Scalable Synthesis of Roxadustat Intermediate via Three-Step Erlenmeyer-Plöchl and Bischler-Napieralski Sequence
Introduction to Advanced Roxadustat Intermediate Synthesis
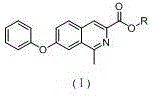
The pharmaceutical landscape for treating renal anemia has been significantly transformed by Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors (HIF-PHIs), with Roxadustat standing as a pioneering molecule in this class. Central to the efficient manufacturing of this vital therapeutic agent is the availability of high-quality key intermediates, specifically 1-methyl-7-phenoxyisoquinoline-3-carboxylic esters. Patent CN109305940B discloses a groundbreaking synthetic methodology that addresses longstanding bottlenecks in the production of these critical building blocks. By leveraging a concise three-step sequence involving Erlenmeyer-Plöchl condensation, alcoholysis, and Bischler-Napieralski cyclization, this technology offers a robust pathway that contrasts sharply with the convoluted multi-step processes previously dominant in the industry. For R&D directors and procurement strategists, understanding this shift is paramount, as it represents a move towards greener, more cost-effective, and scalable chemical manufacturing that aligns with modern supply chain resilience goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
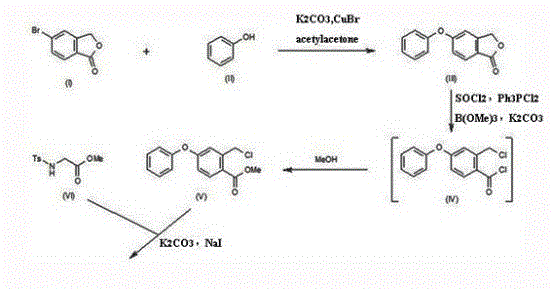
Prior to the innovations detailed in CN109305940B, the synthesis of Roxadustat intermediates was plagued by inefficiencies that severely impacted both cost and operational safety. Historical routes, such as those described in earlier patents, often necessitated upwards of seven to nine distinct reaction steps to reach the target isoquinoline scaffold. These legacy pathways frequently relied on hazardous reagents and extreme conditions; for instance, some methods required the use of cuprous bromide, introducing heavy metal contamination risks that demand expensive and complex purification protocols to meet stringent pharmaceutical standards. Furthermore, other reported syntheses involved the use of thionyl chloride, which poses severe corrosion risks to reactor vessels and necessitates sophisticated tail-gas absorption systems to mitigate environmental pollution. Perhaps most critically from a capital expenditure perspective, certain routes demanded cryogenic conditions as low as -78°C requiring liquid nitrogen cooling, or the use of metallic sodium and flammable gases like trimethylborane, creating significant safety hazards and limiting the feasibility of large-scale industrial application.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach presented in the patent streamlines the entire synthetic trajectory into a highly efficient three-step process. This methodology initiates with the condensation of readily available 4-phenoxybenzaldehyde and N-acetyl glycine, bypassing the need for exotic starting materials like 5-bromo-1(3H)-isobenzofuran. The subsequent alcoholysis and cyclization steps are conducted under mild thermal conditions, typically ranging between 30°C and 130°C, thereby eliminating the energy-intensive requirements for ultra-low temperature refrigeration or high-pressure hydrogenation equipment. By removing transition metal catalysts like copper from the equation, the process inherently reduces the burden on downstream purification, ensuring a cleaner impurity profile without the need for specialized metal scavenging resins. This radical simplification not only shortens the production cycle time drastically but also enhances the overall process safety profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for continuous, uninterrupted manufacturing campaigns.
Mechanistic Insights into Erlenmeyer-Plöchl Condensation and Bischler-Napieralski Cyclization
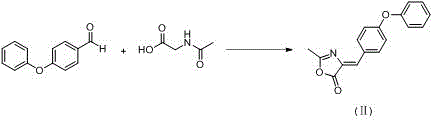
The core of this synthetic innovation lies in the strategic application of two classic organic transformations: the Erlenmeyer-Plöchl condensation and the Bischler-Napieralski cyclization. The first step involves the reaction of 4-phenoxybenzaldehyde with N-acetyl glycine in the presence of acetic anhydride and a base such as sodium acetate. This condensation effectively constructs the carbon-nitrogen backbone required for the heterocyclic ring, generating an azlactone-like intermediate (Formula II) with high atom economy. The mechanistic precision here ensures that the phenoxy substituent remains intact while establishing the necessary connectivity for the subsequent ring closure. Following this, an alcoholysis step converts the intermediate into an acyclic precursor (Formula III), which exists as a mixture of Z and E isomers. This step is crucial as it prepares the molecule for the final cyclization by installing the ester functionality that defines the diverse library of intermediates (methyl, ethyl, isopropyl, etc.) capable of being produced through simple alcohol variation.
