Advanced Synthesis of 2-Fluoro-6-Chlorophenol: Overcoming Regioselectivity and Cost Barriers
The synthesis of halogenated phenols represents a cornerstone challenge in fine chemical manufacturing, particularly for the production of advanced agrochemical intermediates where regioselectivity dictates commercial viability. Patent CN1301949C discloses a robust and economically superior method for preparing 2-fluoro-6-chlorophenol, a critical building block in the pesticide industry, by employing a strategic three-step sequence of sulfonation, chlorination, and hydrolysis. This technical breakthrough addresses the longstanding inefficiencies associated with traditional electrophilic aromatic substitution on activated phenolic rings, specifically targeting the pervasive issue of para-isomer contamination that plagues direct halogenation routes. By introducing a reversible blocking group, the process ensures that the chlorine atom is directed exclusively to the desired ortho-position relative to the hydroxyl group, thereby achieving high yields and exceptional purity without the need for complex separation technologies. For global procurement and R&D teams, this patent signifies a shift towards more predictable, scalable, and cost-efficient manufacturing protocols that leverage commodity reagents rather than exotic starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-fluoro-6-chlorophenol has been hindered by two primary methodologies, both of which suffer from significant economic and technical drawbacks that impact supply chain stability. The first conventional approach involves the direct chlorination of o-fluorophenol, a reaction that is thermodynamically and kinetically prone to forming the undesired para-isomer, 2-fluoro-4-chlorophenol, in quantities up to 1.4 times greater than the target product due to steric and electronic factors. 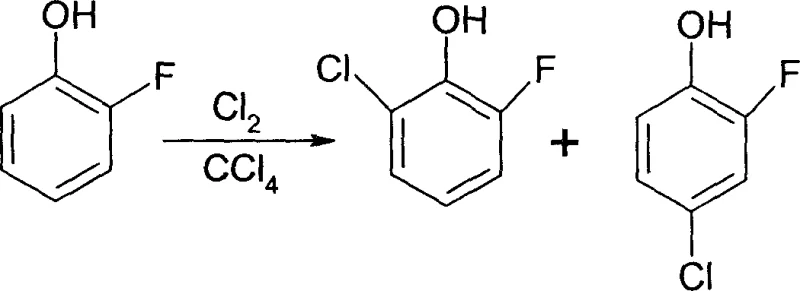 This unfavorable isomer ratio not only drastically reduces the overall yield of the desired 2-fluoro-6-chlorophenol but also necessitates the use of loaded and expensive column chromatography for purification, a technique that is notoriously difficult to scale for tonnage production. The second existing method relies on the hydrolysis of 2-fluoro-6-chloronitrobenzene, which, while chemically feasible, depends on a starting material that is neither readily available nor cost-effective in the global chemical market.
This unfavorable isomer ratio not only drastically reduces the overall yield of the desired 2-fluoro-6-chlorophenol but also necessitates the use of loaded and expensive column chromatography for purification, a technique that is notoriously difficult to scale for tonnage production. The second existing method relies on the hydrolysis of 2-fluoro-6-chloronitrobenzene, which, while chemically feasible, depends on a starting material that is neither readily available nor cost-effective in the global chemical market. 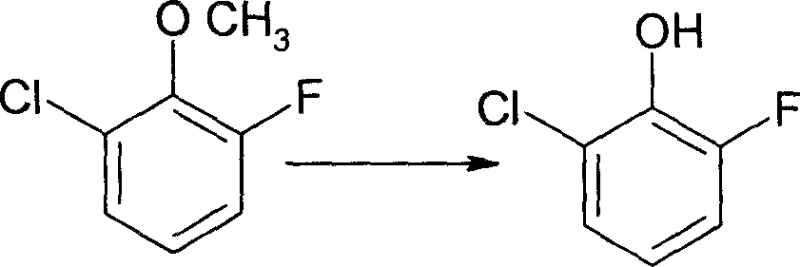 The reliance on such specialized nitro-compounds introduces severe supply chain vulnerabilities and inflates the cost of goods sold, rendering this route uncompetitive for large-scale commercial applications where margin compression is a constant pressure.
