Advanced Synthetic Route for Upatinib Intermediates via Asymmetric Hydrogenation and Scalable Manufacturing
Introduction to Next-Generation Upatinib Intermediate Synthesis
The pharmaceutical landscape for JAK inhibitors continues to evolve, driving an urgent demand for efficient, high-purity synthetic routes for key intermediates like Upatinib. Patent CN109705011B discloses a groundbreaking methodology that addresses the critical bottlenecks of traditional synthesis, specifically focusing on the construction of the chiral pyrrolidine core. This technology leverages a novel acid-catalyzed cyclization followed by highly stereoselective asymmetric hydrogenation, offering a robust alternative to legacy resolution processes. For R&D and procurement leaders, this represents a significant opportunity to optimize supply chains for autoimmune disease therapeutics. The disclosed method not only simplifies the reaction sequence but also drastically improves the stereochemical control, ensuring that the final API meets stringent regulatory standards for chiral purity without the need for cumbersome chromatographic separations.
Furthermore, the versatility of this synthetic platform allows for the generation of multiple protected intermediates, such as Compound 7 and Compound 8, from a common precursor. This flexibility is paramount for process chemists aiming to diversify their pipeline or secure backup supply routes. By integrating mild reaction conditions with commercially available catalysts, the process minimizes safety risks associated with hazardous reagents often found in older literature. As we delve deeper into the technical specifics, it becomes evident that this approach is not merely an incremental improvement but a paradigm shift in how we approach the manufacture of complex heterocyclic pharmaceutical intermediates on a global scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Upatinib intermediates has been plagued by inefficiencies inherent in chemical resolution and unstable functional group manipulations. Prior art, such as WO 2015/061665 A1, relied heavily on chiral preparative columns to separate enantiomers, a technique that is notoriously difficult to scale and results in a theoretical maximum yield of only 50% for the desired isomer. Other approaches, documented in US 2013/0072470 A1, reported resolution yields as low as 13% with enantiomeric excess values that barely met acceptance criteria, creating massive waste streams and inflating production costs. Additionally, strategies involving triflate intermediates, as seen in WO2017066775, suffered from severe stability issues where the triflate group would hydrolyze under necessary alkaline conditions, leading to decomposition and yields dropping to approximately 20%. These legacy methods impose a heavy burden on manufacturing facilities, requiring extensive solvent usage and complex purification protocols that hinder commercial viability.
The Novel Approach
In stark contrast, the methodology outlined in CN109705011B introduces a streamlined cyclization strategy that bypasses these historical pitfalls entirely. The process initiates with the reaction of 2-pentynoate and N-(methoxymethyl)-N-(trimethylsilylmethyl)benzylamine under mild acidic catalysis, typically using trifluoroacetic acid or p-toluenesulfonic acid at temperatures between 10-30°C. This step constructs the pyrrolidine ring with high efficiency, achieving yields of up to 86% in the initial cyclization to form Compound 3. The subsequent transformations utilize robust chemistry that avoids sensitive leaving groups like triflates, thereby eliminating the decomposition pathways that plagued earlier attempts. By shifting the stereocontrol to a catalytic hydrogenation step rather than a resolution step, the theoretical yield ceiling is removed, allowing for near-quantitative conversion of the olefinic precursor into the chiral product.
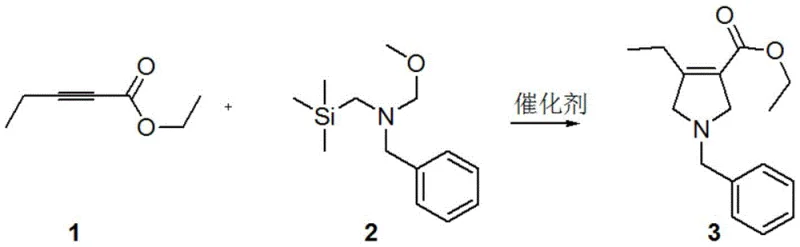
This novel route fundamentally alters the economic equation of Upatinib intermediate manufacturing. The ability to perform these reactions in common solvents like dichloromethane or ethyl acetate, followed by simple aqueous workups and crystallization, significantly reduces the operational complexity. The elimination of chiral column chromatography and the avoidance of low-yielding resolution steps mean that the same amount of raw material produces substantially more final product. For a reliable pharmaceutical intermediates supplier, adopting this technology translates directly into enhanced capacity and the ability to meet large-volume demands without proportional increases in facility footprint or waste treatment costs.
