Advanced Synthesis of Binuclear Ir(III) Supramolecular Cages for Industrial Catalysis
The landscape of supramolecular chemistry is undergoing a significant transformation, driven by the urgent need for stable, functional molecular architectures that can withstand rigorous industrial conditions. Patent CN111154115B introduces a groundbreaking methodology for the preparation of binuclear Ir(III) metal-organic supramolecular cage compounds, addressing critical stability issues that have long plagued the field of coordination polymers and metal-organic frameworks. This innovation leverages a sophisticated dual-strategy approach combining dynamic covalent chemistry with post-assembly reduction locking, resulting in cage structures that maintain their integrity even in challenging solvent environments. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift towards more reliable catalyst systems that offer consistent performance without the risk of structural degradation during storage or reaction. The technology not only enhances the photophysical properties of the iridium centers but also ensures that the supramolecular topology remains intact, thereby guaranteeing reproducible catalytic outcomes in complex organic transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of metal-organic supramolecular cages has relied heavily on the self-assembly of metal ions and organic ligands through coordination bonds. While elegant in theory, this conventional approach suffers from inherent thermodynamic instability due to the reversible nature of coordination interactions. In practical applications, these assemblies are prone to dissociation when exposed to competitive solvents such as acetonitrile, DMSO, or DMF, leading to a loss of the defined cavity structure and a consequent collapse of catalytic activity. Furthermore, the synthesis of specific building units with precise coordination geometries is often fraught with low yields and difficult purification protocols, creating significant bottlenecks in the supply chain. The rigidity of traditional ligands also limits the structural diversity achievable, restricting the ability to tune the electronic environment of the metal center for specific catalytic tasks. These factors collectively result in high production costs and inconsistent batch-to-batch quality, making conventional coordination cages less viable for large-scale industrial adoption where reliability is paramount.
The Novel Approach
The methodology outlined in the patent data circumvents these stability issues by employing a dynamic covalent assembly strategy followed by a chemical reduction step. Instead of relying solely on weak coordination forces, the process utilizes aliphatic diamines to bridge pre-assembled Ir(III) complexes through the formation of Schiff base linkages. This initial assembly is dynamic, allowing for error correction during the formation of the cage structure. Crucially, the subsequent addition of sodium borohydride reduces the labile imine (C=N) double bonds into robust amine (C-N) single bonds. This reduction effectively locks the supramolecular architecture into a permanent covalent state, rendering the cage resistant to dissociation and significantly enhancing its chemical stability. This novel route allows for the use of simpler, more accessible ligand precursors while achieving a level of structural permanence that was previously unattainable with pure coordination assemblies, thereby opening new avenues for the commercial deployment of these advanced materials.
Mechanistic Insights into Binuclear Ir(III) Cage Formation
The core of this technology lies in the precise engineering of the iridium coordination sphere and the subsequent covalent linking mechanism. The process begins with the synthesis of pre-assembled metal-based ligands, specifically Ir(III) complexes functionalized with aldehyde groups. These complexes, denoted as L1 and L2 in the patent documentation, feature cyclometalated ligands that provide strong sigma-donation to the iridium center, ensuring high thermal and photochemical stability. The aldehyde functionalities are strategically positioned to react with aliphatic diamines, such as trans-1,2-cyclohexanediamine or 1,3-propanediamine. Under acidic catalysis, these components undergo condensation to form the intermediate cage structure via imine bonds. The steric bulk of the Ir(III) centers is carefully managed to prevent oligomerization, favoring the formation of discrete binuclear cages. This controlled assembly is critical for maintaining the specific spatial arrangement required for the material's fluorescent and catalytic properties.
