Advanced Synthetic Route for High-Purity R-(+)-Tetrahydrocarbazole Intermediates via Early-Stage Resolution
Advanced Synthetic Route for High-Purity R-(+)-Tetrahydrocarbazole Intermediates via Early-Stage Resolution
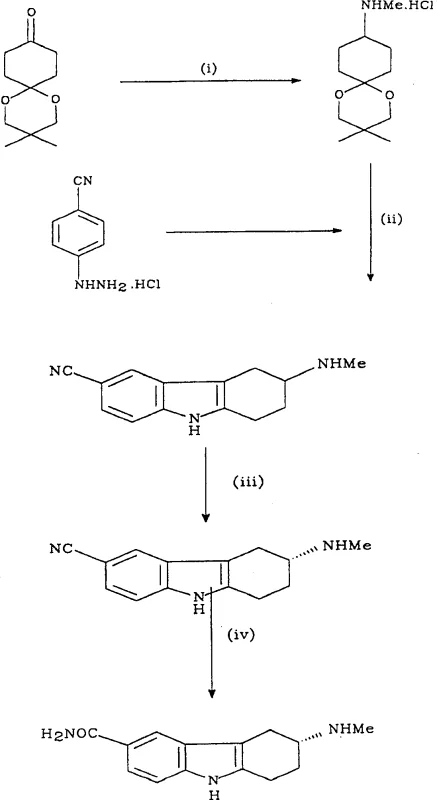
The pharmaceutical industry constantly seeks robust methodologies for producing chiral intermediates, particularly for central nervous system agents like 5HT1 receptor agonists used in migraine treatment. Patent CN1305460A discloses a groundbreaking process for the preparation of R-(+)-6-carboxamido-3-N-methylamino-1,2,3,4-tetrahydrocarbazole, a critical precursor in the synthesis of potent anti-migraine therapeutics. This innovation addresses significant bottlenecks found in prior art, specifically the limitations associated with late-stage resolution and the use of expensive or ineffective resolving agents. By shifting the chiral separation to an earlier indolenitrile intermediate stage, the disclosed method achieves superior solubility profiles and higher overall yields. This technical advancement represents a paradigm shift for manufacturers aiming to optimize their supply chains for complex heterocyclic compounds. The strategic implementation of L-pyroglutamic acid as a resolving agent not only enhances stereochemical control but also leverages the economic benefits of naturally occurring chiral pool resources. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for evaluating potential suppliers capable of delivering high-purity pharmaceutical intermediates at a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO-A-94/14772, typically relied on resolving the final product or late-stage intermediates using D-pyroglutamic acid. These conventional approaches suffered from severe drawbacks, primarily stemming from the poor solubility of the resulting chiral salts, which drastically limited recovery rates and overall process efficiency. Furthermore, attempting resolution at the final amide stage often necessitated extensive protection and deprotection sequences, adding unnecessary complexity and cost to the manufacturing workflow. The reliance on chromatographic purification for enantiomeric enrichment, as seen in some earlier protocols, is fundamentally incompatible with large-scale industrial production due to prohibitive costs and throughput limitations. Additionally, the use of D-pyroglutamic acid was found to yield the incorrect enantiomer in many contexts, forcing manufacturers to invert stereochemistry through additional synthetic steps or accept low yields of the desired isomer. These inefficiencies created significant barriers to entry for generic manufacturers and increased the cost of goods for originator companies seeking reliable contract development and manufacturing organizations.
The Novel Approach
The novel methodology presented in CN1305460A circumvents these historical challenges by introducing a resolution step at the indolenitrile intermediate stage, specifically utilizing L-pyroglutamic acid. This strategic pivot allows for the isolation of the desired R-(+) enantiomer with exceptional optical purity before the final functional group transformations are completed. The process capitalizes on the favorable solubility characteristics of the L-pyroglutamate salt of the indolenitrile, enabling efficient crystallization and filtration without the need for complex chromatographic separations. By resolving the molecule earlier in the synthetic sequence, the process ensures that all subsequent chemical transformations proceed with the correct stereochemistry already established, thereby minimizing the formation of diastereomeric impurities. This approach not only simplifies the downstream processing but also significantly enhances the theoretical yield of the final active pharmaceutical ingredient. The transition from D- to L-pyroglutamic acid also offers a distinct economic advantage, as the L-isomer is a natural product and is generally more cost-effective and readily available in bulk quantities compared to its D-counterpart.
Mechanistic Insights into L-Pyroglutamic Acid Mediated Chiral Resolution
The core of this innovative process lies in the diastereomeric salt formation between the racemic indolenitrile intermediate and L-pyroglutamic acid. Mechanistically, the interaction between the basic nitrogen of the tetrahydrocarbazole ring and the carboxylic acid groups of the pyroglutamic acid creates a stable salt complex. The stereochemical environment provided by the L-pyroglutamic acid favors the crystallization of the R-(+) enantiomer of the indolenitrile over the S-(-) enantiomer due to differences in lattice energy and solubility in alcoholic solvents. The patent data highlights the critical role of solvent composition, specifically the use of methanol or ethanol with controlled water content, in modulating the solubility equilibrium to drive the selective precipitation of the desired diastereomer. The addition of acetic acid during the crystallization phase further refines this selectivity by adjusting the pH and ionic strength of the solution, promoting the nucleation of the target salt while keeping the unwanted enantiomer in the mother liquor. This precise control over crystallization kinetics is vital for achieving optical purities exceeding 98% ee, which is a stringent requirement for downstream pharmaceutical applications. The robustness of this resolution step ensures that the chiral integrity is maintained throughout the subsequent hydrolysis and salt formation stages.