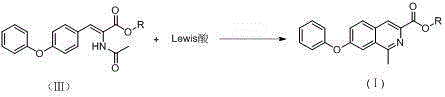
The final and most critical transformation is the Bischler-Napieralski cyclization, which closes the isoquinoline ring system. This step utilizes a Lewis acid, such as phosphorus oxychloride (POCl3), zinc chloride, or aluminum chloride, to activate the amide carbonyl for intramolecular electrophilic aromatic substitution. The reaction proceeds efficiently in solvents like dichloromethane or chloroform at moderate temperatures (30-90°C), driving the formation of the aromatic isoquinoline core (Formula I). From an impurity control perspective, this method is superior because it avoids the generation of halogenated byproducts associated with thionyl chloride usage in older routes. The robustness of the Lewis acid mediated cyclization ensures high conversion rates and minimizes the formation of regio-isomers, thereby simplifying the crystallization and purification stages. This mechanistic clarity provides R&D teams with a predictable and controllable process window, essential for maintaining the stringent quality specifications required for API intermediates.
How to Synthesize 1-Methyl-7-Phenoxyisoquinoline-3-Carboxylic Ester Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity, particularly during the cyclization phase where moisture sensitivity can be a factor. The process begins with the careful addition of reactants for the condensation step, followed by a controlled workup to isolate the intermediate before proceeding to alcoholysis. The final cyclization demands anhydrous conditions and the slow addition of the Lewis acid to manage exotherms effectively. While the general procedure is straightforward, scaling this chemistry requires attention to heat transfer and mixing efficiency to ensure uniform reaction progress throughout the batch. For detailed operational protocols, stoichiometry ratios, and specific workup procedures tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Perform Erlenmeyer-Plöchl condensation between 4-phenoxybenzaldehyde and N-acetyl glycine using acetic anhydride and sodium acetate at 90-130°C.
- Conduct alcoholysis of the resulting intermediate with alcohol (e.g., methanol, ethanol) in the presence of a base like sodium carbonate at 60-70°C.
- Execute Bischler-Napieralski cyclization using a Lewis acid such as phosphorus oxychloride in a solvent like dichloromethane at 30-90°C to form the final isoquinoline ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this three-step synthesis route offers transformative benefits that extend far beyond simple chemical efficiency. The primary advantage lies in the drastic reduction of process complexity, which directly correlates to lower operational expenditures and reduced risk of supply disruption. By eliminating the need for seven to nine reaction steps, manufacturers can significantly reduce the consumption of solvents, reagents, and labor hours, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the reliance on commodity chemicals like 4-phenoxybenzaldehyde and acetic anhydride ensures that raw material sourcing is stable and less susceptible to the volatility often seen with specialized, low-volume reagents. This stability is crucial for maintaining consistent lead times and securing long-term supply contracts with key stakeholders in the pharmaceutical value chain.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as metallic sodium, cuprous bromide, and trimethylborane removes the necessity for specialized handling equipment and extensive waste treatment protocols. Without the need for cryogenic cooling down to -78°C or high-pressure hydrogenation reactors, the capital investment required for production facilities is markedly lower, allowing for more flexible allocation of resources. Additionally, the removal of heavy metal catalysts simplifies the purification workflow, reducing the consumption of costly chromatography media or metal scavengers, which further drives down the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of supply bottlenecks that often plague complex synthetic routes dependent on custom-synthesized precursors. Since the reaction conditions are mild and do not require extreme temperatures or pressures, the process can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure means that production can be scaled up rapidly without the need for lengthy equipment fabrication or installation timelines, ensuring that delivery schedules are met consistently even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly friendlier, avoiding the generation of corrosive sulfur dioxide gas and toxic heavy metal waste streams. The simplified three-step sequence reduces the total volume of organic waste generated per kilogram of product, lowering disposal costs and easing the regulatory burden associated with environmental permits. The absence of pyrophoric reagents and the use of moderate reaction temperatures enhance overall plant safety, reducing insurance premiums and minimizing the risk of unplanned shutdowns due to safety incidents, thereby ensuring a continuous and reliable supply of high-purity intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process robustness and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis route over prior art methods?
A: This route reduces the synthesis from 7-9 steps down to only 3 steps, eliminates the need for hazardous reagents like metallic sodium or cryogenic conditions (-78°C), and avoids heavy metal catalysts like copper bromide, significantly simplifying purification and environmental compliance.
Q: Which Lewis acids are suitable for the final cyclization step?
A: The patent specifies several effective Lewis acids including phosphorus trichloride, phosphorus pentachloride, phosphorus oxychloride, zinc chloride, and aluminum chloride, offering flexibility in reagent sourcing and process optimization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials, operates under mild temperatures (30-130°C), avoids specialized high-pressure or ultra-low temperature equipment, and demonstrates high yields, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-7-Phenoxyisoquinoline-3-Carboxylic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the global supply of life-saving medications like Roxadustat. Our team of expert chemists has extensively analyzed the pathway disclosed in CN109305940B and possesses the technical capability to execute this three-step sequence with precision and reliability. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 1-methyl-7-phenoxyisoquinoline-3-carboxylic ester we deliver is ready for immediate use in downstream API synthesis.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced synthetic route for their Roxadustat programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined process for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities, allowing you to validate the superior quality and cost-efficiency of our intermediates firsthand.