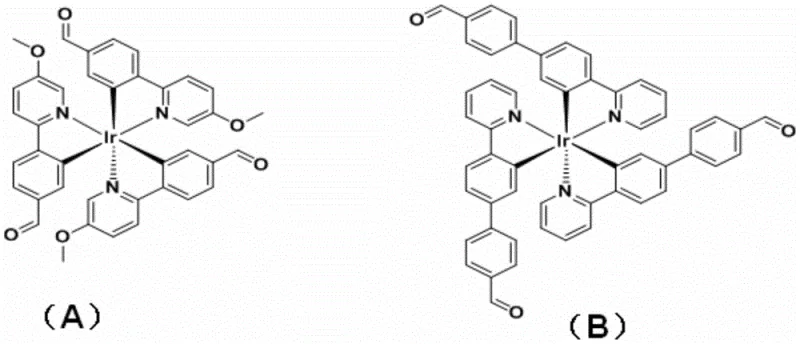
Following the assembly, the reduction mechanism plays a vital role in defining the final product's utility. The addition of sodium borohydride in a methanol solution at low temperatures selectively targets the C=N bonds of the Schiff base linkers without affecting the robust Ir-C or Ir-N coordination bonds. This chemoselective reduction transforms the dynamic imine bridges into static secondary amine linkers. From a mechanistic standpoint, this eliminates the reversibility that characterizes traditional supramolecular systems. The resulting binuclear cage possesses a rigid, well-defined cavity that can host guest molecules or facilitate specific catalytic cycles, such as the activation of molecular oxygen. The stability of the reduced cage ensures that the iridium centers remain in close proximity, which is essential for the cooperative effects observed in the catalytic oxidation of beta-keto esters. This mechanistic robustness translates directly to operational reliability in industrial reactors, where solvent compatibility and thermal endurance are non-negotiable requirements.
How to Synthesize Binuclear Ir(III) Complexes Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing these high-value intermediates, balancing yield with operational simplicity. The process is divided into distinct stages: the preparation of the iridium precursor, the functionalization of the ligand, the cage assembly, and the final reduction. Each step utilizes standard laboratory equipment and common organic solvents, minimizing the need for specialized high-pressure or cryogenic infrastructure. The initial formation of the chloro-bridged iridium dimer is achieved through reflux in glycol-water mixtures, a technique well-suited for scale-up. Subsequent ligand exchange and aldehyde functionalization are performed under inert atmospheres to prevent oxidation of sensitive intermediates. The final assembly and reduction steps are conducted in polar protic solvents, facilitating easy workup and purification via standard column chromatography or recrystallization techniques. For detailed standardized synthesis steps, please refer to the guide below.
- Preparation of Pre-assembled Ir(III) Ligands: React iridium trichloride with functionalized cyclometalating ligands (such as 4-(5-methoxypyridin-2-yl)benzaldehyde derivatives) under reflux in ethylene glycol/water or ethanol/water mixtures to form chloro-bridged dimers, followed by conversion to monomeric precursors using silver salts.
- Dynamic Covalent Assembly: Mix the pre-assembled Ir(III) aldehyde-functionalized ligands with aliphatic diamines (e.g., trans-1,2-cyclohexanediamine) in a mixed solvent system with acid catalysis to form the Schiff base-linked supramolecular cage structure.
