Advanced Semi-Synthesis of Deserpidine: Overcoming Selectivity Challenges for Commercial Scale-up
Advanced Semi-Synthesis of Deserpidine: Overcoming Selectivity Challenges for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex indole alkaloids, particularly those with significant antihypertensive activity like Deserpidine. Patent CN1934110A discloses a groundbreaking semi-synthetic method that transforms Reserpine Lactone into Deserpidine with high efficiency and selectivity. Historically, obtaining Deserpidine relied heavily on extraction from Rauwolfia canescens roots, where its natural abundance is critically low, ranging merely from 0.003% to 0.005%. This scarcity creates substantial bottlenecks for reliable pharmaceutical intermediates supplier networks aiming to meet global demand. The disclosed invention addresses these limitations by utilizing Reserpine, which is more abundant (0.1-0.2% in roots), as a starting material and converting it through a specialized lactone intermediate. This strategic shift from direct extraction to semi-synthesis represents a pivotal advancement in cost reduction in API manufacturing, ensuring a stable supply of this vital therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional attempts to synthesize Deserpidine directly from Reserpine or its esters have been plagued by severe chemoselectivity issues. The molecular structure of Reserpine contains multiple oxygenated functional groups, specifically a phenolic hydroxyl at the 11-position and a methoxy group at the 17-position. Conventional demethylation reagents typically lack the discrimination required to remove the 11-methoxy group (in precursors) or modify the 11-position without affecting the 17-methoxy group. Furthermore, direct reduction of the phenolic ring to a benzene ring in the presence of other sensitive functionalities often leads to over-reduction or decomposition of the complex yohimbane skeleton. These technical hurdles result in low yields and difficult purification processes, making conventional methods economically unviable for commercial scale-up of complex pharmaceutical intermediates. The inability to selectively manipulate the 11-position without protecting group strategies that are cumbersome to implement has long hindered the industrial production of Deserpidine.
The Novel Approach
The innovative process described in the patent circumvents these selectivity problems by employing Reserpine Lactone (II) as a key precursor. The formation of the lactone ring induces a critical conformational change in the molecule. In the lactone form, the substituents at positions 16, 17, and 18 adopt a specific spatial arrangement where the 17-methoxy group is locked in an axial position. This axial conformation sterically hinders the approach of demethylating reagents to the 17-position, thereby shielding it from reaction. Consequently, reagents like boron tribromide can selectively target the 11-position for demethylation or modification. This conformational locking mechanism allows for the efficient conversion of compound (II) to 11-O-deserpinic acid methyl lactone (III) and subsequently to Deserpidine Lactone (V). 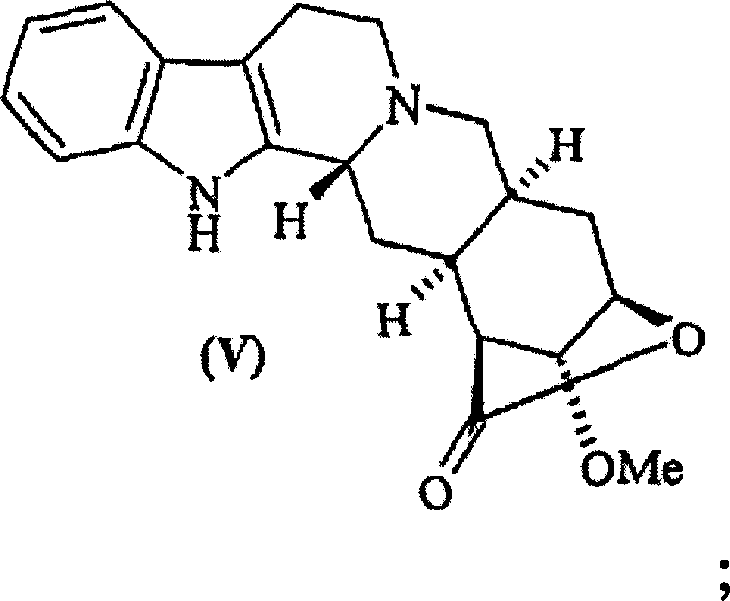
Mechanistic Insights into Selective Demethylation and Phenol Reduction
The core of this synthetic strategy lies in the precise manipulation of stereochemistry and electronic effects within the indole alkaloid framework. The selective demethylation step (Step a) utilizes strong Lewis acids such as boron tribromide (BBr3). In the Reserpine Lactone scaffold, the electron density and steric environment around the 11-oxygen differ significantly from the 17-methoxy group due to the rigid lactone bridge. The reaction proceeds via the coordination of the boron atom to the oxygen lone pairs, followed by nucleophilic attack by bromide ions. Because the 17-methoxy group is axially oriented and sterically encumbered by the lactone ring system, the activation energy for its cleavage is significantly higher than that for the 11-position. This kinetic differentiation ensures that the 11-O-demethylation occurs exclusively, preserving the integrity of the rest of the molecule and preventing the formation of unwanted side products that would complicate downstream processing.
Following demethylation, the conversion of the phenolic hydroxyl group to a benzene ring (Step b) requires the activation of the oxygen as a leaving group. The patent details the conversion of the 11-hydroxyl intermediate into a sulfonate ester, specifically a tosylate, or an isourea derivative. 
This activation transforms the poor leaving group (hydroxyl) into an excellent leaving group (tosylate or similar), facilitating the subsequent reductive cleavage. The reduction is typically carried out using catalytic hydrogenation with Raney Nickel for sulfonates or Palladium on Carbon for isoureas. The mechanism involves the adsorption of the substrate onto the metal catalyst surface, followed by the oxidative addition of hydrogen and the cleavage of the carbon-oxygen bond. This step effectively removes the oxygen functionality at the 11-position, restoring the aromatic benzene ring characteristic of Deserpidine. The specificity of Raney Nickel for sulfonate reduction is crucial here, as it avoids reducing other sensitive double bonds or functional groups within the complex alkaloid structure, ensuring high purity of the resulting Deserpidine Lactone (V).
