Advanced Synthesis of 4,6-Dihydrofuro[3,4-d]imidazol-6-one Derivatives for High-Purity API Production
Advanced Synthesis of 4,6-Dihydrofuro[3,4-d]imidazol-6-one Derivatives for High-Purity API Production
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for complex antihypertensive agents. Patent CN1467209A introduces a groundbreaking methodology for the preparation of 4,6-dihydrofuro[3,4-d]imidazol-6-one derivatives, which serve as pivotal intermediates in the synthesis of Angiotensin II receptor antagonists such as Olmesartan. This technology addresses critical bottlenecks found in legacy manufacturing processes by offering a route that minimizes side reactions and streamlines purification. The core innovation lies in the specific structural configuration of the intermediates, which prevents unwanted etherification during subsequent coupling steps. By leveraging this intellectual property, manufacturers can achieve superior control over the impurity profile, ensuring that the final Active Pharmaceutical Ingredient meets the stringent regulatory standards required for global markets. ![General structural formula of 4,6-dihydrofuro[3,4-d]imidazol-6-one derivatives showing variable R groups](/insights/img/furo-imidazol-one-derivatives-synthesis-pharma-supplier-20260309004846-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Olmesartan intermediates has been fraught with significant technical challenges that impact both cost and timeline. Traditional methods often suffer from harsh reaction conditions that degrade sensitive functional groups, leading to a complex mixture of by-products that are difficult to separate. Specifically, the hydroxyl group on the imidazole ring is prone to unwanted etherification with the biphenyl moiety, creating structurally similar impurities that compromise the purity of the final drug substance. These issues necessitate extensive downstream processing, including multiple recrystallizations and chromatographic separations, which drastically reduce the overall yield and increase the environmental footprint of the manufacturing process. Furthermore, the reliance on unstable intermediates in older routes often results in batch-to-batch variability, posing a risk to supply chain consistency for large-scale API producers.
The Novel Approach
The methodology disclosed in the patent data presents a sophisticated solution by utilizing a protected lactone structure that inherently stabilizes the reactive centers during synthesis. This approach effectively blocks the problematic hydroxyl group until the precise moment of coupling, thereby eliminating the formation of etherification by-products. The result is a much cleaner reaction profile that allows for simpler work-up procedures and higher isolated yields. 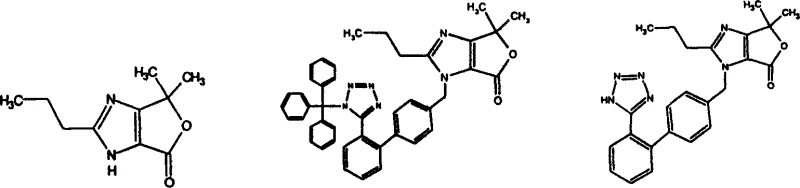
Moreover, the versatility of this synthetic route allows for the introduction of various substituents at the R1, R2, and R3 positions, providing flexibility for the development of analogues. The process utilizes readily available starting materials and standard reagents, making it highly adaptable for existing manufacturing infrastructure. By shifting to this novel pathway, pharmaceutical companies can significantly reduce the number of processing steps required to reach the final intermediate, directly translating to lower operational costs and faster time-to-market for generic versions of blockbuster antihypertensive drugs.
Mechanistic Insights into Acid-Catalyzed Lactonization
The heart of this synthetic innovation is the efficient cyclization of the hydroxy-acid precursor into the fused furo-imidazole ring system. This transformation is achieved through a carefully controlled condensation reaction where the carboxylic acid moiety reacts with the tertiary hydroxyl group to form a stable lactone ring. The mechanism typically involves the activation of the carboxyl group, either through protonation by strong mineral acids or via the formation of a mixed anhydride with reagents like trifluoroacetic anhydride. This activation renders the carbonyl carbon highly electrophilic, facilitating a nucleophilic attack by the oxygen atom of the adjacent hydroxyl group. The resulting intramolecular cyclization is thermodynamically favored due to the formation of the stable five-membered lactone ring fused to the imidazole core. 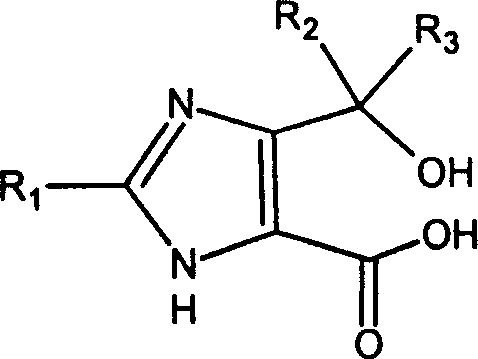
Crucially, the reaction conditions are optimized to prevent the dehydration of the tertiary alcohol into an alkene, a common side reaction in acid-catalyzed processes. By maintaining temperatures between 0°C and 50°C and selecting appropriate solvents such as dichloromethane or pyridine, the process ensures high selectivity for the desired lactone product. The use of condensing agents like dicyclohexylcarbodiimide (DCC) further enhances the efficiency of this step by driving the equilibrium towards product formation through the removal of water. This mechanistic precision is vital for maintaining the integrity of the chiral centers and ensuring that the intermediate possesses the correct stereochemistry required for biological activity in the final drug product.