The reliance on such specialized nitro-compounds introduces severe supply chain vulnerabilities and inflates the cost of goods sold, rendering this route uncompetitive for large-scale commercial applications where margin compression is a constant pressure.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a clever "blocking group" strategy that fundamentally alters the reaction landscape to favor the target isomer with high precision. The process initiates with the sulfonation of o-fluorophenol using concentrated sulfuric acid at elevated temperatures (95°C to 100°C), which selectively installs a sulfonic acid group at the para-position, effectively shielding it from subsequent electrophilic attack. 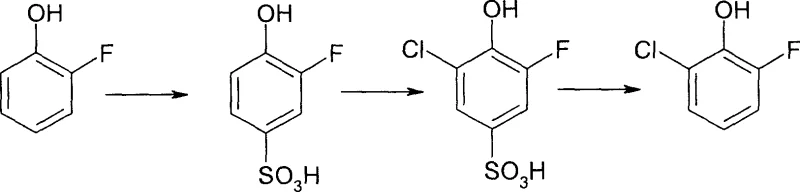 Once the para-position is blocked, the intermediate undergoes chlorination at low temperatures (-25°C to -20°C) using agents such as trichloroisocyanuric acid, ensuring that the chlorine atom substitutes exclusively at the remaining open ortho-position adjacent to the hydroxyl group. The final step involves hydrolysis, where the temporary sulfonic acid blocking group is removed under acidic distillation conditions, regenerating the phenolic ring with the chlorine atom locked in the desired 6-position. This sequence not only eliminates the formation of the problematic para-isomer but also allows for the use of inexpensive, commodity-grade reagents, thereby transforming the economic model of producing this valuable intermediate.
Once the para-position is blocked, the intermediate undergoes chlorination at low temperatures (-25°C to -20°C) using agents such as trichloroisocyanuric acid, ensuring that the chlorine atom substitutes exclusively at the remaining open ortho-position adjacent to the hydroxyl group. The final step involves hydrolysis, where the temporary sulfonic acid blocking group is removed under acidic distillation conditions, regenerating the phenolic ring with the chlorine atom locked in the desired 6-position. This sequence not only eliminates the formation of the problematic para-isomer but also allows for the use of inexpensive, commodity-grade reagents, thereby transforming the economic model of producing this valuable intermediate.
Mechanistic Insights into Sulfonation-Directed Electrophilic Substitution
The core chemical innovation of this process lies in the manipulation of electrophilic aromatic substitution rules through the temporary installation of a sulfonic acid moiety, which acts as a powerful directing and blocking agent. In the initial sulfonation step, the hydroxyl group of o-fluorophenol acts as a strong ortho/para director; however, the para-position is sterically more accessible and thermodynamically favored for the bulky sulfonic acid group under the specified reaction conditions of 95°C to 100°C. By occupying the para-site, the sulfonic acid group deactivates that specific carbon atom towards further electrophilic substitution, effectively forcing the incoming chlorine electrophile (generated from trichloroisocyanuric acid or similar agents) to attack the only remaining activated position, which is the ortho-site next to the hydroxyl group. This mechanistic control is critical because, without the blocking group, the electron-donating resonance effect of the hydroxyl group would typically favor para-substitution or a mixture of ortho/para products, leading to the impurity profile observed in direct chlorination methods.
Furthermore, the reversibility of the sulfonation reaction is ingeniously exploited in the final hydrolysis step to restore the original phenolic structure without leaving residual impurities. After the chlorination is complete, the reaction mixture is heated to boiling in the presence of water, which facilitates the desulfonation reaction, cleaving the carbon-sulfur bond and releasing sulfuric acid back into the solution. This step is particularly elegant from a process chemistry perspective because it avoids the need for additional reagents to remove the blocking group; the conditions required for product isolation (distillation) simultaneously drive the removal of the sulfonate. Consequently, the final product stream contains primarily the target 2-fluoro-6-chlorophenol, water, and regenerated sulfuric acid, simplifying the workup procedure and minimizing the generation of hazardous organic waste streams that are typical of chromatographic separations or multi-step functional group interconversions.
How to Synthesize 2-Fluoro-6-Chlorophenol Efficiently
The operational protocol for this synthesis is designed for scalability and safety, utilizing standard reactor configurations commonly found in fine chemical manufacturing facilities. The process begins by charging o-fluorophenol into a reactor equipped with agitation and temperature control, followed by the controlled addition of concentrated sulfuric acid while maintaining the temperature between 95°C and 100°C to ensure complete para-sulfonation. Following a one-hour reaction period, the mixture is cooled significantly to a range of -25°C to -20°C before the gradual addition of the chlorinating agent, a critical step that manages the exotherm and maintains the selectivity of the chlorination.
