Optimizing Antiviral Intermediate Production via Novel N-5-Protected Pyrimidine Synthesis Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for antiviral nucleotide derivatives, where the efficiency of intermediate production directly impacts global drug availability. Patent CN1077193A introduces a groundbreaking methodology for producing N-5-protected 2,5-diamino-4,6-dichloropyrimidines, compounds that serve as pivotal building blocks in modern medicinal chemistry. This technology addresses long-standing inefficiencies in heterocyclic synthesis by streamlining the chlorination process, thereby reducing the reliance on complex reagent systems that traditionally plague manufacturing scalability. By leveraging a novel approach to pyrimidine functionalization, this patent offers a strategic advantage for organizations aiming to secure a reliable antiviral intermediate supplier capable of meeting stringent purity standards. The core innovation lies in the ability to achieve high conversion rates without the excessive salt burdens associated with legacy methods, fundamentally altering the economic landscape of fine chemical production. For R&D teams evaluating process viability, this represents a significant opportunity to enhance yield consistency while minimizing environmental waste streams. As we delve into the technical specifics, it becomes clear that this synthesis route is not merely an incremental improvement but a substantial leap forward in process chemistry optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-diamino-4,6-dichloropyrimidine derivatives has been fraught with chemical inefficiencies that hinder commercial viability and increase operational costs. Prior art, such as the methods described by Legraverend et al., relied heavily on the use of phosphorus oxychloride in conjunction with phosphorus pentachloride and quaternary ammonium salts to drive the chlorination reaction to completion. This multi-reagent approach inevitably leads to the formation of substantial inorganic salt byproducts, creating a heavy salt burden that complicates downstream processing and waste management. Furthermore, these conventional routes often necessitate harsh reaction conditions to achieve acceptable conversion, which can degrade sensitive functional groups and lead to unpredictable impurity profiles. The reliance on ammonium salts or tertiary amine hydrochlorides, as seen in earlier PCT applications, further exacerbates the issue by introducing additional purification steps required to remove residual amines from the final product. Consequently, manufacturers face increased lead times and higher disposal costs, making cost reduction in pharmaceutical intermediate manufacturing a critical challenge. These limitations underscore the urgent need for a cleaner, more direct synthetic strategy that eliminates unnecessary reagents while maintaining high product integrity.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN1077193A achieves the desired chlorination using phosphorus oxychloride as the sole chlorinating agent, remarkably eliminating the need for auxiliary salts or catalysts. This simplification allows the reaction to proceed smoothly at moderate temperatures between 80°C and 90°C, significantly reducing energy consumption and thermal stress on the reaction mixture. By removing the requirement for quaternary ammonium salts, the process inherently avoids the generation of problematic salt wastes, thereby facilitating a much cleaner workup procedure and higher isolation yields. The absence of these additives also means that the risk of amine contamination in the final active pharmaceutical ingredient is drastically minimized, ensuring superior quality control for downstream drug synthesis. This streamlined approach not only enhances the commercial scale-up of complex heterocyclic compounds but also aligns with modern green chemistry principles by reducing the overall E-factor of the synthesis. For procurement specialists, this translates to a more predictable supply chain with fewer variables affecting batch-to-batch consistency. The ability to produce high-purity pyrimidine derivatives through such a direct route establishes a new benchmark for efficiency in the production of antiviral intermediates.
