Advanced Synthesis of 3,5-Dihalobenzotrifluoride for Scalable Pharmaceutical and Agrochemical Manufacturing
The chemical industry is constantly seeking more efficient pathways to synthesize critical fluorinated building blocks, particularly for the agrochemical and pharmaceutical sectors. A significant breakthrough in this domain is detailed in patent CN112110790A, which outlines a robust preparation method for 3,5-dihalobenzotrifluoride and its downstream derivative, 3'-chloro-5'-(trifluoromethyl)phenyl trifluoroacetone. This technology addresses the longstanding challenges of high production costs and complex processing associated with traditional halogenation methods. By leveraging a diazotization deamination strategy starting from readily available 3,5-dihalo-4-aminobenzotrifluoride, the process offers a streamlined route that maintains high yields while drastically simplifying operational requirements. For global procurement teams and R&D directors, understanding this methodology is crucial for securing a reliable supply chain of high-purity intermediates essential for next-generation pesticide and drug development.
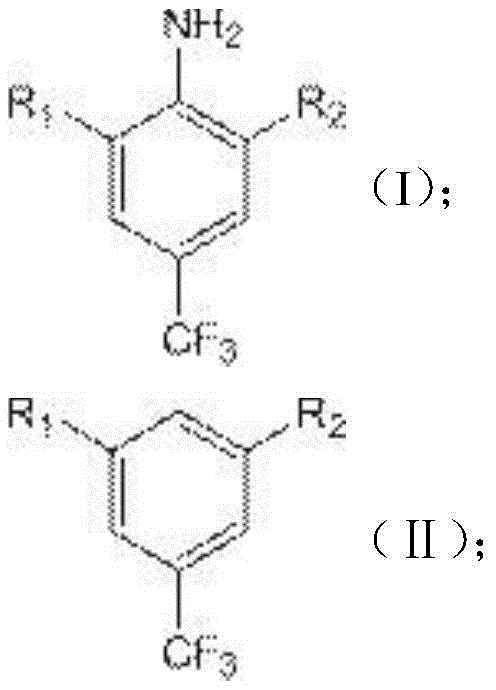
The limitations of conventional methods for producing 3,5-dihalobenzotrifluoride often stem from the reliance on direct halogenation of benzotrifluoride derivatives, which typically requires harsh reaction conditions, expensive catalysts, and generates difficult-to-separate isomeric byproducts. Traditional routes frequently involve multiple protection and deprotection steps, leading to cumulative yield losses and increased waste generation. Furthermore, the necessity for strict anhydrous conditions and specialized equipment to handle corrosive halogenating agents adds substantial capital expenditure and operational complexity. These factors collectively inflate the cost of goods sold (COGS) and create bottlenecks in the supply chain, making it difficult for manufacturers to respond agilely to market demands for agrochemical intermediates.
In stark contrast, the novel approach disclosed in the patent utilizes a diazotization deamination reaction that transforms an amino group directly into a hydrogen atom, effectively removing the nitrogen functionality to reveal the desired halogenated aromatic core. This method bypasses the need for complex halogen exchange reactions or direct electrophilic substitution on deactivated rings. By starting with 3,5-dihalo-4-aminobenzotrifluoride, where the halogen atoms are already in place, the synthesis focuses solely on the removal of the amino group. This strategic shift allows the reaction to proceed under mild acidic conditions using common reagents like sodium nitrite and hypophosphorous acid. The result is a process that is not only chemically elegant but also economically superior, offering a viable solution for cost reduction in pharmaceutical intermediates manufacturing without compromising on product quality or safety standards.
Mechanistic Insights into Diazotization Deamination and Grignard Coupling
The core of this synthesis lies in the precise control of the diazotization mechanism followed by a reductive elimination. Initially, the 3,5-dihalo-4-aminobenzotrifluoride is dissolved in a solvent such as toluene and acidified with sulfuric acid at low temperatures ranging from -10°C to 0°C. This step ensures the formation of the ammonium salt, which is crucial for the subsequent reaction with the nitrous acid reagent. Upon addition of sodium nitrite, the amino group is converted into a diazonium salt intermediate. This intermediate is inherently unstable and must be handled carefully; however, the presence of electron-withdrawing halogen groups and the trifluoromethyl moiety stabilizes the aromatic ring sufficiently to allow for controlled processing. The use of cuprous chloride as a catalyst facilitates the electron transfer processes required for the stability of the diazonium species prior to reduction.
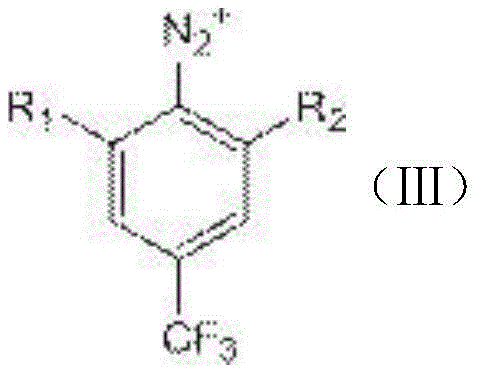
Following the formation of the diazonium intermediate, the system undergoes a reductive deamination using hypophosphorous acid. This step is critical as it replaces the diazo group with a hydrogen atom, regenerating the aromatic system as the final 3,5-dihalobenzotrifluoride product. The reaction temperature is allowed to rise gently to around 20°C, promoting the decomposition of the diazonium salt and the evolution of nitrogen gas, which serves as a driving force for the reaction. Impurity control is managed through the careful selection of acid concentration and the rate of reagent addition. By maintaining the sulfuric acid concentration between 70% and 95%, the process minimizes side reactions such as hydrolysis or polymerization. Furthermore, the subsequent work-up involving extraction with inorganic salt solutions effectively removes residual acids and metal catalysts, ensuring the final product achieves purity levels exceeding 99%, which is vital for downstream applications in sensitive catalytic cycles.
