Scalable Synthesis of Tetranuclear Rectangular Macrocycles for Advanced Catalysis and Nanotechnology
Introduction to Breakthrough Supramolecular Architecture
The landscape of nanotechnology and supramolecular chemistry is being reshaped by the precise engineering of metal-organic frameworks, specifically through the innovations detailed in patent CN101693726A. This intellectual property introduces a sophisticated yet robust methodology for constructing tetranuclear rectangular macrocyclic complexes utilizing half-sandwich organometallic units of ruthenium, iridium, or rhodium. Unlike traditional coordination polymers that often suffer from structural ambiguity, this technology leverages the steric bulk of p-cymene or pentamethylcyclopentadienyl ligands to direct the self-assembly process into well-defined rectangular geometries. For R&D directors and procurement strategists in the fine chemical sector, this represents a pivotal shift towards high-purity, structurally predictable nanomaterials that are essential for next-generation catalytic applications and molecular recognition systems. The ability to tune the internal cavity size and electronic properties through simple ligand substitution offers a versatile platform for developing specialized functional materials.
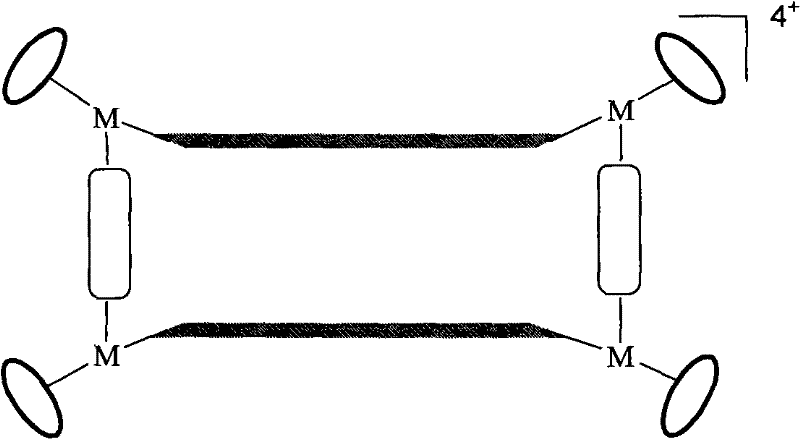
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-nuclearity organometallic cages has been plagued by thermodynamic unpredictability and kinetic traps that lead to polydisperse mixtures rather than discrete species. Conventional methods often rely on flexible ligands or unhindered metal centers that result in uncontrolled polymerization or the formation of insoluble precipitates, severely limiting their utility in homogeneous catalysis or solution-phase sensing. The lack of stereochemical control in traditional assembly processes means that extensive purification steps, such as repeated recrystallization or preparative HPLC, are required to isolate the desired macrocyclic topology, driving up both time and operational costs. Furthermore, the absence of protective groups on the metal centers often leads to decomposition or side reactions with solvent molecules, compromising the stability and shelf-life of the final product. These inefficiencies create significant bottlenecks for supply chain managers who require consistent, high-volume batches of specialized intermediates for downstream applications.
The Novel Approach
The methodology disclosed in CN101693726A overcomes these historical barriers by employing a "half-sandwich" strategy that inherently restricts the coordination geometry of the metal centers. By utilizing oxamide-bridged dinuclear precursors where the metal is shielded on one face by an aromatic cap, the synthesis forces the remaining coordination sites to align in a specific manner that favors the formation of a rectangular motif upon addition of linear bidentate linkers. This geometric constraint drastically improves the selectivity of the self-assembly process, minimizing the formation of oligomeric byproducts and simplifying the isolation of the target tetranuclear species. The process operates under relatively mild conditions, utilizing standard laboratory solvents like tetrahydrofuran and methanol, which facilitates easier solvent recovery and waste management compared to harsher traditional protocols. This streamlined approach not only enhances the chemical purity of the output but also aligns perfectly with the goals of cost reduction in organometallic catalyst manufacturing by reducing the number of unit operations required.
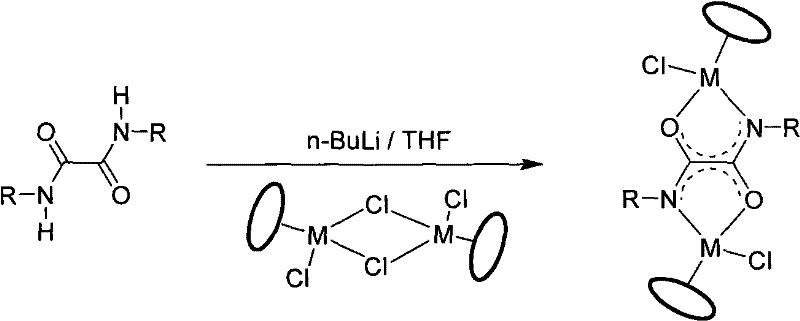
Mechanistic Insights into Oxamide-Bridged Self-Assembly
The core of this technological advancement lies in the precise orchestration of ligand deprotonation and metal coordination kinetics. The process initiates with the quantitative deprotonation of the oxamide ligand using n-butyllithium at cryogenic temperatures (-78°C), generating a highly reactive dianionic species that serves as a rigid bridging unit. This intermediate is then reacted with stable metal dimers, such as [(p-cymene)RuCl2]2, to form the foundational dinuclear building blocks. The rigidity of the oxamide bridge is critical, as it pre-organizes the two metal centers at a fixed distance and angle, setting the stage for the subsequent expansion into a larger architecture. Without this pre-organization, the entropy penalty for forming a large macrocycle would be too high, favoring smaller rings or chains instead. The use of the half-sandwich motif ensures that the chloride ligands are positioned cis to the bridging ligand, making them accessible for the subsequent abstraction step which is the driving force for the final assembly.
