Mastering the Total Synthesis of Salvianolic Acid A for Scalable Pharmaceutical Production
Mastering the Total Synthesis of Salvianolic Acid A for Scalable Pharmaceutical Production
The pharmaceutical industry constantly seeks reliable sources for bioactive natural products that are otherwise difficult to source from nature due to low abundance or stability issues. Patent CN102827002A presents a groundbreaking chemical total-synthetic method for Salvianolic Acid A, a potent water-soluble component derived from Salvia miltiorrhiza (Danshen). This patent addresses the critical bottleneck of relying on plant extraction, which often results in unstable mixtures with low purity. By establishing a robust synthetic pathway, this technology enables the production of high-purity pharmaceutical intermediates that meet stringent quality standards required for modern cardiovascular therapeutics. The synthesis strategically dissects the complex molecule into two manageable fragments, utilizing classic organic transformations like the Darzens reaction and Wittig olefination to construct the intricate carbon skeleton with high precision.
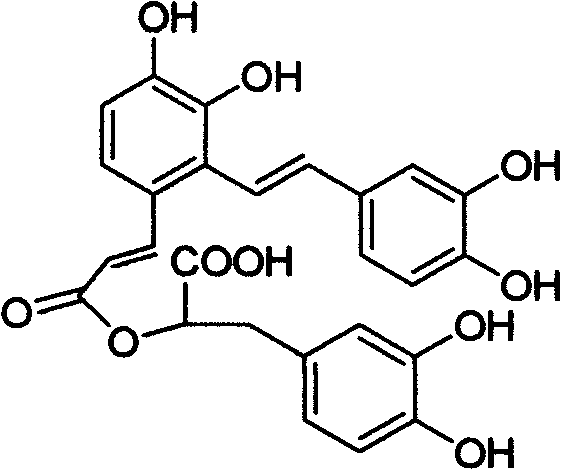
For R&D directors and procurement specialists, the shift from extraction to synthesis represents a paradigm shift in supply chain security. The natural product is known for its pharmacological actions, including anti-thrombosis and platelet aggregation inhibition, but its clinical application has been hindered by the instability of its catechol structure, which is prone to oxidation and color deepening upon storage. The synthetic approach detailed in this patent not only solves the resource scarcity problem but also allows for structural modifications that can enhance stability. As a reliable pharmaceutical intermediate supplier, understanding this synthetic logic is crucial for evaluating the feasibility of long-term commercial partnerships and ensuring a continuous supply of this critical active ingredient for drug development pipelines globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of Salvianolic Acid A has relied heavily on extraction from the roots of Salvia miltiorrhiza. However, this conventional method suffers from severe inherent limitations that impact both cost reduction in API manufacturing and product consistency. The content of Salvianolic Acid A in the plant is extremely low, necessitating the processing of massive quantities of raw biomass to obtain negligible yields of the target compound. Furthermore, the extraction process yields a complex mixture of water-soluble components rather than a pure substance. These components interact with each other, leading to character instability and making purification exceptionally difficult and costly. The presence of multiple catechol groups makes the extracted material highly susceptible to oxidation, resulting in deepening color and degradation of potency over time. This instability creates significant challenges for formulation scientists and quality control teams, as batch-to-batch variability is high, and shelf-life is compromised, ultimately influencing the quality of clinical applications and limiting the further exploitation of this valuable natural drug.
