Advanced Synthesis of Tunable Near-Infrared II Dyes for Deep Tissue Imaging Applications
Advanced Synthesis of Tunable Near-Infrared II Dyes for Deep Tissue Imaging Applications
The rapid evolution of biomedical imaging technologies has created an urgent demand for fluorophores capable of operating within the second near-infrared window (NIR-II, 900-1700 nm). Patent CN111849196B addresses this critical market need by disclosing a novel class of organic fluorescent dyes characterized by the general Formula I. These compounds represent a significant leap forward in optical performance, offering exceptional chemical stability, photostability, and a large Stokes shift that minimizes background interference. Unlike traditional cyanine dyes which often suffer from aggregation-caused quenching and poor stability in physiological environments, the molecular architecture defined in this patent provides a robust platform for deep-tissue visualization. The invention specifically targets the limitations of current imaging agents by introducing tunable structural sites that allow for precise modulation of photophysical properties. 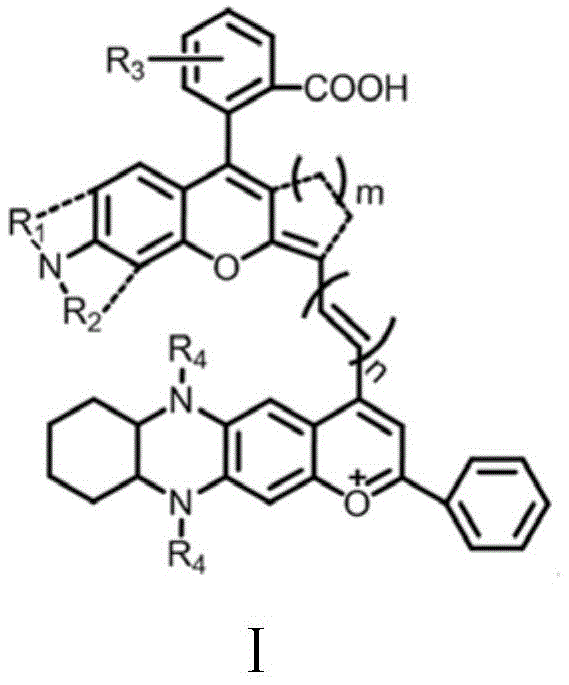 For procurement specialists and supply chain managers in the life sciences sector, understanding the synthetic accessibility of these high-value intermediates is paramount. The patent outlines a convergent synthesis strategy that relies on readily available commodity chemicals, suggesting a pathway for cost-effective manufacturing without compromising on the purity required for in vivo applications. As a reliable NIR-II dye supplier, analyzing the technical nuances of this intellectual property reveals opportunities for optimizing the production of next-generation imaging probes.
For procurement specialists and supply chain managers in the life sciences sector, understanding the synthetic accessibility of these high-value intermediates is paramount. The patent outlines a convergent synthesis strategy that relies on readily available commodity chemicals, suggesting a pathway for cost-effective manufacturing without compromising on the purity required for in vivo applications. As a reliable NIR-II dye supplier, analyzing the technical nuances of this intellectual property reveals opportunities for optimizing the production of next-generation imaging probes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of fluorescence imaging has been dominated by fluorophores operating in the visible and first near-infrared (NIR-I) regions, such as Indocyanine Green (ICG). However, these conventional agents face severe physical limitations when applied to deep-tissue imaging. The primary drawback is the strong autofluorescence of biological tissues in the visible spectrum, which creates a high background noise level and drastically reduces the signal-to-noise ratio. Furthermore, photon scattering increases significantly at shorter wavelengths, limiting the penetration depth to merely a few millimeters and preventing high-resolution visualization of structures located centimeters beneath the surface. From a chemical stability perspective, many existing NIR-I dyes are prone to rapid photobleaching under continuous laser irradiation, leading to signal loss during long-term surgical guidance or longitudinal studies. Additionally, the phenomenon of solvatochromism often results in unpredictable shifts in emission maxima when the dye moves from an organic solvent to an aqueous biological environment, complicating quantitative analysis. These inherent deficiencies necessitate the development of new molecular scaffolds that can overcome the optical window barriers while maintaining structural integrity under physiological conditions.