- Reduction and Stabilization: Treat the intermediate cage compound with sodium borohydride in methanol at low temperature to reduce the imine (C=N) bonds to amine (C-N) bonds, locking the supramolecular structure into a stable, non-dissociable cage.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers substantial strategic benefits beyond mere technical performance. The elimination of unstable coordination intermediates drastically simplifies the logistics of storage and transportation, as the final reduced cage products exhibit shelf-stable characteristics comparable to traditional small molecule organics. This stability reduces the risk of material degradation during transit, ensuring that the specifications received match the specifications ordered. Furthermore, the reliance on commodity chemicals like iridium trichloride and simple diamines mitigates the risk of supply chain disruptions associated with exotic, single-source reagents. The process design inherently supports continuous manufacturing improvements, allowing for incremental cost optimizations as production volumes increase. By adopting this technology, organizations can secure a more resilient supply of high-performance catalysts while simultaneously reducing the total cost of ownership associated with catalyst handling and replacement.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers production costs by eliminating the need for expensive stabilizing agents or complex purification protocols required for unstable coordination cages. The use of sodium borohydride as a reducing agent is economically favorable compared to other hydrogenation methods, and the high yields reported in the patent examples indicate efficient atom economy. Additionally, the robustness of the final product reduces waste generation caused by material decomposition, leading to lower disposal costs and improved overall process efficiency. The ability to use standard solvents like ethanol and acetonitrile further aligns the process with existing solvent recovery infrastructure, maximizing resource utilization.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various aliphatic diamines and substituted benzaldehydes, are widely available from multiple global suppliers, reducing dependency on single vendors. This diversification of the supply base enhances negotiation leverage and ensures continuity of supply even during market fluctuations. The simplified synthetic steps also reduce the lead time for batch production, allowing manufacturers to respond more agilely to changes in demand. The stability of the intermediates and final products means that safety stock can be maintained without the fear of rapid degradation, providing a buffer against unexpected supply shocks.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, removing the safety hazards and capital expenditures associated with high-pressure hydrogenation or extreme thermal conditions. This mild operating window facilitates easier scale-up from pilot plants to commercial production facilities without requiring extensive re-engineering of reactor systems. Moreover, the reduction in solvent complexity and the potential for solvent recycling contribute to a smaller environmental footprint, aiding compliance with increasingly stringent global environmental regulations. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying waste treatment and lowering the environmental burden of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this binuclear Ir(III) cage technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating these catalysts into existing process lines. The answers highlight the balance between advanced supramolecular design and practical industrial applicability, ensuring that stakeholders have a clear understanding of the technology's capabilities and limitations.
Q: What distinguishes this binuclear Ir(III) cage from traditional coordination assemblies?
A: Unlike traditional supramolecular cages held together solely by reversible coordination bonds which often dissociate in solution, this method utilizes a dynamic covalent assembly strategy followed by chemical reduction. The Schiff base linkages formed between the Ir-ligands and diamines are reduced to stable secondary amines, creating a robust covalent framework that resists dissociation in polar solvents like DMSO or DMF.
Q: What is the primary catalytic application of these Ir(III) complexes?
A: These binuclear Ir(III) complexes function as efficient photocatalysts for the alpha-hydroxylation of beta-keto esters. Upon visible light irradiation, the complex activates triplet oxygen to generate singlet oxygen, facilitating the oxidation of substrates like methyl 1-oxo-2,3-dihydro-1-indene-2-carboxylate with high yields reaching up to 83%.
Q: Are the starting materials for this synthesis commercially scalable?
A: Yes, the synthesis relies on readily available commodity chemicals such as iridium trichloride, various aliphatic diamines (ethylenediamine, 1,3-propanediamine), and substituted benzaldehydes. The process avoids exotic reagents and utilizes standard organic solvents like ethanol, acetonitrile, and dichloromethane, making it highly suitable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binuclear Ir(III) Complexes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced supramolecular catalysts in driving the next generation of chemical synthesis. As a premier CDMO partner, we possess the technical expertise and infrastructure necessary to translate complex laboratory protocols like those in CN111154115B into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot to plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of binuclear Ir(III) complexes meets the exacting standards required for high-value applications in pharmaceuticals and fine chemicals. Our commitment to quality assurance ensures that the structural integrity and catalytic activity of these sophisticated materials are preserved throughout the manufacturing lifecycle.
We invite you to collaborate with us to optimize your supply chain and unlock the full potential of this innovative catalytic technology. By leveraging our process development capabilities, you can achieve significant operational efficiencies and secure a competitive advantage in the market. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your project goals. Let us help you navigate the complexities of supramolecular chemistry and deliver reliable, high-quality solutions for your business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