The Novel Approach
In stark contrast to the unpredictability of extraction, the novel approach outlined in patent CN102827002A employs a rational retrosynthetic strategy that splits the target molecule into two distinct fragments for independent construction. This fragment-based assembly allows for rigorous quality control at each stage of the synthesis, ensuring that impurities are managed before the final coupling. The first fragment is constructed via a Darzens reaction followed by catalytic hydrogenation, establishing the chiral hydroxy acid moiety with high fidelity. The second fragment is built through a sequence involving reduction, bromination, and a Wittig reaction to install the necessary styryl double bond geometry. By condensing these two pre-validated fragments, the synthesis achieves a level of purity and structural definition that extraction simply cannot match. This method effectively bypasses the oxidation issues associated with crude plant extracts, providing a stable, well-defined chemical entity that is suitable for rigorous structure-activity relationship studies and eventual commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Fragment Condensation and Selective Deprotection
The core of this synthetic success lies in the meticulous orchestration of reaction conditions to preserve sensitive functional groups while building molecular complexity. The synthesis begins with the Darzens condensation of 3,4-dimethoxy benzaldehyde and methyl chloroacetate under alkaline conditions, typically using sodium methylate or triethylamine at temperatures ranging from -10°C to 50°C. This step generates an epoxy ester, which is subsequently subjected to catalytic hydrogenation using Pd/C in ethyl acetate. This hydrogenation step is critical as it opens the epoxide ring to form the 2-hydroxy-3-(3,4-dimethoxyphenyl) methyl propionate fragment. Simultaneously, the second fragment is prepared from 2,3-dimethoxy benzaldehyde. A key mechanistic feature here is the use of n-butyllithium for debromination and subsequent aldehyde addition, which allows for the precise extension of the carbon chain. The final coupling involves a Knoevenagel reaction to form the cinnamic acid derivative, which is then esterified with the first fragment. The choice of reagents is deliberate; for instance, the use of mild bases like piperidine or triethylamine in the Knoevenagel step prevents unwanted side reactions on the electron-rich aromatic rings.
Perhaps the most sophisticated aspect of this mechanism is the selective deprotection strategy employed in the final stages. The molecule contains both carboxylic acid methyl esters and phenolic methyl ethers, both of which need to be removed to reveal the bioactive polyphenol. Standard basic hydrolysis would be non-selective and could lead to decomposition. The patent innovatively utilizes trimethyltin hydroxide (Me3SnOH) to selectively hydrolyze the carboxylate methyl ester at temperatures between 20°C and 100°C without touching the phenolic protecting groups. Following this, the phenolic methyl groups are removed using a Lewis acid such as boron tribromide (BBr3) or aluminum chloride in non-protonic solvents like methylene dichloride. This orthogonal deprotection strategy is vital for maintaining the integrity of the ester linkage while unveiling the free phenolic hydroxyls. For R&D teams, this mechanistic insight highlights the importance of reagent selection in preserving the delicate balance of functionality in polyphenolic natural products, ensuring that the final product retains its intended biological activity without degradation.
How to Synthesize Salvianolic Acid A Efficiently
The synthesis of Salvianolic Acid A requires a disciplined approach to reaction monitoring and purification to ensure high overall yield and purity. The process involves ten distinct chemical steps, starting from readily available benzaldehyde derivatives. The initial phases focus on building the two key fragments independently, allowing for parallel processing which can significantly reduce lead time for high-purity pharmaceutical intermediates. Critical attention must be paid to the stereochemistry during the Darzens reaction and the geometric integrity of the double bonds formed during the Wittig and Knoevenagel reactions. The patent specifies the use of column chromatography with petroleum ether and ethyl acetate systems for purification at various stages, indicating that while the chemistry is robust, careful separation of diastereomers and byproducts is necessary. Detailed standardized synthetic steps are provided below to guide process chemists in replicating this high-value pathway.
- Synthesize Fragment 1 by reacting 3,4-dimethoxy benzaldehyde with methyl chloroacetate via Darzens reaction, followed by catalytic hydrogenation to form the hydroxy ester.
- Prepare Fragment 2 starting from 2,3-dimethoxy benzaldehyde through reduction, bromination, Wittig reaction, and Knoevenagel condensation to build the styryl backbone.
