Advanced Ortho-Halogenation Technology for High-Purity Pharmaceutical Intermediates Manufacturing
The landscape of aromatic halide synthesis is undergoing a significant transformation driven by the urgent need for more efficient and environmentally sustainable manufacturing processes, particularly for high-value intermediates used in the pharmaceutical and agrochemical sectors. Patent CN113620811A introduces a groundbreaking methodology that leverages a novel proton transfer strategy to achieve highly selective ortho-halogenation of aromatic compounds such as arylamines, carbazoles, and phenols. This technology represents a paradigm shift from conventional electrophilic substitution reactions, which have long been plagued by issues regarding regioselectivity control, harsh reaction conditions, and the generation of substantial hazardous waste. By employing inexpensive metal sulfonate salts as catalysts, this invention enables the direct conversion of readily available starting materials into corresponding ortho-substituted bromination and chlorination products with moderate to excellent yields. The strategic implementation of this catalytic system not only addresses the critical demand for precise molecular architecture in drug discovery but also aligns with the global push towards greener chemistry practices by minimizing the reliance on stoichiometric amounts of toxic reagents and complex purification protocols.
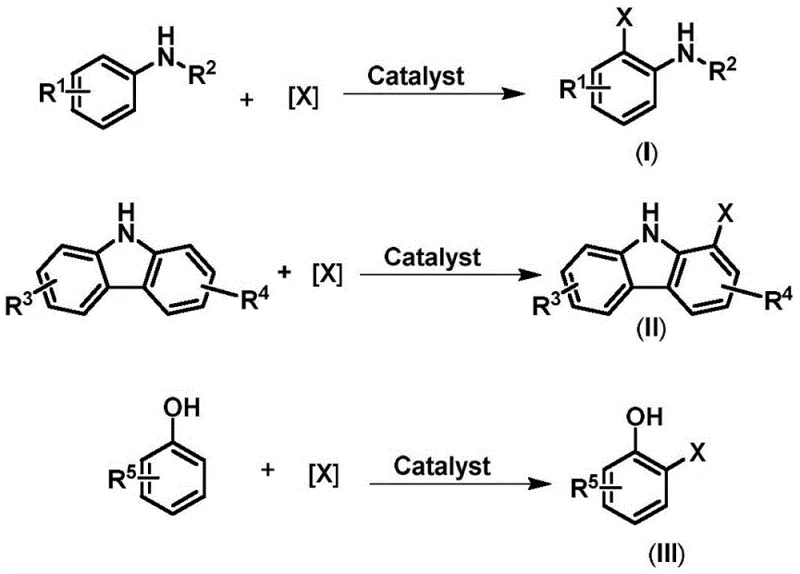
For decades, the industrial production of ortho-halogenated aromatic amines, such as o-chloroaniline, has relied heavily on the reduction of o-nitrochlorobenzene, a process fraught with significant operational and environmental challenges. The traditional iron powder reduction method, while historically prevalent, is increasingly being phased out due to its backward technological profile, high production costs, severe pollution issues, and inconsistent product quality that complicates downstream processing. Although catalytic hydrogenation offers a cleaner alternative with higher efficiency, it introduces the risk of hydrogenolysis dechlorination, leading to the formation of aniline byproducts that compromise reaction efficiency and cause serious equipment corrosion. Furthermore, existing direct electrophilic halogenation methods often suffer from poor selectivity control, requiring expensive catalysts and strict reaction conditions that limit their economic feasibility for large-scale applications. In contrast, the novel approach disclosed in the patent utilizes a unique combination of metal sulfonate catalysts and common halogenating agents like NBS or TCCA to overcome these limitations. This method achieves superior ortho-selectivity through specific intermolecular interactions, effectively bypassing the thermodynamic preference for para-substitution that typically dominates traditional Friedel-Crafts type reactions, thereby providing a robust solution for the synthesis of high-purity intermediates.