How to Synthesize 3,5-Dihalobenzotrifluoride Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and reagent stoichiometry to maximize yield. The process begins with the dissolution of the amine precursor in toluene, followed by careful acidification and cooling to prevent thermal runaway during diazotization. The addition of the nitrite source must be controlled to maintain the system below 0°C, ensuring the selective formation of the diazonium salt without degradation. Once the diazotization is complete, the reducing agent is introduced, and the mixture is stirred until gas evolution ceases, indicating the completion of the deamination. Detailed standardized synthetic steps see the guide below.
- Dissolve 3,5-dihalo-4-aminobenzotrifluoride in a solvent like toluene and cool to -10 to 0°C, then acidify with sulfuric acid to form the salt.
- Add a nitrous acid reagent (e.g., sodium nitrite) at low temperature to diazotize the amino group, forming the diazonium intermediate.
- Reduce the diazonium group using hypophosphorous acid and a cuprous chloride catalyst at 20°C to yield the final 3,5-dihalobenzotrifluoride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the significant reduction of raw material costs. The starting material, 3,5-dihalo-4-aminobenzotrifluoride, is commercially available at a fraction of the cost of specialized halogenating agents or protected intermediates required by older methods. Additionally, the reagents used in the process, such as sulfuric acid, sodium nitrite, and hypophosphorous acid, are commodity chemicals with stable global supply chains, reducing the risk of procurement disruptions. This stability allows for better long-term planning and inventory management, ensuring continuous production schedules for critical agrochemical and pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of cryogenic conditions (below -78°C) drastically lower energy consumption and equipment maintenance costs. The process operates efficiently at temperatures achievable with standard industrial chillers, removing the need for specialized low-temperature reactors. Furthermore, the simplified work-up procedure, which relies on liquid-liquid extraction and distillation rather than complex chromatography or crystallization steps, reduces solvent usage and waste disposal fees. These factors combine to create a leaner manufacturing process with a substantially lower cost base, enhancing the overall competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route that depends on widely available bulk chemicals, the supply chain becomes more resilient to regional shortages or geopolitical fluctuations affecting specialty reagents. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without extensive re-validation, providing flexibility in sourcing strategies. This adaptability is crucial for maintaining supply continuity for high-purity intermediates, especially when facing unexpected demand surges in the agricultural or healthcare sectors. The ability to source raw materials locally in various regions further shortens lead times and reduces logistics costs associated with long-distance transportation of hazardous chemicals.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, as evidenced by successful scale-up examples in the patent data. The generation of acidic wastewater is manageable through standard neutralization and treatment protocols, aligning with increasingly stringent environmental regulations. The absence of heavy metal residues in the final product simplifies the regulatory approval process for pharmaceutical applications, reducing the time to market for new drug candidates. Moreover, the high atom economy of the deamination step minimizes the generation of organic byproducts, contributing to a greener manufacturing footprint. This alignment with sustainability goals is increasingly important for corporate social responsibility initiatives and meeting the eco-friendly criteria of major multinational clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader production workflows.
Q: What are the key advantages of the diazotization deamination method for 3,5-dihalobenzotrifluoride?
A: The method utilizes cheap and readily available raw materials like 3,5-dihalo-4-aminobenzotrifluoride. It operates under mild conditions (below 0°C for diazotization, 20°C for reduction) without requiring anhydrous or oxygen-free environments, significantly lowering production costs and simplifying equipment requirements compared to traditional halogenation routes.
Q: How does this process ensure high purity for pharmaceutical intermediates?
A: The process achieves high purity (>99%) through controlled low-temperature reactions that suppress side products. The work-up involves extracting with inorganic salt solutions to remove acidic impurities and drying with agents like anhydrous magnesium sulfate, ensuring the final distillate meets stringent quality specifications for downstream synthesis.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up from laboratory to pilot scales (e.g., Example 35). The use of common solvents like toluene, stable reagents like sodium nitrite, and manageable acidic waste streams makes the process highly adaptable for commercial scale-up of complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dihalobenzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of advanced agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN112110790A can be translated into reliable industrial reality. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3,5-dihalobenzotrifluoride meets the exacting standards required for downstream Grignard reactions and nucleophilic additions. Our commitment to quality assurance ensures that your final active ingredients are free from detrimental impurities that could affect efficacy or safety profiles.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your project timelines and budgetary goals. Let us be your partner in delivering high-purity chemical solutions that drive innovation in the global life sciences industry.