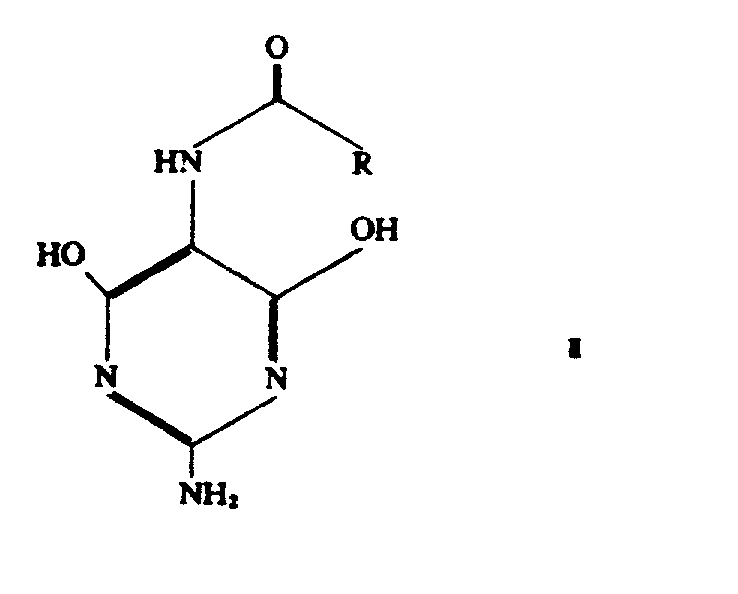
Mechanistic Insights into Phosphorus Oxychloride-Mediated Chlorination
The mechanistic elegance of this synthesis lies in the direct nucleophilic substitution of the hydroxyl groups on the pyrimidine ring by chlorine atoms using phosphorus oxychloride. In the intermediate stage, represented by the N-5-protected 4,6-dihydroxy-2,5-diaminopyrimidine structure, the hydroxyl groups are activated for substitution without the need for external activation by ammonium salts. The reaction mechanism likely involves the formation of a reactive phosphorylated intermediate which subsequently undergoes displacement by chloride ions generated in situ from the excess phosphorus oxychloride solvent. This pathway is highly selective, ensuring that the N-5 protecting group remains intact while the 4 and 6 positions are efficiently dichlorinated. The use of alkali metal alkoxides in the preceding cyclization step ensures that the pyrimidine ring is formed with high regioselectivity, setting the stage for this clean chlorination. Understanding this mechanism is crucial for R&D directors focused on impurity control, as it highlights how avoiding alternative chlorinating agents prevents the formation of side products associated with radical pathways or over-chlorination. The structural integrity of the final molecule is preserved, ensuring that the subsequent coupling reactions in antiviral drug synthesis proceed without unexpected steric or electronic hindrances.
Impurity control is further enhanced by the simplicity of the reaction matrix, which lacks the complex ionic species found in traditional ammonium salt-mediated processes. The absence of quaternary ammonium cations eliminates the possibility of ion-pairing interactions that can trap impurities within the crystal lattice of the product during precipitation. Additionally, the use of excess phosphorus oxychloride acts as both reagent and solvent, ensuring homogeneous reaction conditions that prevent localized hot spots which could lead to decomposition. The workup procedure, involving pouring the reaction mixture onto ice and adjusting pH, allows for the hydrolysis of excess phosphorylating agents into water-soluble phosphoric acid derivatives that are easily separated from the organic product. This phase separation is critical for achieving the stringent purity specifications required for pharmaceutical intermediates. By controlling the pH during the quench phase, manufacturers can selectively precipitate the desired dichloropyrimidine while keeping acidic byproducts in the aqueous layer. This level of control is essential for reducing lead time for high-purity antiviral intermediates, as it minimizes the need for extensive recrystallization or chromatographic purification steps that often bottleneck production schedules.
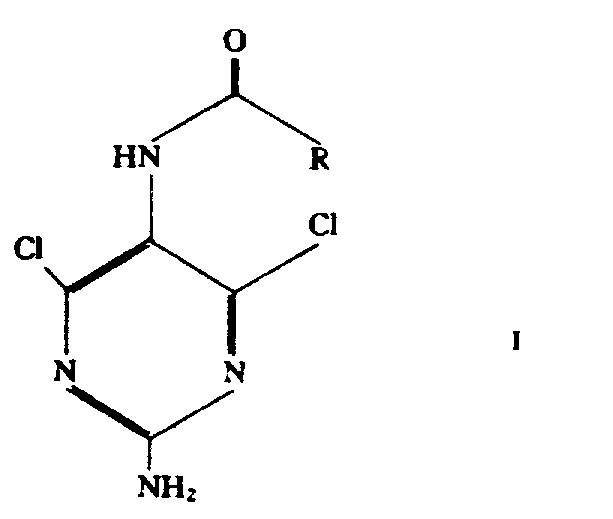
How to Synthesize N-5-Protected 2,5-Diamino-4,6-Dichloropyrimidine Efficiently
The synthesis of this critical antiviral intermediate begins with the preparation of N-protected aminomalonate esters, which serve as the foundational precursors for the pyrimidine ring construction. These starting materials are reacted with guanidine in the presence of alkali metal alkoxides, typically sodium ethoxide or methoxide, to effect cyclization under reflux conditions. This step generates the N-5-protected 4,6-dihydroxy-2,5-diaminopyrimidine intermediate, which is then isolated and subjected to the key chlorination step. The detailed standardized synthesis steps see the guide below, outlining the precise stoichiometric ratios and temperature controls necessary to replicate the patent's success on a larger scale. Adhering to these parameters ensures that the protecting group stability is maintained throughout the sequence, preventing premature deprotection that could compromise the final yield. Operators must carefully monitor the exotherm during the addition of reagents to maintain safety and product quality.