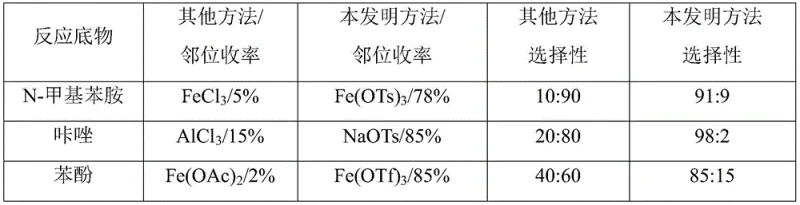
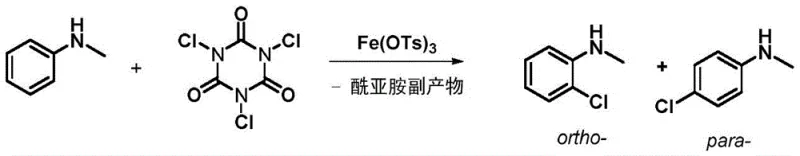
The mechanistic underpinning of this innovative halogenation process lies in the sophisticated interplay between the metal sulfonate catalyst and the aromatic substrate, which fundamentally alters the electronic environment of the reaction center. The catalyst, typically a salt formed by a metal with certain Lewis acidity and a sulfonate anion capable of transferring protons, engages in a specific O-H hydrogen bond interaction with the reaction substrate. This interaction, coupled with the weak coordination between the metal center and the nitrogen atom of the amine or heterocycle, creates a transient supramolecular assembly that directs the incoming electrophilic halogen species exclusively to the ortho-position. This proton migration strategy breaks through conventional textbook knowledge which suggests that Lewis acids generally reduce reaction efficiency in electrophilic halogenation, instead demonstrating that the right choice of counter-ion can enhance both rate and selectivity. The result is a highly controlled reaction pathway that minimizes the formation of para-isomers and poly-halogenated byproducts, ensuring a cleaner impurity profile that is critical for regulatory compliance in pharmaceutical manufacturing. For instance, the chlorination of N-methylaniline using Iron p-toluenesulfonate yields the ortho-product with a remarkable selectivity ratio of 91:9 over the para-isomer, showcasing the precision of this catalytic system.
Mechanistic Insights into Metal Sulfonate-Catalyzed Ortho-Halogenation
The depth of control achieved in this reaction stems from the dual functionality of the metal sulfonate catalyst, which acts not merely as a Lewis acid activator but as a structural director through non-covalent interactions. The sulfonate group facilitates proton transfer events that stabilize the transition state leading to ortho-substitution, while the metal center coordinates weakly with the heteroatom of the substrate to lock the conformation in a way that exposes the ortho-carbon to the electrophile. This mechanism effectively suppresses the inherent electronic bias of the aromatic ring that would otherwise favor para-attack, allowing for the functionalization of electron-deficient or sterically hindered substrates that are typically unreactive under standard conditions. Moreover, the catalyst system exhibits remarkable tolerance towards a wide array of functional groups, including ethers, esters, and halogens, which means that complex molecules can be halogenated late-stage in the synthesis without the need for extensive protecting group manipulations. This level of chemoselectivity is paramount for R&D teams aiming to rapidly iterate on lead compounds, as it reduces the number of synthetic steps and accelerates the timeline for generating diverse libraries of halogenated building blocks for structure-activity relationship studies.
How to Synthesize Ortho-Halogenated Aromatics Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to existing laboratory and pilot plant infrastructure, requiring minimal specialized equipment beyond standard glassware and heating mantles. The general procedure involves charging a reaction vessel with the aromatic substrate, the metal sulfonate catalyst, and the halogenating reagent in a suitable organic solvent such as toluene or dioxane, followed by heating the mixture to a moderate temperature range of 90°C to 110°C. The reaction proceeds under an air atmosphere, eliminating the need for inert gas lines or glovebox conditions, which significantly simplifies the operational complexity and reduces the capital expenditure associated with setting up the process. After the designated reaction time, which can range from a few hours to overnight depending on the substrate reactivity, the mixture is cooled, extracted with an organic solvent, and purified using standard column chromatography or crystallization techniques. Detailed standardized synthesis steps for specific substrates like N-methylaniline or carbazole derivatives are provided in the guide below to ensure reproducibility and optimal yield.
- Charge a reaction vessel with a metal sulfonate catalyst (e.g., Fe(OTs)3), the aromatic substrate (e.g., N-methylaniline), an electrophilic halogenating reagent (e.g., TCCA or NBS), and a suitable organic solvent like toluene.
- Heat the reaction mixture to a temperature between 25°C and 180°C, preferably 90°C to 110°C, and maintain stirring under an air atmosphere for a duration ranging from 30 seconds to 48 hours.