Mechanistic Insights into S-BINAP-Ru Catalyzed Asymmetric Hydrogenation
The cornerstone of this synthetic advancement is the application of transition metal-catalyzed asymmetric hydrogenation to establish the critical chiral centers at the C3 and C4 positions of the pyrrolidine ring. The patent specifies the use of an S-BINAP-Ru(OAc)2 complex, a sophisticated catalyst system known for its ability to differentiate between enantiotopic faces of a double bond with exceptional precision. In this mechanism, the ruthenium center coordinates with the chiral BINAP ligand to create a sterically defined environment that directs the addition of hydrogen atoms to the olefinic substrate (Compound 4 or 9). This coordination ensures that the hydrogen adds exclusively to one face of the molecule, resulting in the formation of the (3S, 4R) configuration with an HPLC chiral purity of not less than 99%. Such high fidelity in stereocontrol is essential for the biological activity of the final JAK inhibitor, as the wrong enantiomer could be inactive or even toxic.
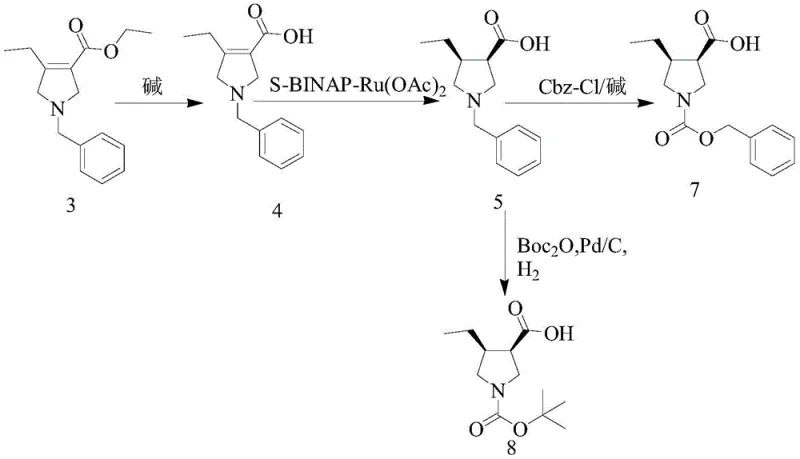
Beyond stereochemistry, the mechanistic design of this process inherently supports superior impurity control. The hydrogenation is conducted under controlled hydrogen pressures ranging from 20 to 60 atm and temperatures between 40-80°C, conditions that favor the desired reduction while minimizing side reactions such as over-reduction or racemization. The use of methanol as a solvent further aids in maintaining catalyst stability and solubility of the intermediates. Following the hydrogenation, the crude product can be purified via simple recrystallization from ethyl acetate, leveraging the high diastereomeric purity generated by the catalyst to exclude minor impurities. This contrasts sharply with resolution methods where impurities often co-crystallize or require repeated recrystallizations to achieve acceptable purity levels. The result is a process that delivers high-purity [high-purity pharmaceutical intermediates] consistently, batch after batch, reducing the risk of regulatory delays caused by impurity profile variations.
How to Synthesize Upatinib Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the cyclization and hydrogenation stages. The process begins with the preparation of the pyrrolidine core, followed by hydrolysis to the free acid, which serves as the substrate for the chiral step. The standardization of these steps is critical for reproducibility, especially when scaling from laboratory to pilot plant. The patent details specific molar ratios, such as using 1.5 to 3 equivalents of the amine component relative to the alkyne, and catalyst loadings of 0.01 to 0.1 molar equivalent for the ruthenium complex, providing a clear roadmap for process optimization. Operators must ensure strict exclusion of oxygen during the hydrogenation phase to prevent catalyst deactivation and maintain the high turnover numbers required for cost-effective production.
- Perform cyclization of 2-pentynoate and N-(methoxymethyl)-N-(trimethylsilylmethyl)benzylamine using trifluoroacetic acid catalyst to form the pyrrolidine core.
- Hydrolyze the ester group under alkaline conditions to generate the carboxylic acid intermediate.
- Execute asymmetric hydrogenation using S-BINAP-Ru(OAc)2 catalyst under high hydrogen pressure to establish chirality.
- Protect the nitrogen atom using benzyl chloroformate (Cbz-Cl) or Boc anhydride to finalize the intermediate structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By replacing multi-step resolution sequences with a direct catalytic asymmetric synthesis, the number of unit operations is significantly decreased. This reduction in processing steps means less equipment occupancy time, lower labor costs, and diminished consumption of utilities such as steam and cooling water. Furthermore, the use of commodity chemicals like 2-pentynoate and benzyl chloroformate ensures that the supply chain is not dependent on exotic or single-source reagents, mitigating the risk of raw material shortages that can disrupt production schedules.