Following the successful resolution, the process proceeds with the hydrolysis of the nitrile group to the corresponding carboxamide, a transformation that requires careful condition selection to avoid over-hydrolysis to the carboxylic acid. The use of a boron trifluoride/acetic acid complex serves as a mild yet effective Lewis acid catalyst that facilitates the hydration of the nitrile functionality under controlled thermal conditions. This specific reagent system is chosen to minimize side reactions and preserve the sensitive tetrahydrocarbazole core structure. The mechanistic pathway involves the activation of the nitrile nitrogen by the Lewis acid, followed by nucleophilic attack by water, leading to the formation of the primary amide. Impurity control is rigorously managed through this step by monitoring the reaction progress via HPLC to ensure complete conversion without degradation. The final conversion to the succinate salt involves a straightforward acid-base reaction in a mixed solvent system, where temperature control during cooling dictates the crystal habit and purity of the final isolated solid. This comprehensive understanding of the reaction mechanism allows for precise scale-up and consistent quality assurance in a GMP manufacturing environment.
How to Synthesize R-(+)-Tetrahydrocarbazole Efficiently
The synthesis of this high-value intermediate begins with the construction of the tetrahydrocarbazole scaffold via a Fischer indole synthesis, followed by the critical chiral resolution and functional group modification. The process is designed to be telescoped where possible to minimize unit operations and solvent usage, aligning with green chemistry principles. Operators must pay close attention to the stoichiometry of the resolving agent and the cooling profiles during crystallization to maximize yield and optical purity. The detailed standard operating procedures for each reaction stage, including specific temperature ramps and aging times, are critical for reproducibility. For a comprehensive guide on the exact molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Perform Fischer Indole Synthesis by reacting 4-cyanophenylhydrazine with 4-methylaminocyclohexanone ketal under acidic aqueous conditions to form the racemic indolenitrile.
- Resolve the racemic indolenitrile mixture using L-pyroglutamic acid in methanol/ethanol to form the diastereomeric salt of the desired R-(+) enantiomer.
- Hydrolyze the resolved nitrile intermediate using boron trifluoride/acetic acid complex to convert the cyano group to the carboxamido group, followed by succinate salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible benefits that extend beyond mere technical feasibility. The elimination of chromatographic purification steps significantly reduces the consumption of silica gel and organic solvents, leading to a drastic simplification of the waste management profile and a reduction in overall manufacturing costs. The switch to L-pyroglutamic acid, a naturally derived and economically favorable resolving agent, further drives down the raw material costs compared to synthetic or less abundant D-isomers. This cost structure improvement allows for more competitive pricing models when sourcing this critical pharmaceutical intermediate from reliable suppliers. Furthermore, the high yield and purity achieved through this crystallization-based resolution reduce the need for reprocessing or recycling of off-spec material, thereby enhancing the overall throughput of the production facility. These efficiencies translate directly into shorter lead times and greater supply security for downstream API manufacturers who depend on a steady flow of high-quality chiral building blocks.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chiral HPLC columns and reduces solvent consumption by relying on crystallization for purification. By avoiding extensive protection and deprotection sequences required in older methods, the number of synthetic steps is effectively optimized, lowering labor and utility costs. The use of L-pyroglutamic acid provides a cost-effective alternative to other resolving agents, contributing to a leaner cost of goods sold. These cumulative savings allow for substantial cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 4-cyanophenylhydrazine and protected cyclohexanedione ensures a robust supply chain with multiple sourcing options. The crystallization-based purification method is inherently more scalable and less prone to equipment bottlenecks than chromatographic techniques, ensuring consistent batch-to-batch availability. This reliability is crucial for maintaining continuous API production schedules and mitigating the risk of stockouts. Suppliers utilizing this technology can offer greater flexibility in order volumes and delivery timelines, strengthening the partnership between chemical manufacturers and pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up, utilizing standard reactor equipment and avoiding hazardous reagents that require specialized handling. The reduction in solvent waste and the avoidance of heavy metal catalysts align with increasingly stringent environmental regulations and corporate sustainability goals. The high atom economy of the Fischer indole synthesis combined with the efficient resolution step minimizes the environmental footprint of the manufacturing process. This compliance facilitates easier regulatory approval and supports the long-term viability of the supply chain in a regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of R-(+)-6-carboxamido-3-N-methylamino-1,2,3,4-tetrahydrocarbazole. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process validation. The responses cover aspects of stereochemistry, process safety, and quality control metrics that are critical for pharmaceutical grade materials.
Q: Why is L-pyroglutamic acid preferred over D-pyroglutamic acid in this synthesis?
A: The patent data indicates that using D-pyroglutamic acid yields the 'wrong' enantiomer with poor solubility, whereas L-pyroglutamic acid selectively crystallizes the desired R-(+) enantiomer with high optical purity and superior solubility characteristics.
Q: What are the critical control points for ensuring high optical purity?
A: Critical control points include the precise temperature management during crystallization (cooling to 0-5°C), the addition of acetic acid to facilitate selective crystallization, and the recrystallization of the L-pyroglutamate salt from aqueous ethanol to enhance ee values above 98%.
Q: How does this process improve scalability compared to previous methods?
A: By performing the resolution at the indolenitrile stage rather than the final product stage, the process avoids extensive protection and deprotection steps and eliminates the need for preparative chromatography, making it highly suitable for large-scale industrial batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-(+)-Tetrahydrocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation migraine therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution chemistry described in CN1305460A can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the optical purity and chemical identity of every batch. Our commitment to quality assurance means that we can deliver intermediates that meet the exacting standards required for global regulatory filings, providing our partners with peace of mind and supply chain stability.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your commercial manufacturing needs. Let us be your strategic partner in bringing life-saving medications to market faster and more efficiently.
