The high atom economy of the transformation ensures that the majority of the input mass is converted into the desired product, minimizing the generation of hazardous byproducts. This environmental compatibility is increasingly important for meeting the stringent sustainability criteria imposed by major pharmaceutical buyers and regulatory bodies worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this synthesis technology. These insights are derived directly from the experimental data and characterization results provided in the patent documentation, offering clarity on the practical aspects of producing and utilizing 2-hydroxyphenyl-5-pyrazinyl ketone. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing pipelines.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The patented method achieves the target pyrazine derivative in a single synthetic step from chromone-3-formaldehyde, eliminating the need for complex multi-step sequences typically required for constructing the pyrazine core.
Q: Can this compound be utilized as a catalyst in other organic transformations?
A: Yes, beyond its role as an intermediate, the compound demonstrates significant catalytic activity in the condensation of benzophenone imine with trimethylsilyl cyanide, achieving high conversion rates.
Q: What are the critical purification parameters for this process?
A: Effective isolation is achieved through column chromatography utilizing a specific 1:1 volume ratio of petroleum ether to dichloromethane, ensuring the removal of unreacted starting materials and side products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyphenyl-5-Pyrazinyl Ketone Supplier
As the demand for specialized heterocyclic intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies backed by rigorous quality systems. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art analytical instrumentation and stringent purity specifications protocols, guaranteeing that every batch of 2-hydroxyphenyl-5-pyrazinyl ketone meets the exacting standards required for pharmaceutical applications. We understand the critical nature of timeline adherence in drug development and are committed to delivering reliable supply continuity for our global partners.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined process. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your transition to this advanced intermediate is seamless and scientifically sound.