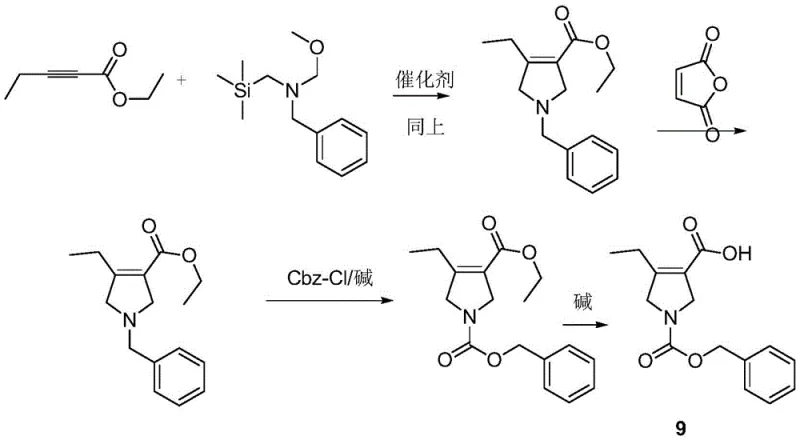
The economic benefits are further amplified by the potential for telescoped operations, as illustrated in the one-pot synthesis variants described in the patent. In these scenarios, intermediates like Compound 3 are generated and consumed in situ without isolation, saving substantial amounts of solvent and eliminating the yield losses associated with drying and handling solid intermediates. This approach leads to substantial cost savings in [cost reduction in pharmaceutical intermediates manufacturing] by maximizing the throughput of existing reactor volumes. Additionally, the environmental footprint of the process is smaller due to reduced solvent waste and higher atom economy, aligning with modern green chemistry principles and easing the burden on waste treatment facilities. For a [reliable pharmaceutical intermediates supplier], these efficiencies translate into more competitive pricing and the ability to offer flexible volume commitments to global partners.
- Cost Reduction in Manufacturing: The elimination of chiral resolution columns and the high yield of the catalytic hydrogenation step remove the most expensive and wasteful parts of the traditional process. By avoiding the 50% yield loss inherent in resolution, the effective cost per kilogram of the active chiral intermediate is nearly halved, excluding catalyst costs which are recoverable. The use of simple workup procedures like extraction and crystallization instead of column chromatography further drives down processing costs, making the final product economically viable for high-volume generic drug production.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures a robust supply chain that is resistant to market fluctuations. Unlike processes requiring custom-synthesized chiral auxiliaries or unstable triflate intermediates, this route uses bulk chemicals that can be sourced from multiple vendors globally. This diversification of the supply base reduces the risk of single-point failures and ensures consistent availability of the intermediate, which is critical for maintaining the continuity of API production for life-saving medications like Upatinib.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize standard stainless steel equipment, facilitating easy scale-up from 100 kgs to 100 MT annual commercial production without the need for specialized reactors. The reduced solvent usage and absence of heavy metal waste (beyond the recoverable Ruthenium catalyst) simplify environmental compliance and waste disposal. This scalability ensures that the manufacturing process can grow in tandem with market demand for JAK inhibitors, providing a future-proof solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or licensing. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the enantiomeric purity achieved in this synthetic route?
A: The process utilizes S-BINAP-Ru(OAc)2 catalytic hydrogenation, achieving an HPLC chiral purity of not less than 99% ee, significantly surpassing traditional resolution methods.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly describes one-pot telescoped reactions and simple post-treatment methods like extraction and crystallization, making it highly suitable for commercial scale-up from kilograms to tons.
Q: How does this method compare to prior art regarding yield?
A: Unlike prior resolution methods yielding only 13% or triflate routes with 20% yield, this novel cyclization and hydrogenation route achieves overall yields exceeding 80% in key steps with simplified purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Upatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-quality chiral intermediates for the growing JAK inhibitor market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify that every batch of Upatinib intermediate meets the >99% ee standard required for downstream API synthesis. Our commitment to quality assurance means that you can rely on us not just as a vendor, but as a strategic partner dedicated to the success of your pharmaceutical projects.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic method for your specific production volumes. Please contact us to request specific COA data, route feasibility assessments, and samples for your evaluation. Together, we can optimize the manufacturing of Upatinib intermediates, driving down costs and accelerating the availability of vital treatments for patients worldwide.
