Advancing Antiemetic API Production: Safe and Scalable Fosaprepitant Dimeglumine Synthesis
Advancing Antiemetic API Production: Safe and Scalable Fosaprepitant Dimeglumine Synthesis
The pharmaceutical landscape for oncology support care continues to evolve, driven by the demand for safer and more efficient manufacturing processes for critical antiemetic agents. Patent CN108948080B introduces a transformative approach to the preparation of fosaprepitant dimeglumine, a water-soluble phosphoryl prodrug of aprepitant widely used to prevent chemotherapy-induced nausea and vomiting. This innovation addresses long-standing safety and scalability bottlenecks associated with conventional synthetic routes by replacing hazardous high-pressure hydrogen gas with triethylsilane as a hydrogen donor. For global procurement and R&D teams, this shift represents a significant opportunity to optimize supply chain resilience while maintaining the stringent purity standards required for injectable pharmaceutical intermediates. The technology enables the production of high-quality active pharmaceutical ingredients without the capital intensity and safety risks of traditional高压 hydrogenation, marking a pivotal advancement in process chemistry for neurokinin-1 receptor antagonists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of fosaprepitant dimeglumine has relied heavily on palladium-catalyzed hydrogenation using molecular hydrogen gas to remove benzyl protecting groups from the phosphate ester intermediates. This traditional methodology, as documented in earlier patents such as WO9523798 and WO2006010110, necessitates the use of specialized high-pressure reaction kettles capable of withstanding significant internal pressures. The reliance on gaseous hydrogen introduces severe safety hazards, including the risk of explosion and leakage, which mandates rigorous and costly safety protocols and infrastructure. Furthermore, the requirement for high-pressure equipment severely limits the batch size and production throughput, creating a bottleneck for manufacturers attempting to scale up to meet global demand. The operational complexity of managing high-pressure hydrogenation also increases maintenance costs and downtime, making the supply chain vulnerable to disruptions and limiting the flexibility of production scheduling in multi-purpose facilities.
The Novel Approach
The innovative method disclosed in patent CN108948080B circumvents these critical limitations by employing triethylsilane as a liquid hydrogen source in the presence of a palladium catalyst. This transfer hydrogenation strategy allows the deprotection of benzyloxy groups to proceed efficiently under ambient pressure and mild temperature conditions, typically between 20°C and 40°C. By eliminating the need for gaseous hydrogen, the process removes the requirement for expensive high-pressure autoclaves, allowing the reaction to be conducted in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. This transition not only drastically enhances operational safety by removing explosion risks but also simplifies the engineering controls required for the reaction. The result is a streamlined process that yields fosaprepitant dimeglumine with superior physical appearance and high chemical purity, making it ideally suited for continuous, large-scale industrial production without the constraints of legacy high-pressure technologies.
Mechanistic Insights into Palladium-Catalyzed Transfer Hydrogenation
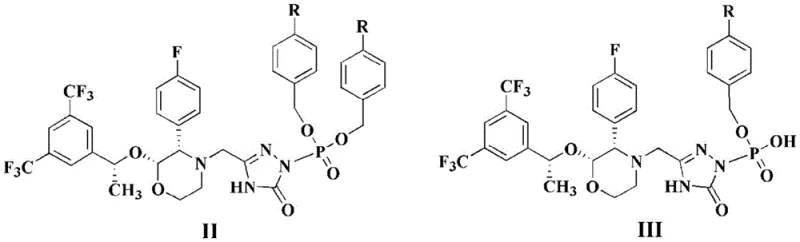
The core of this technological breakthrough lies in the mechanism of palladium-catalyzed transfer hydrogenation, where triethylsilane serves as the hydride donor for the cleavage of benzyl ethers and esters. In this catalytic cycle, the palladium surface activates the silicon-hydrogen bond of triethylsilane, generating a reactive palladium-hydride species in situ. This active hydride species then attacks the benzylic carbon of the phosphate ester intermediates (Structure II or III), facilitating the scission of the carbon-oxygen bond and releasing toluene as a byproduct while regenerating the free phosphoric acid moiety. Unlike direct hydrogenation which relies on the diffusion of gas into the liquid phase, this liquid-phase hydrogen transfer ensures more uniform contact between the reductant and the substrate, leading to consistent reaction kinetics. The simultaneous presence of N-methyl-D-glucamine in the reaction mixture allows for the immediate formation of the pharmaceutically acceptable meglumine salt, stabilizing the acidic phosphate group and driving the equilibrium towards the desired product.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods by minimizing the formation of over-reduced byproducts or phosphonate impurities often seen in harsh hydrogenation conditions. The mild reaction environment preserves the stereochemical integrity of the chiral centers within the morpholine and triazole rings, which is critical for the biological activity of the final NK-1 receptor antagonist. The use of triethylsilane also avoids the potential for metal leaching issues that can sometimes be exacerbated by high-pressure hydrogen flows, ensuring that the final product meets stringent heavy metal specifications. Furthermore, the byproduct of the silane oxidation is easily separable, and the palladium catalyst can be efficiently removed via simple filtration, resulting in a crude product with purity levels exceeding 99.5% prior to final recrystallization. This high level of selectivity reduces the burden on downstream purification steps, directly contributing to higher overall yields and reduced waste generation.