The final transformation into the tetranuclear rectangle is driven by halide abstraction using silver triflate (AgOTf), which removes the chloride ligands to generate coordinatively unsaturated cationic metal centers. These electrophilic sites exhibit a strong affinity for the nitrogen donors of the added bidentate pyridine ligands, such as 4,4'-bipyridine. The self-assembly is enthalpically driven by the formation of strong metal-nitrogen coordinate bonds and entropically favored by the precipitation of silver chloride. The result is a highly symmetric cationic complex balanced by triflate anions, exhibiting remarkable stability and solubility characteristics. This mechanistic pathway allows for the modular exchange of the metal center (Ru, Ir, Rh) and the bridging ligands, providing a tunable system for optimizing electronic properties for specific catalytic or electronic applications without redesigning the entire synthetic route.
How to Synthesize Tetranuclear Rectangular Macrocycles Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value complexes with consistency. The procedure begins with the preparation of the dinuclear precursors under strict inert atmosphere conditions to prevent moisture sensitivity issues associated with organolithium reagents. Following the isolation of the intermediate, the self-assembly step is conducted in anhydrous methanol, where the stoichiometry between the metal precursor, the silver salt, and the linker ligand is critical for maximizing yield. Detailed standard operating procedures for temperature control, stirring rates, and filtration techniques are essential to replicate the high selectivity reported in the examples. For a comprehensive breakdown of the specific molar ratios, reaction times, and workup procedures required to achieve optimal results, please refer to the standardized synthesis guide below.
- Deprotonate oxamide ligands with n-BuLi at -78°C and react with metal dimers to form dinuclear intermediates.
- Perform halide abstraction on the dinuclear compound using AgOTf in anhydrous methanol under nitrogen protection.
- Add nitrogen-containing bidentate ligands to the filtrate to induce self-assembly into the final tetranuclear rectangular macrocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this self-assembly technology offers profound advantages for organizations seeking to secure a reliable supply of advanced organometallic materials. The reliance on commercially available starting materials, such as simple oxamides and commodity metal dimers, mitigates the risk of raw material shortages that often plague the supply chains of exotic fine chemicals. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, leading to more consistent batch-to-batch reproducibility which is a key metric for supply chain reliability. Furthermore, the high selectivity of the reaction reduces the burden on downstream purification teams, allowing for faster throughput and lower consumption of chromatography media and solvents. This efficiency translates directly into a more resilient supply chain capable of meeting the demanding timelines of research and development projects in the pharmaceutical and electronic materials sectors.
- Cost Reduction in Manufacturing: The elimination of complex templating agents and the use of straightforward precipitation methods for product isolation significantly lowers the operational expenditure associated with production. By avoiding the need for expensive transition metal catalysts in the assembly step itself—relying instead on stoichiometric self-assembly—the overall material cost is optimized. The high yields reported in the patent examples indicate that raw material utilization is efficient, minimizing waste generation and the associated costs of disposal. Additionally, the simplicity of the workup procedure, which primarily involves filtration and solvent evaporation, reduces the energy consumption and labor hours required per kilogram of product, contributing to substantial cost savings in large-scale manufacturing scenarios.
- Enhanced Supply Chain Reliability: The synthetic route utilizes reagents that are widely sourced from global chemical suppliers, reducing dependency on single-source vendors for critical intermediates. The stability of the dinuclear precursors allows them to be stockpiled if necessary, providing a buffer against potential disruptions in the supply of the final linking ligands. This modularity ensures that production can continue even if one component faces temporary availability issues, as the precursors can be synthesized in advance. For procurement managers, this means a lower risk of project delays due to material shortages and a greater ability to negotiate favorable terms with multiple suppliers for the basic building blocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like methanol and dichloromethane which are standard in industrial chemical plants and have established recovery and recycling infrastructure. The generation of silver chloride as a byproduct is advantageous as it is a solid that can be easily filtered off and potentially recycled for silver recovery, aligning with green chemistry principles. The absence of hazardous gaseous byproducts or extreme pressure requirements simplifies the engineering controls needed for scale-up, making it easier to transition from gram-scale laboratory synthesis to multi-kilogram commercial production while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this supramolecular synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding the nuances of the reaction mechanism and the physical properties of the resulting macrocycles is crucial for integrating these materials into existing product pipelines. We encourage technical teams to review these answers to assess the feasibility of adopting this methodology for their specific application requirements.
Q: What is the key advantage of the half-sandwich structure in this synthesis?
A: The p-cymene or pentamethylcyclopentadienyl groups shield half of the metal center, enforcing directional bonding that favors the formation of discrete rectangular macrocycles over random polymers.
Q: What are the typical yields for these tetranuclear complexes?
A: According to patent data, the self-assembly process demonstrates high selectivity and yields ranging significantly, with some examples achieving up to 89% isolated yield for the final macrocyclic products.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states that the synthesis uses simple steps and common solvents like THF and methanol, making the production easy to realize industrialization and scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetranuclear Rectangular Macrocycles Supplier
As the demand for precision-engineered nanomaterials grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities backed by rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless and efficient. We understand the critical importance of stringent purity specifications in the field of organometallic catalysis and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and purity of every batch. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that empower your R&D initiatives and accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this patented technology can be adapted to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this self-assembly route for your supply chain. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality tetranuclear macrocycles that meet the exacting standards of the global fine chemical industry. Contact us today to explore a partnership that combines innovation with reliability.
