Scalable Synthesis of Valiolamine Intermediates via Novel Radical Dehalogenation for Commercial API Production
Scalable Synthesis of Valiolamine Intermediates via Novel Radical Dehalogenation for Commercial API Production
The pharmaceutical industry's relentless pursuit of effective alpha-glucosidase inhibitors for treating diabetes and obesity has placed significant emphasis on the efficient production of key intermediates like valiolamine. Patent CN101155820A introduces a groundbreaking preparation method for 6,7,8-trihydroxy-1-hydroxymethyl-3-oxo-2-oxa-4-azabicyclo[3.3.1]nonane, a critical precursor in this therapeutic class. This technology addresses long-standing safety and scalability issues associated with traditional dehalogenation processes, offering a robust pathway for high-purity intermediate manufacturing. By leveraging a phosphite-based radical reduction mechanism, this innovation eliminates the need for hazardous reagents and complex purification steps, positioning it as a superior choice for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
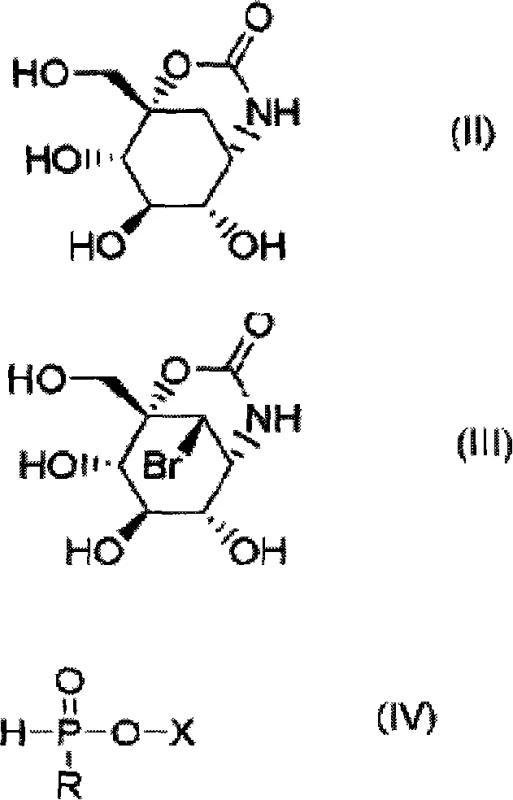
Historically, the dehalogenation of bromo-sugar derivatives like Compound (III) has been fraught with significant operational hazards and technical bottlenecks that hinder large-scale industrial application. Conventional protocols often rely on metal hydride complexes such as sodium borohydride or lithium aluminum hydride, which notoriously generate explosive hydrogen gas during the reaction, necessitating rigorous safety controls and specialized venting systems. Alternatively, catalytic hydrogenation using palladium on carbon requires high-pressure hydrogenation reactors, representing a substantial capital expenditure and introducing risks associated with handling compressed hydrogen gas. Furthermore, methods utilizing organic tin hydrides, while chemically effective, introduce severe toxicity concerns due to the difficulty in removing trace tin residues from the final product, rendering them unsuitable for pharmaceutical grade materials without extensive and costly purification efforts.
The Novel Approach
The methodology disclosed in CN101155820A fundamentally shifts the paradigm by employing phosphite derivatives, specifically sodium hypophosphite, as a free radical reducing agent. This approach circumvents the generation of explosive gases and eliminates the requirement for high-pressure equipment, allowing the reaction to proceed safely in standard glass-lined or stainless steel reactors under atmospheric pressure. The process utilizes readily available and inexpensive reagents, significantly lowering the raw material costs compared to precious metal catalysts or complex hydride sources. Moreover, the workup procedure is drastically simplified; the phosphite byproducts are highly water-soluble and can be easily separated from the organic product through simple filtration and solvent exchange, avoiding the need for complex column chromatography which is often a bottleneck in ton-scale manufacturing.
Mechanistic Insights into Phosphite-Mediated Radical Dehalogenation
The core of this technological advancement lies in the efficient radical chain mechanism facilitated by the interaction between the bromo-intermediate and the phosphite reducing agent in the presence of a radical initiator like AIBN. Upon thermal decomposition, the initiator generates free radicals that abstract a hydrogen atom from the phosphite species, creating a phosphorus-centered radical capable of reducing the carbon-bromine bond in Compound (III). This homolytic cleavage results in the formation of the desired C-H bond in Compound (II) while oxidizing the phosphite to a phosphate species. The elegance of this mechanism is its chemoselectivity; it effectively targets the halogen atom without affecting other sensitive functional groups present on the carbohydrate scaffold, such as the hydroxyl groups or the carbamate protecting group, thereby maintaining the stereochemical integrity essential for biological activity.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal-catalyzed routes. The absence of heavy metals like palladium or tin means there is no risk of metal leaching into the product stream, a critical parameter for regulatory compliance in API synthesis. The primary byproducts are inorganic phosphates which are easily removed during the aqueous workup, leading to a crude product of exceptionally high purity. This reduces the burden on downstream purification processes and minimizes the loss of yield often associated with aggressive purification techniques. The use of mixed solvent systems, particularly water and lower alcohols like methanol or ethanol, further enhances the solubility profile of the reagents while facilitating the crystallization of the final product, ensuring consistent quality across batches.
