Advanced Stereoselective Synthesis of Cyclic Phosphonate Intermediates for Commercial Scale-Up
Advanced Stereoselective Synthesis of Cyclic Phosphonate Intermediates for Commercial Scale-Up
The pharmaceutical landscape for metabolic disorders is rapidly evolving, with nonalcoholic steatohepatitis (NASH) emerging as a critical therapeutic area. Central to the development of next-generation treatments is the efficient production of high-purity intermediates, such as the cyclic phosphonate compound known as Formula 1 (CAS: 852948-13-1). This molecule serves as a key precursor for novel oral thyroid hormone beta receptor agonists (THR-beta agonists), which regulate gene expression to reduce liver fat and improve lipid profiles. A recent breakthrough detailed in patent CN113336792A introduces a robust preparation method that addresses long-standing challenges in stereoselectivity and yield. By leveraging specific Lewis acid catalysts and precise temperature controls, this technology enables the reliable production of the desired cis-configuration, a critical quality attribute for biological activity.
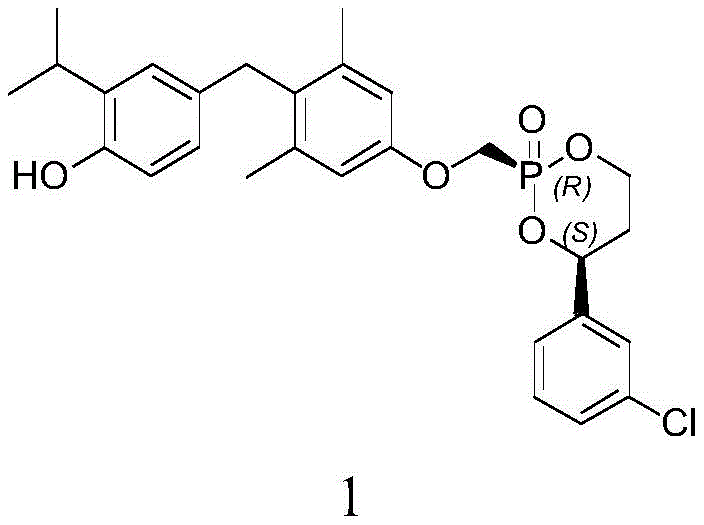
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex cyclic phosphonates has relied on condensation reactions using carbodiimide coupling agents. For instance, prior art such as patent application WO2006128055 describes reacting phosphonic acids with propylene glycol derivatives in the presence of pyridine and 1,3-dicyclohexylcarbodiimide (DCC). While chemically feasible, this conventional approach suffers from significant drawbacks when applied to chiral molecules like Formula 1. The existence of two chiral centers leads to the formation of both cis and trans isomers, but the traditional method exhibits poor stereocontrol, typically yielding a cis-to-trans ratio of merely 1.4:1 to 1.7:1. Furthermore, the overall isolated yield of the target cis-isomer is often unacceptably low, hovering around 12% to 17%. This lack of selectivity necessitates extensive and costly downstream purification processes to remove the unwanted trans-isomer and urea byproducts generated by the DCC reagent, creating a bottleneck for commercial scale-up.
The Novel Approach
The methodology disclosed in CN113336792A represents a paradigm shift in constructing the phosphonate ring system. Instead of direct condensation, the process employs a two-step strategy involving the activation of the phosphonic acid (Formula 9) into a highly reactive phosphonochloride intermediate (Formula 11). This activated species is then reacted with the chiral diol (Formula 10) under the influence of specific Lewis acids. This novel pathway dramatically alters the reaction kinetics and thermodynamics, favoring the formation of the biologically active cis-configuration. Experimental data demonstrates that this approach can achieve cis-to-trans ratios as high as 7.0:1 and substantially boost yields to over 60%. By eliminating the need for carbodiimide coupling and utilizing a more controlled activation-cyclization sequence, the new method offers a cleaner reaction profile and superior process efficiency.
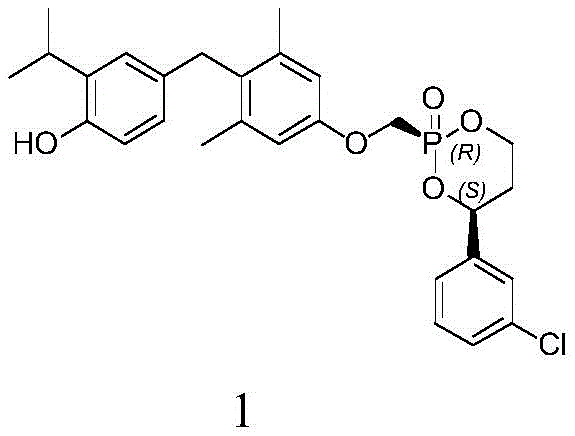
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The success of this synthesis hinges on the precise activation of the phosphorus center and the subsequent nucleophilic attack by the chiral diol. In the first stage, the phosphonic acid is converted to a phosphonochloride using chlorinating agents like thionyl chloride or oxalyl chloride, often catalyzed by trace amounts of DMF. This generates the electrophilic intermediate 11, which is far more reactive towards alcohols than the parent acid. The critical innovation lies in the second step, where a Lewis acid such as Titanium Tetrachloride (TiCl4), Tin Tetrachloride (SnCl4), or Iron(III) Chloride (FeCl3) is introduced. These Lewis acids coordinate with the oxygen atoms of the diol and potentially the phosphoryl group, organizing the transition state in a manner that kinetically favors the formation of the cis-ring closure. Interestingly, the patent notes that not all Lewis acids are suitable; for example, Aluminum Chloride (AlCl3) resulted in poor selectivity (1.4:1) and very low yields (5%), highlighting the specific electronic and steric requirements of the catalyst for this transformation.