How to Synthesize Deserpidine Efficiently
The synthesis of Deserpidine via this lactone pathway offers a reproducible and scalable protocol for laboratory and pilot plant operations. The process begins with the cyclization of Reserpine or Methylreserpate to the lactone form, followed by the critical selective demethylation and reduction steps described above. The final stages involve hydrolysis of the lactone ring to regenerate the methyl ester and subsequent esterification with 3,4,5-trimethoxybenzoyl chloride to install the final side chain. Each step has been optimized to maximize yield and minimize impurity formation, making it an ideal candidate for technology transfer. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical control points for each transformation.
- Cyclize Reserpine or Methylreserpate to form Reserpine Lactone (II) using aluminum isopropoxide in xylene.
- Perform selective demethylation at the 11-position using Boron Tribromide (BBr3) to yield 11-O-deserpinic acid methyl lactone (III).
- Convert the 11-hydroxyl group to a leaving group (e.g., tosylate) and reduce using Raney Nickel to form Deserpidine Lactone (V).
- Hydrolyze the lactone ring with sodium methoxide to obtain Methyl Deserpidine (VI), followed by esterification with 3,4,5-trimethoxybenzoyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from plant extraction to this semi-synthetic route offers profound strategic benefits. The primary advantage is the decoupling of production from agricultural variables. Natural extraction of Deserpidine is subject to seasonal fluctuations, crop failures, and geopolitical instability in sourcing regions. By synthesizing Deserpidine from the more abundant Reserpine, manufacturers can establish a continuous, year-round production schedule that is immune to harvest cycles. This stability significantly enhances supply chain reliability, ensuring that downstream API manufacturers receive consistent deliveries without the risk of raw material shortages. Furthermore, the use of well-defined chemical intermediates allows for better inventory management and forecasting, reducing the need for large safety stocks of volatile botanical extracts.
- Cost Reduction in Manufacturing: The semi-synthetic approach drastically simplifies the purification burden associated with natural product isolation. Extracting minute quantities of Deserpidine from plant matter requires massive volumes of solvent and extensive chromatographic separation to remove structurally similar alkaloids. In contrast, the synthetic route starts with a purified precursor (Reserpine Lactone) and proceeds through discrete chemical steps where impurities can be removed at each stage. This stepwise purification is far more efficient and less resource-intensive than bulk extraction. Additionally, the high selectivity of the demethylation step minimizes the loss of valuable starting material to side reactions, improving overall material throughput and reducing the cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: Relying on a chemical synthesis pathway mitigates the risks associated with the variability of botanical raw materials. Plant-derived alkaloids often exhibit batch-to-batch variations in impurity profiles due to soil conditions and climate, which can trigger costly re-validation efforts for API producers. A synthetic route provides a consistent chemical identity and impurity profile, streamlining the regulatory approval process for drug manufacturers. This consistency reduces lead time for high-purity pharmaceutical intermediates by eliminating the need for extensive incoming quality control testing associated with natural extracts. Suppliers can guarantee specifications with greater confidence, fostering stronger long-term partnerships with global pharmaceutical clients who prioritize supply security.
- Scalability and Environmental Compliance: The reagents and conditions employed in this patent, such as boron tribromide demethylation and catalytic hydrogenation, are well-established in industrial organic chemistry. These processes are readily scalable from kilogram to multi-ton quantities using standard reactor equipment. Unlike some exotic biocatalytic or enzymatic methods that may require specialized infrastructure, this chemical route leverages existing manufacturing capabilities. Moreover, the ability to recycle solvents and recover catalysts (such as Raney Nickel) aligns with modern green chemistry principles. The process avoids the generation of massive amounts of plant waste biomass associated with extraction, resulting in a lower environmental footprint and easier compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the semi-synthesis of Deserpidine. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their own production lines. The clarity provided here aims to resolve uncertainties regarding selectivity, reagent choice, and process robustness.
Q: Why is Reserpine Lactone used as a precursor instead of direct demethylation of Reserpine?
A: Direct demethylation of Reserpine lacks regioselectivity between the 11-OH and 17-OMe groups. The lactone conformation locks the 17-methoxy group in an axial position, sterically protecting it and allowing selective attack at the 11-position.
Q: What reducing agents are suitable for the phenol-to-benzene conversion step?
A: The patent specifies that Raney Nickel is essential for reducing sulfonate esters (like tosylates), while Palladium on Carbon is preferred for reducing isourea derivatives formed during the activation of the phenolic hydroxyl group.
Q: How does this synthetic route impact the purity profile compared to plant extraction?
A: Synthetic routes avoid the complex alkaloid mixtures found in Rauwolfia roots. By using defined chemical transformations, the process minimizes structurally related impurities, resulting in a cleaner impurity profile suitable for pharmaceutical grade API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deserpidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the technology disclosed in CN1934110A and is fully prepared to implement this advanced semi-synthetic pathway. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for antihypertensive APIs. We understand that the transition from lab scale to commercial manufacturing requires meticulous attention to detail, particularly in handling sensitive intermediates like the Reserpine Lactone derivatives discussed in this report.
We invite you to collaborate with us to leverage this innovative technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to not just a product, but a comprehensive solution that includes specific COA data and route feasibility assessments. Contact us today to discuss how our expertise in indole alkaloid synthesis can optimize your supply chain and reduce your overall manufacturing costs.