How to Synthesize 6,7,8-Trihydroxy-1-hydroxymethyl-3-oxo-2-oxa-4-azabicyclo[3.3.1]nonane Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for converting the bromo-precursor into the target dehalogenated intermediate with high efficiency. The process begins by suspending the starting material in a mixture of water and an alcohol, creating a homogeneous reaction environment that supports the radical propagation. The addition of sodium hypophosphite and a catalytic amount of AIBN initiates the reduction upon heating, typically maintained between 80°C and 85°C to ensure optimal reaction kinetics without degrading the sensitive sugar moiety.
- Suspend the bromo-intermediate (Compound III) in a mixed solvent system of water and methanol or ethanol.
- Add sodium hypophosphite as the radical reducing agent and AIBN as the initiator to the reaction mixture under inert atmosphere.
- Heat the mixture to 80-85°C for 3-5 hours, then filter, concentrate, and crystallize the product using methanol to obtain high-purity Compound II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phosphite-based dehalogenation route translates into tangible strategic benefits regarding cost stability and operational continuity. The shift away from precious metal catalysts and hazardous high-pressure operations significantly reduces the total cost of ownership for the manufacturing process. By utilizing commodity chemicals like sodium hypophosphite and standard solvents, the supply chain becomes less vulnerable to the price volatility often seen with specialized catalytic reagents. Furthermore, the simplified purification workflow reduces the consumption of silica gel and organic solvents typically required for column chromatography, leading to substantial cost savings in waste disposal and raw material procurement.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium on carbon removes a major cost driver from the bill of materials, while also negating the need for costly metal scavenging steps post-reaction. The use of sodium hypophosphite, a bulk chemical with stable pricing, ensures predictable production costs and protects margins against market fluctuations. Additionally, the ability to isolate the product through simple crystallization rather than complex chromatography drastically reduces labor hours and solvent usage, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like sodium hypophosphite and AIBN mitigates the risk of supply disruptions that can occur with specialized or imported catalysts. The process does not require custom-built high-pressure reactors, meaning production can be scheduled flexibly across multiple standard facilities without bottlenecks. This flexibility enhances the overall resilience of the supply chain, ensuring consistent delivery of high-purity pharmaceutical intermediates even during periods of high market demand or logistical constraints.
- Scalability and Environmental Compliance: The inherent safety of the process, characterized by the absence of explosive hydrogen gas generation, simplifies regulatory compliance and reduces insurance premiums associated with hazardous operations. The aqueous nature of the reaction and the non-toxic byproducts align well with green chemistry principles, minimizing the environmental footprint of the manufacturing process. This makes the technology highly scalable from pilot plant to commercial production, allowing for seamless capacity expansion to meet growing global demand for anti-diabetic therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehalogenation technology in industrial settings. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, purity, and scalability.
Q: Why is the phosphite-based dehalogenation method safer than traditional catalytic hydrogenation?
A: Traditional methods using metal hydrides or palladium catalysts often generate explosive hydrogen gas or require high-pressure hydrogenation reactors. The phosphite method operates at atmospheric pressure without generating explosive gases, significantly enhancing operational safety.
Q: How does this process improve the impurity profile for pharmaceutical intermediates?
A: Unlike organotin hydride methods which leave toxic tin residues difficult to remove, the phosphite byproducts are water-soluble and easily removed during the aqueous workup, ensuring a cleaner final product suitable for API synthesis.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common solvents like water and methanol and standard heating equipment, eliminating the need for specialized high-pressure reactors or complex chromatography columns, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7,8-Trihydroxy-1-hydroxymethyl-3-oxo-2-oxa-4-azabicyclo[3.3.1]nonane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN101155820A can be translated into reliable supply solutions. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of valiolamine intermediate meets the exacting standards required for downstream API synthesis, providing our partners with peace of mind and regulatory confidence.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your development timelines and enhance your competitive advantage in the marketplace.