The Novel Approach
The technology disclosed in CN111849196B introduces a sophisticated molecular design that effectively circumvents the pitfalls of earlier generations of dyes. By extending the conjugated pi-system and incorporating rigid fused-ring structures, the inventors have successfully shifted the emission wavelength into the NIR-II region, where tissue absorption and scattering are minimized. The core structure, as depicted in Formula I, integrates a benzopyrylium moiety linked to a functionalized phenazine derivative, creating a push-pull electronic system that enhances intramolecular charge transfer. This architectural choice not only redshifts the absorption and emission but also imparts remarkable resistance to solvent-induced quenching. 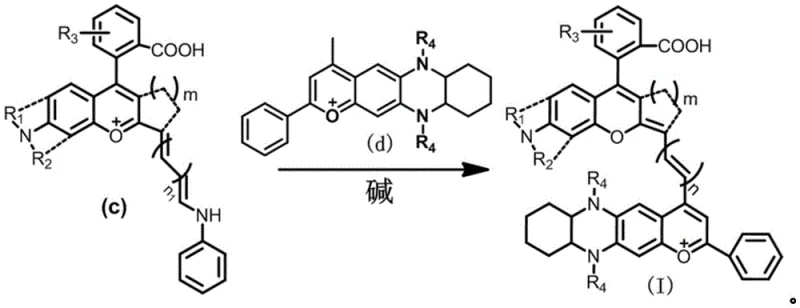 The synthetic methodology described is equally innovative, employing a modular approach that facilitates structural diversification. The final condensation step utilizes acetic anhydride as both solvent and dehydrating agent, driving the reaction to completion with high efficiency. This contrasts sharply with older methods that might require toxic chlorinated solvents or expensive transition metal catalysts, thereby simplifying the downstream purification process. For manufacturers, this translates to a streamlined workflow that reduces waste generation and lowers the overall cost of goods sold, making high-performance NIR-II imaging more accessible for widespread clinical and research adoption.
The synthetic methodology described is equally innovative, employing a modular approach that facilitates structural diversification. The final condensation step utilizes acetic anhydride as both solvent and dehydrating agent, driving the reaction to completion with high efficiency. This contrasts sharply with older methods that might require toxic chlorinated solvents or expensive transition metal catalysts, thereby simplifying the downstream purification process. For manufacturers, this translates to a streamlined workflow that reduces waste generation and lowers the overall cost of goods sold, making high-performance NIR-II imaging more accessible for widespread clinical and research adoption.
Mechanistic Insights into the Convergent Condensation Strategy
The synthesis of the target NIR-II dyes relies on a classic yet highly effective Knoevenagel-type condensation mechanism, adapted here for the construction of a complex polymethine bridge. The reaction involves the coupling of an activated methyl group on the benzopyrylium intermediate (Intermediate C) with an aldehyde functionality present on the phenazine-based Intermediate D. In the presence of a mild base such as potassium acetate or potassium carbonate, the acidic protons alpha to the positively charged oxygen in the benzopyrylium ring are abstracted, generating a nucleophilic carbanion. This nucleophile then attacks the electrophilic carbonyl carbon of the aldehyde group on Intermediate D, forming a beta-hydroxy intermediate. Subsequent dehydration, facilitated by the elevated reaction temperature of 110-130°C and the dehydrating nature of the acetic anhydride solvent, yields the extended conjugated double bond system essential for NIR-II emission. This mechanism is particularly advantageous because it avoids the use of harsh bases that could degrade the sensitive heterocyclic cores, ensuring high fidelity in the final product structure.
Impurity control is another critical aspect addressed by this mechanistic pathway. The use of acetic anhydride serves a dual purpose: it acts as the reaction medium and effectively scavenges water produced during the condensation, pushing the equilibrium towards the desired product and minimizing the formation of hydrolysis byproducts. Furthermore, the distinct polarity differences between the starting intermediates and the final highly conjugated dye allow for efficient purification via silica gel column chromatography. The patent specifies the use of dichloromethane-ethanol gradients, which effectively separate the target dye from unreacted starting materials and side products. This level of control over the impurity profile is vital for pharmaceutical applications, where strict limits on organic impurities must be met to ensure biocompatibility and regulatory compliance for in vivo diagnostic use.
How to Synthesize NIRII-RT1 Efficiently
The practical implementation of this technology is best illustrated through the specific embodiment of NIRII-RT1, where m=1 and n=1. The process begins with the independent preparation of the two key building blocks: the benzopyrylium salt (Intermediate C) and the phenazine derivative (Intermediate D). Intermediate C is synthesized by heating a substituted flavylium salt with an aromatic aldehyde in acetonitrile, a polar aprotic solvent that supports the ionic nature of the intermediates. Meanwhile, Intermediate D requires a multi-step sequence starting from 4-methoxy o-phenylenediamine and 1,2-cyclohexanedione, involving reduction and cyclization steps to establish the electron-rich donor core. Once both intermediates are purified to high standards, they are combined in the final coupling reaction. 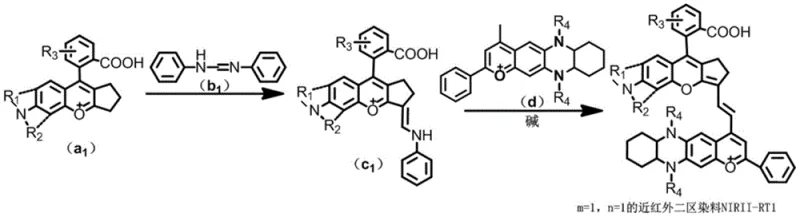 The detailed standardized synthesis steps for producing this high-purity fluorophore are outlined below.