How to Synthesize 4,6-Dihydrofuro[3,4-d]imidazol-6-one Efficiently
Implementing this synthesis requires a systematic approach to reagent selection and process control to maximize yield and safety. The procedure begins with the preparation of the hydroxy-acid precursor, followed by the critical cyclization step and final alkylation. Each stage demands strict adherence to stoichiometric ratios and temperature profiles to avoid the formation of trace impurities that could complicate regulatory filing. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process engineers to adapt to their specific reactor configurations and scale requirements.
- Prepare the hydroxy-acid precursor by reacting the diethyl ester with a Grignard reagent followed by hydrolysis.
- Perform the critical lactonization cyclization using a condensing agent such as trifluoroacetic anhydride or hydrobromic acid under controlled temperatures.
- Execute the final alkylation with a protected biphenyl tetrazole derivative in the presence of a base to form the final active intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical superiority. The primary advantage lies in the drastic simplification of the purification workflow, which directly correlates to reduced manufacturing cycle times and lower utility consumption. By eliminating the need for complex chromatographic separations to remove etherification by-products, facilities can increase their throughput capacity without requiring additional capital investment in equipment. This efficiency gain is particularly valuable in the context of high-volume generic drug production, where marginal cost savings per kilogram compound into substantial annual financial improvements. Furthermore, the robustness of the reaction conditions ensures consistent quality output, minimizing the risk of batch failures that can disrupt supply commitments to downstream API formulators.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage for purification steps lead to a significantly lower cost of goods sold. The process relies on commodity chemicals such as hydrobromic acid and common organic solvents, which are subject to less price volatility than specialized reagents. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final intermediate, optimizing the material balance and reducing waste disposal costs associated with failed batches or low-efficiency runs.
- Enhanced Supply Chain Reliability: The synthetic route utilizes starting materials that are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures continuity of operations even during periods of market fluctuation or logistical disruptions. The stability of the intermediates also allows for safer storage and transportation, reducing the need for specialized cold-chain logistics and enabling the establishment of regional inventory hubs to support just-in-time manufacturing models for major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, operating within standard pressure and temperature ranges that are easily manageable in large-scale stainless steel reactors. The reduction in hazardous by-products aligns with increasingly stringent environmental regulations, lowering the burden on wastewater treatment facilities. This green chemistry aspect not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for top-tier multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: How does this novel route improve upon conventional Olmesartan intermediate synthesis?
A: The patented method significantly reduces by-product formation, specifically avoiding the etherification of the hydroxyl group on the imidazole ring which plagues older methods, thereby simplifying purification and boosting overall yield.
Q: What are the preferred condensing agents for the lactonization step?
A: The process demonstrates high flexibility, successfully utilizing inorganic acids like hydrobromic acid, carbodiimides such as DCC, or acid anhydrides like trifluoroacetic anhydride, allowing manufacturers to select based on cost and availability.
Q: Is this synthesis method scalable for commercial API production?
A: Yes, the reaction conditions operate within moderate temperature ranges (-20°C to 100°C) and utilize common industrial solvents, making the transition from laboratory scale to multi-ton commercial production technically feasible and robust.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dihydrofuro[3,4-d]imidazol-6-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful launch of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 4,6-dihydrofuro[3,4-d]imidazol-6-one derivatives meets the highest international standards. Our commitment to quality assurance means that our clients can rely on us for consistent supply without compromising on the chemical integrity required for regulatory approval.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your pharmaceutical manufacturing supply chain.