- Condense the two fragments via esterification, selectively remove the carboxyl methyl ester using trimethyltin hydroxide, and finally deprotect phenolic groups with Lewis acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to a fully synthetic route for Salvianolic Acid A offers profound strategic advantages that go beyond simple availability. The primary benefit is the decoupling of supply from agricultural variables. Unlike plant extraction, which is subject to seasonal fluctuations, weather patterns, and geographical constraints, chemical synthesis can be performed year-round in a controlled factory environment. This ensures a consistent and predictable supply chain, eliminating the risks associated with crop failures or raw material shortages that often plague botanical sourcing. Furthermore, the synthetic route utilizes commodity chemicals such as dimethoxy benzaldehydes and methyl chloroacetate, which are abundant and cost-effective. This shifts the cost structure from one dominated by expensive biomass processing and low-yield extraction to one driven by efficient chemical transformations, potentially leading to substantial cost savings in the long run.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for large-scale biomass handling and the extensive solvent usage associated with extracting trace components from plant roots. By using high-concentration chemical reactions, the process intensity is significantly increased, meaning smaller reactors can produce the same amount of product compared to extraction tanks. Additionally, the selective deprotection strategy using trimethyltin hydroxide avoids the harsh conditions that often degrade product quality, thereby reducing waste and rework costs. The ability to synthesize the molecule from cheap starting materials means that the variable cost of goods sold (COGS) is drastically lower than that of purified natural extracts. This economic efficiency allows for more competitive pricing strategies in the global market for cardiovascular therapeutics.
- Enhanced Supply Chain Reliability: Relying on a synthetic route mitigates the geopolitical and logistical risks associated with sourcing raw herbs from specific regions. The starting materials for this synthesis are bulk petrochemical derivatives available from multiple global suppliers, ensuring redundancy in the supply base. This diversification protects the supply chain from disruptions caused by trade restrictions or local agricultural issues. Moreover, the synthetic product is a single chemical entity with a defined specification, simplifying the qualification process with regulatory bodies. This predictability allows supply chain planners to forecast demand more accurately and maintain optimal inventory levels without the fear of sudden quality deviations that are common in natural product supplies.
- Scalability and Environmental Compliance: The reactions described in the patent, such as hydrogenation and esterification, are well-understood unit operations that scale linearly from the laboratory to the pilot plant and eventually to commercial production. There are no exotic catalysts or extreme conditions that would pose engineering challenges at scale. From an environmental perspective, the synthetic route generates less organic waste compared to the massive volumes of plant residue generated by extraction. The use of recyclable solvents like ethyl acetate and toluene further aligns the process with green chemistry principles. This scalability ensures that as demand for Salvianolic Acid A grows in the pharmaceutical sector, production capacity can be expanded rapidly to meet market needs without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of synthetically derived Salvianolic Acid A. These answers are derived directly from the technical specifications and experimental data provided in patent CN102827002A. Understanding these details is essential for technical procurement teams evaluating the feasibility of integrating this intermediate into their drug development pipelines. The focus is on clarity regarding purity, stability, and the specific advantages of the synthetic method over traditional extraction.
Q: Why is total synthesis preferred over plant extraction for Salvianolic Acid A?
A: Plant extraction yields low purity mixtures prone to oxidation and instability due to the catechol structure. Total synthesis provides a consistent, high-purity single compound with improved stability profiles for clinical applications.
Q: What is the key challenge in the deprotection step of this synthesis?
A: The molecule contains both carboxylic esters and phenolic methyl ethers. The patent utilizes trimethyltin hydroxide for selective hydrolysis of the carboxyl ester without affecting the phenolic protection, followed by specific Lewis acid conditions for final deprotection.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the route utilizes standard organic transformations such as Darzens condensation, catalytic hydrogenation, and Wittig reactions, which are well-established and amenable to scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Salvianolic Acid A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation cardiovascular therapies. Our team of expert process chemists has extensively analyzed the synthetic route described in patent CN102827002A and possesses the technical capability to execute this complex multi-step synthesis with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or full-scale manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Salvianolic Acid A we produce. We understand that consistency is key, and our robust quality management systems are designed to deliver a product that exceeds the expectations of global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and timeline. Our technical procurement team is ready to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how our synthetic Salvianolic Acid A can optimize your supply chain. Contact us today to discuss how we can support your R&D and commercial goals with a reliable, high-purity supply of this vital pharmaceutical intermediate.