The detailed standardized synthesis steps for producing this high-purity fluorophore are outlined below.
- Synthesize Intermediate C by reacting a substituted benzopyrylium salt (Compound A) with an aldehyde derivative (Compound B) in acetonitrile at 70-90°C.
- Prepare Intermediate D via a multi-step sequence involving the condensation of 4-methoxy o-phenylenediamine with 1,2-cyclohexanedione, followed by reduction and cyclization with 1-phenyl-1,3-butanedione.
- Perform the final coupling reaction by heating Intermediate C and Intermediate D with potassium acetate in acetic anhydride at 110-130°C for 2-3 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic route described in CN111849196B offers tangible strategic benefits beyond mere technical performance. The reliance on commodity feedstocks such as cyclohexanedione, phenylenediamines, and acetic anhydride ensures a stable and resilient supply chain, insulating production from the volatility often associated with exotic or scarce reagents. Furthermore, the avoidance of precious metal catalysts in the final coupling step eliminates the need for expensive metal scavenging processes and rigorous heavy metal testing, which are significant cost drivers in fine chemical manufacturing. This simplification of the downstream processing workflow directly contributes to substantial cost savings and faster batch turnover times. The robustness of the reaction conditions, which tolerate moderate temperatures and standard atmospheric pressure, further enhances the feasibility of scaling this process from gram-scale laboratory synthesis to multi-kilogram commercial production without requiring specialized high-pressure equipment.
- Cost Reduction in Manufacturing: The synthetic pathway leverages inexpensive, bulk-available organic solvents like acetonitrile and acetic anhydride, avoiding the need for costly anhydrous conditions or cryogenic temperatures often required in organometallic chemistry. By eliminating transition metal catalysts from the final bond-forming step, the process removes the financial burden associated with catalyst recovery and the validation of residual metal levels, leading to a significantly simplified purification protocol. This streamlining of the manufacturing process reduces both material costs and labor hours, resulting in a more competitive price point for the final high-purity NIR-II dye intermediates.
- Enhanced Supply Chain Reliability: The starting materials identified in the patent, including substituted benzopyrylium salts and cyclohexanediones, are established industrial chemicals with multiple global sources, mitigating the risk of single-supplier dependency. The synthetic route is designed with operational simplicity in mind, utilizing standard glass-lined reactors and filtration equipment that are ubiquitous in fine chemical facilities. This compatibility with existing infrastructure means that production can be ramped up quickly to meet surging demand for imaging agents without the need for capital-intensive plant modifications, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its homogeneous reaction conditions and the absence of hazardous gaseous reagents. The use of acetic anhydride, while requiring careful handling, allows for efficient solvent recovery and recycling systems, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing operation. Additionally, the high selectivity of the condensation reaction minimizes the generation of complex byproduct mixtures, simplifying waste treatment and disposal procedures, which is a critical factor for maintaining regulatory compliance in increasingly stringent environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced imaging materials. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these details is essential for partners looking to integrate these dyes into their own diagnostic or therapeutic pipelines.
Q: What are the primary advantages of the NIR-II dyes disclosed in CN111849196B compared to traditional ICG?
A: The dyes described in this patent exhibit superior chemical and photostability, larger Stokes shifts, and reduced solvent-induced quenching compared to commercial dyes like ICG and IR1061, enabling deeper tissue penetration and higher resolution imaging.
Q: Is the synthesis process scalable for commercial production of these fluorophores?
A: Yes, the synthetic route utilizes robust reaction conditions, such as heating in acetic anhydride and acetonitrile, and avoids sensitive transition metal catalysts in the final step, making it highly amenable to kilogram-scale manufacturing.
Q: How does the structural tunability of Formula I benefit probe design?
A: The presence of variable substituents (R1-R4) and adjustable conjugation lengths (m and n) allows chemists to fine-tune the emission wavelength within the 900-1700nm window and optimize solubility or bioconjugation properties for specific biological targets.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NIR-II Dye Supplier
The technological breakthroughs detailed in CN111849196B underscore the immense potential of next-generation NIR-II fluorophores in revolutionizing medical diagnostics and biological research. At NINGBO INNO PHARMCHEM, we recognize the complexity involved in translating such sophisticated molecular designs into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of high-purity near-infrared organic fluorescent dyes meets the exacting standards required for in vivo applications.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your specific imaging needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your project's economic viability. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate the development of your advanced fluorescence imaging solutions.