- Preparation of N-protected aminomalonate esters using alkyl haloformates or trifluoroacetyl halides under controlled low-temperature conditions.
- Cyclization reaction with guanidine in the presence of alkali metal alkoxides to form the N-5-protected 4,6-dihydroxy intermediate.
- Direct chlorination of the dihydroxy intermediate using excess phosphorus oxychloride without additional ammonium salts or catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound benefits for procurement managers and supply chain heads tasked with optimizing operational expenditures. The primary advantage stems from the drastic simplification of the reagent profile, which eliminates the need for purchasing and handling hazardous phosphorus pentachloride or expensive quaternary ammonium salts. This reduction in raw material complexity directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering both material acquisition costs and hazardous waste disposal fees. Furthermore, the simplified workup procedure reduces the consumption of solvents and utilities associated with extensive purification cycles, leading to substantial cost savings in overall production overhead. For supply chain planners, the robustness of this method implies a more reliable antiviral intermediate supplier capability, as the process is less susceptible to variability caused by reagent quality fluctuations. The ability to scale this process from laboratory to commercial production without significant re-engineering ensures continuity of supply, a critical factor for long-term drug development projects. By mitigating the risks associated with complex multi-step purifications, companies can achieve faster turnaround times and more predictable delivery schedules for their clients.
- Cost Reduction in Manufacturing: The elimination of auxiliary salts and secondary chlorinating agents significantly lowers the bill of materials, while the simplified isolation process reduces labor and utility costs associated with downstream processing. By avoiding the formation of heavy salt burdens, the facility saves on waste treatment expenses and extends the lifecycle of reactor equipment by reducing corrosion risks associated with aggressive salt mixtures. This lean manufacturing approach allows for better margin management in a competitive market environment.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available reagents like phosphorus oxychloride and alkali metal alkoxides ensures that raw material sourcing is not constrained by niche supplier availability. This ubiquity of inputs reduces the risk of supply disruptions, enabling a more resilient supply chain that can withstand market volatility. Additionally, the robustness of the reaction conditions means that production batches are less likely to fail quality control, ensuring consistent inventory levels for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous reaction nature and manageable thermal profile, facilitating the commercial scale-up of complex heterocyclic compounds without requiring specialized high-pressure equipment. From an environmental standpoint, the reduction in salt waste and the use of recyclable solvents align with increasingly strict global environmental regulations, future-proofing the manufacturing site against compliance risks. This sustainability angle is increasingly important for partnerships with major pharmaceutical corporations prioritizing green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's experimental data and claims. These insights are intended to clarify the operational benefits and chemical feasibility for stakeholders evaluating this route for potential licensing or contract manufacturing agreements. Understanding these details is vital for making informed decisions about integrating this technology into existing production portfolios. The answers reflect the specific advantages regarding yield improvement and process simplification documented in the intellectual property.
Q: How does this synthesis method improve upon conventional chlorination techniques?
A: Unlike conventional methods requiring phosphorus pentachloride or quaternary ammonium salts, this patent utilizes phosphorus oxychloride alone, eliminating significant salt burdens and simplifying downstream purification.
Q: What are the primary applications of N-5-protected 2,5-diamino-4,6-dichloropyrimidine?
A: This compound serves as a critical valuable intermediate for the production of antiviral nucleotide derivatives, specifically referenced in PCT applications for pharmaceutical development.
Q: Does the process require harsh reaction conditions for the chlorination step?
A: No, the chlorination proceeds effectively between 80°C and 90°C under reflux without the need for severe conditions or additional catalytic additives, enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-5-Protected 2,5-Diamino-4,6-Dichloropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the race to develop new antiviral therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1077193A are fully realized in a GMP-compliant environment. We are committed to delivering high-purity pyrimidine derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our expertise in heterocyclic chemistry allows us to navigate the nuances of this chlorination process, guaranteeing batch-to-batch consistency that R&D directors can trust for their clinical and commercial programs. We understand that time-to-market is essential, and our optimized workflows are designed to accelerate your development timeline without compromising on quality or safety standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your supply chain goals. Let us be your partner in transforming innovative patent chemistry into reliable commercial reality.