How to Synthesize Fosaprepitant Dimeglumine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing fosaprepitant dimeglumine with high efficiency and reproducibility. The process begins with the charging of the benzyl-protected intermediate, either the dibenzyl phosphate (II) or monobenzyl phosphate (III), into a reaction vessel containing a suitable solvent such as methanol or ethanol. To this mixture, a stoichiometric amount of N-methyl-D-glucamine and a catalytic loading of palladium on carbon are added, followed by the controlled addition of triethylsilane. The reaction proceeds under gentle stirring at room temperature, typically completing within 1 to 6 hours, after which the catalyst is removed by filtration. The detailed standardized synthesis steps, including specific solvent ratios, crystallization temperatures, and drying parameters required to achieve GMP-grade quality, are provided in the technical guide below.
- Charge intermediate II or III, palladium catalyst, N-methyl-D-glucamine, and triethylsilane into a reaction solvent such as methanol.
- Stir the mixture at ambient temperature (20-40°C) for 1-6 hours to effect catalytic transfer hydrogenation and salt formation.
- Filter off the catalyst, add an anti-solvent like isopropanol to crystallize the crude product, then recrystallize from methanol/acetone for final purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this triethylsilane-based synthesis route offers compelling economic and logistical benefits that extend beyond simple chemical yield. By shifting away from high-pressure hydrogenation, manufacturers can significantly reduce capital expenditure (CAPEX) associated with specialized pressure vessels and safety infrastructure, translating into lower fixed costs per kilogram of produced API. The use of liquid reagents like triethylsilane simplifies logistics and storage compared to managing bulk hydrogen gas supplies, enhancing the reliability of raw material sourcing. Additionally, the ability to run reactions in standard reactors increases facility utilization rates, allowing for greater production flexibility and faster response times to market demand fluctuations. These factors collectively contribute to a more resilient and cost-effective supply chain for this critical antiemetic intermediate.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements leads to substantial savings in both equipment investment and maintenance overheads. Operating at ambient pressure reduces energy consumption associated with compression and heating, while the high selectivity of the reaction minimizes the loss of expensive chiral starting materials. The simplified work-up procedure, involving straightforward filtration and crystallization, further lowers labor and utility costs, ensuring a highly competitive cost structure for the final pharmaceutical salt.
- Enhanced Supply Chain Reliability: Utilizing liquid hydrogen donors removes the dependency on industrial gas suppliers and the complex logistics of hydrogen transport, which can be prone to disruptions. The reagents used, such as triethylsilane and palladium carbon, are commercially available from multiple global sources, mitigating the risk of single-source bottlenecks. This diversification of the supply base ensures continuous production capability, safeguarding against shortages that could impact the availability of life-supporting oncology medications.
- Scalability and Environmental Compliance: The process is inherently scalable, as it does not face the geometric limitations of high-pressure reactors, allowing for seamless transition from pilot batches to multi-ton commercial production. From an environmental standpoint, the reaction generates minimal hazardous waste, and the absence of high-pressure gas reduces the risk of fugitive emissions. The high purity of the product reduces the need for solvent-intensive recrystallization steps, aligning with green chemistry principles and reducing the overall environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process safety, quality attributes, and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing portfolios.
Q: How does the triethylsilane method improve safety over traditional hydrogenation?
A: Traditional methods require high-pressure hydrogen gas (H2) and specialized autoclaves, posing significant explosion risks. The patented triethylsilane route operates at ambient pressure using a liquid hydrogen donor, eliminating high-pressure equipment requirements and drastically improving operational safety.
Q: What purity levels can be achieved with this novel preparation method?
A: The process yields fosaprepitant dimeglumine with exceptional purity, typically exceeding 99.5% by HPLC, with single impurities controlled below 0.1%. This high quality reduces the need for extensive downstream purification.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, by removing the dependency on high-pressure hydrogenation reactors, the process allows for the use of standard glass-lined or stainless steel reactors, facilitating easier scale-up from pilot batches to multi-ton commercial production without significant capital expenditure on pressure vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosaprepitant Dimeglumine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safe and efficient manufacturing processes for high-value pharmaceutical intermediates like fosaprepitant dimeglumine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle complex catalytic reactions, including the advanced transfer hydrogenation techniques described in recent patents, guaranteeing a consistent and reliable supply of this essential antiemetic agent.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's success and ensure uninterrupted access to high-quality fosaprepitant dimeglumine.