- Upon completion, cool the system to room temperature, extract the product with an organic solvent such as dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal sulfonate-catalyzed halogenation technology offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. The primary driver of value is the drastic simplification of the supply chain for raw materials, as the process utilizes cheap, commercially available metal salts and common halogenating agents instead of exotic or proprietary catalysts that are subject to volatile pricing and long lead times. This shift to commodity-grade reagents ensures a stable and continuous supply of critical intermediates, mitigating the risk of production stoppages due to material shortages. Furthermore, the elimination of expensive transition metals like palladium or platinum from the catalyst formulation removes the necessity for costly and time-consuming heavy metal scavenging steps during downstream processing, which translates into significant reductions in both material costs and waste disposal fees. The high selectivity of the reaction also means that less starting material is wasted on unwanted isomers, improving the overall atom economy and reducing the volume of solvent and energy required for purification, thereby enhancing the sustainability profile of the manufacturing process.
- Cost Reduction in Manufacturing: The utilization of inexpensive metal sulfonate catalysts such as iron or aluminum salts replaces costly noble metal catalysts, leading to a substantial decrease in direct material costs per kilogram of product. Additionally, the high ortho-selectivity minimizes the formation of difficult-to-separate para-isomers, which reduces the burden on purification units and lowers the consumption of solvents and energy associated with chromatographic separation or recrystallization. By avoiding the need for complex protecting group strategies and multi-step sequences often required in traditional routes, the overall process mass intensity is significantly lowered, resulting in a more lean and cost-efficient manufacturing workflow that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable chemical reagents ensures that the supply chain is robust against market fluctuations and geopolitical disruptions that often affect specialized fine chemicals. Since the catalysts and halogenating agents are commodity items with multiple global suppliers, procurement teams can easily qualify alternative vendors to secure competitive pricing and guarantee uninterrupted production schedules. The simplicity of the reaction conditions, which tolerate air and moisture to a greater extent than sensitive organometallic catalysts, also reduces the logistical complexities associated with storage and transportation, further stabilizing the supply chain and reducing the risk of inventory degradation or loss.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation under mild thermal conditions and atmospheric pressure, allowing for seamless translation from gram-scale laboratory experiments to ton-scale commercial production without the need for specialized high-pressure reactors. The reduced generation of hazardous waste, particularly heavy metal residues and toxic byproducts, simplifies compliance with increasingly stringent environmental regulations and lowers the costs associated with wastewater treatment and hazardous waste disposal. This environmental advantage not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing site, making it a more attractive partner for global pharmaceutical companies seeking sustainable supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ortho-halogenation technology, providing clarity on its operational parameters and strategic benefits for potential partners. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring that the information provided is accurate and relevant to real-world manufacturing scenarios. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production pipelines or for procurement officers assessing the long-term value proposition of switching to this new catalytic system.
Q: What is the primary advantage of the metal sulfonate catalyst over traditional Lewis acids?
A: Unlike traditional Lewis acids which often reduce reaction efficiency or require strict anhydrous conditions, the metal sulfonate catalyst utilizes a proton transfer strategy. This allows for excellent ortho-selectivity through O-H hydrogen bond interactions and weak metal-N coordination, significantly improving yield and reducing byproduct formation.
Q: Can this method be applied to sensitive functional groups?
A: Yes, the method demonstrates excellent substrate applicability and compatibility with various chemical functional groups including ether, ester, methoxyl, halogen, methyl, and phenyl groups, making it suitable for synthesizing complex pharmaceutical intermediates without protecting group strategies.
Q: Is the process scalable for industrial production?
A: The process is designed for industrial viability, utilizing cheap and commercially available metal salt catalysts and operating under air atmosphere at moderate temperatures. The simplified workup and high selectivity reduce purification costs, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Halogenated Aromatics Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the fast-evolving landscape of fine chemical and pharmaceutical intermediate manufacturing. Our team of expert process chemists has thoroughly evaluated the metal sulfonate-catalyzed halogenation method described in CN113620811A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the market. We are committed to delivering high-purity ortho-halogenated intermediates that meet stringent purity specifications, leveraging our rigorous QC labs and state-of-the-art analytical capabilities to ensure every batch conforms to the highest industry standards. Our facility is equipped to handle the specific solvent systems and reaction conditions outlined in the patent, ensuring a smooth and efficient technology transfer that minimizes ramp-up time and maximizes yield consistency.
We invite you to engage with our technical procurement team to discuss how this cutting-edge halogenation technology can be tailored to your specific project needs, offering a pathway to significant cost optimization and supply chain security. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your target molecules, along with specific COA data and route feasibility assessments that demonstrate our capability to deliver. Let us partner with you to accelerate your development timelines and secure a reliable source of high-quality aromatic halides for your next generation of therapeutic or agrochemical products.