- Sulfonation: React o-fluorophenol with concentrated sulfuric acid at 95-100°C for 1 hour to introduce a sulfonic acid blocking group at the para-position.
- Chlorination: Cool the mixture to -25°C to -20°C and add a chlorinating agent (e.g., trichloroisocyanuric acid) to introduce chlorine at the remaining ortho-position.
- Hydrolysis: Heat the reaction mixture to boiling with water distillation to remove the sulfonic acid group and isolate the target product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic advantages by decoupling production from volatile niche markets and anchoring it in stable commodity chemical sectors. The primary cost driver in the traditional nitro-route is the specialized starting material, 2-fluoro-6-chloronitrobenzene, which often commands a premium price due to limited manufacturer capacity and complex upstream synthesis requirements. By switching to o-fluorophenol and sulfuric acid, manufacturers can leverage the massive global production scales of these base chemicals, resulting in a drastic reduction in raw material expenditure and insulating the supply chain from shortages of exotic intermediates. Additionally, the elimination of column chromatography—a batch-intensive and solvent-heavy purification method—in favor of continuous or semi-continuous distillation significantly lowers operational expenditures related to solvent recovery, waste disposal, and labor hours.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the substitution of expensive, hard-to-source nitro-compounds with ubiquitous and low-cost reagents like concentrated sulfuric acid and trichloroisocyanuric acid. Since sulfuric acid is one of the most produced industrial chemicals globally, its price stability ensures predictable manufacturing costs, while the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts. Furthermore, the ability to purify the final product via distillation rather than chromatography removes the need for large volumes of organic solvents and silica gel, leading to significant savings in both material costs and environmental compliance fees associated with hazardous waste treatment.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals inherently reduces supply chain risk, as o-fluorophenol and sulfuric acid are produced by numerous vendors worldwide, preventing single-source bottlenecks. In contrast, the conventional nitro-route depends on a specific fluorinated nitrobenzene derivative that may have a fragmented supplier base, making the supply chain vulnerable to disruptions. The robustness of the new method also means that production can be easily scaled up or down in response to market demand without the lead time delays associated with sourcing specialized custom intermediates, ensuring consistent delivery schedules for downstream agrochemical formulators.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is highly favorable because it avoids the generation of complex organic waste streams associated with chromatographic purification. The use of a reversible blocking group means that the only major byproduct is dilute sulfuric acid, which can often be recycled or neutralized with greater ease than mixed organic halogenated wastes. This simplified waste profile facilitates easier permitting for larger production capacities and aligns with increasingly stringent global environmental regulations, allowing manufacturers to expand capacity with lower capital investment in waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, focusing on the specific improvements over prior art methods. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, highlighting the practical benefits for industrial application.
Q: How does this process solve the para-isomer contamination issue found in direct chlorination?
A: The process utilizes a sulfonic acid group as a temporary blocking agent at the para-position. This sterically and electronically prevents chlorination at the 4-position, forcing the chlorine atom exclusively to the 6-position (ortho to the hydroxyl group), thereby eliminating the formation of the difficult-to-separate 2-fluoro-4-chlorophenol byproduct.
Q: Why is this method more cost-effective than the nitrobenzene hydrolysis route?
A: The conventional nitro-route relies on 2-fluoro-6-chloronitrobenzene, a specialized and expensive starting material with limited supply chain availability. In contrast, this patented method starts with o-fluorophenol and uses commodity chemicals like sulfuric acid and trichloroisocyanuric acid, drastically reducing raw material costs and dependency on niche suppliers.
Q: What purification methods are required for the final product?
A: Unlike the direct chlorination method which often requires tedious column chromatography to separate isomers, this process allows for purification via standard distillation. The high selectivity of the reaction yields a crude product of sufficient quality that can be refined to 99% purity using conventional distillation techniques, simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-6-Chlorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated sulfonation-chlorination chemistry described in CN1301949C can be executed with precision and consistency. Our facilities are equipped with state-of-the-art corrosion-resistant reactors capable of handling strong acids and low-temperature exotherms, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the global agrochemical industry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this high-yield process impacts your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more reliable and cost-effective source of this critical intermediate.