Temperature control acts as another vital mechanistic lever in this process. The reaction between the activated phosphonochloride and the Lewis acid-complexed diol is highly exothermic and sensitive to thermal conditions. The patent data reveals that maintaining the reaction temperature strictly between -40°C and -30°C during the addition phase is optimal. At higher temperatures, such as 0°C, the stereoselectivity drops significantly (cis:trans ≈ 3.2:1), while at excessively low temperatures like -50°C, the reaction kinetics slow down too much, leading to incomplete conversion of starting materials. This narrow operational window ensures that the energy barrier for the desired cis-pathway is surmounted while suppressing the formation of the trans-isomer and preventing decomposition of the sensitive intermediates.
How to Synthesize Cyclic Phosphonate Compound 1 Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding reagent stoichiometry and thermal management. The process begins with the preparation of the phosphonochloride intermediate in a solvent like dichloromethane, followed by the separate preparation of the diol-Lewis acid complex. The integration of these two streams must be performed with high precision to maintain the critical temperature range. Below is a summary of the standardized operational protocol derived from the patent examples, designed to ensure maximum yield and purity for commercial production.
- Convert phosphonic acid compound 9 to phosphonochloride intermediate 11 using thionyl chloride and DMF catalyst.
- Prepare the chiral diol solution (compound 10) with a Lewis acid (TiCl4, SnCl4, or FeCl3) and an organic base like triethylamine.
- Add the diol mixture dropwise to the intermediate 11 solution at -40 to -30°C to maximize cis-isomer formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits beyond mere chemical elegance. The primary value driver is the substantial increase in process efficiency, which directly translates to cost reduction in pharmaceutical intermediate manufacturing. By shifting from a low-yielding DCC coupling (approx. 15% yield) to a high-yielding Lewis acid catalyzed process (approx. 60-65% yield), the amount of raw material required to produce a kilogram of the final API intermediate is drastically reduced. This efficiency gain minimizes waste generation and lowers the cost of goods sold (COGS), making the final therapeutic more economically viable. Additionally, the elimination of dicyclohexylurea byproducts simplifies the workup procedure, reducing solvent consumption and processing time.
- Cost Reduction in Manufacturing: The switch to thionyl chloride or oxalyl chloride for activation, followed by Lewis acid catalysis, removes the dependency on expensive coupling reagents like DCC or T3P. These traditional reagents are not only costly but also generate stoichiometric amounts of solid waste that are difficult to filter and dispose of on a large scale. The new method utilizes commodity chemicals that are readily available in the global market, ensuring stable pricing and supply continuity. Furthermore, the improved stereoselectivity reduces the burden on chromatographic purification, allowing for more cost-effective crystallization or extraction techniques to be used for final polishing.
- Enhanced Supply Chain Reliability: The robustness of the new process contributes significantly to supply chain stability. The tolerance for specific Lewis acids (TiCl4, SnCl4, FeCl3) provides flexibility; if one reagent faces temporary scarcity, alternatives within the approved list can be substituted without re-validating the entire process. This redundancy mitigates the risk of production stoppages due to raw material shortages. Moreover, the higher yields mean that existing reactor capacity can produce significantly more product per batch, effectively increasing manufacturing throughput without the need for capital investment in new equipment.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the new route is superior. The avoidance of carbodiimides eliminates the generation of allergenic urea byproducts. The solvents used, such as dichloromethane or tetrahydrofuran, are standard industrial solvents with well-established recovery and recycling protocols. The ability to run the reaction at controlled low temperatures (-30°C) is easily achievable with standard jacketed reactors equipped with glycol cooling systems, ensuring that the process is fully scalable from pilot plant to multi-ton commercial production without encountering unexpected thermal runaways or safety hazards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclic phosphonate synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on process optimization and quality control.
Q: What is the primary advantage of the new Lewis acid method over conventional DCC coupling?
A: The new method significantly improves stereoselectivity, achieving cis:trans ratios up to 7.0:1 compared to roughly 1.5:1 with traditional carbodiimide coupling, while also increasing yields from approximately 15% to over 60%.
Q: Which Lewis acids are most effective for this cyclic phosphonate synthesis?
A: Titanium tetrachloride (TiCl4), Tin tetrachloride (SnCl4), and Iron(III) chloride (FeCl3) are preferred. Aluminum chloride (AlCl3) was found to be unfavorable, resulting in poor selectivity and low yields.
Q: Why is temperature control critical in the cyclization step?
A: Maintaining the reaction temperature between -40°C and -30°C during the addition of the diol mixture is crucial. Deviating from this range can lead to incomplete conversion or reduced stereoselectivity, impacting the purity of the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Phosphonate Supplier
The development of NASH therapeutics requires partners who understand the intricacies of complex organic synthesis and can deliver consistent quality at scale. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with advanced cryogenic reactors capable of maintaining the strict -30°C conditions required for this Lewis acid-catalyzed cyclization, guaranteeing the high stereoselectivity demanded by regulatory agencies. We adhere to stringent purity specifications and operate rigorous QC labs to verify the cis:trans ratio and impurity profile of every batch, providing you with confidence in your supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for this critical intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can lower your overall project costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from clinical trials to commercial launch.
