Advanced Synthesis of Chiral Spiroalkene Derivatives for High-Performance OLED Applications
Advanced Synthesis of Chiral Spiroalkene Derivatives for High-Performance OLED Applications
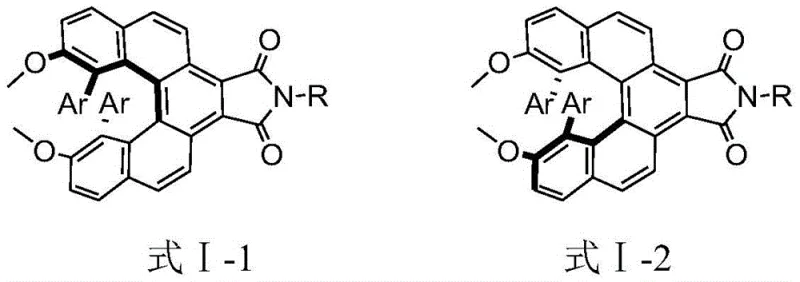
The rapid evolution of next-generation display technologies has intensified the demand for advanced organic luminescent materials capable of emitting circularly polarized light (CPL). Patent CN111559973B discloses a groundbreaking class of phthalimide-substituted spiroalkene derivatives that address critical stability and efficiency challenges in this field. These novel compounds, characterized by a rigid helical chirality and a large pi-conjugated skeleton, offer a robust solution for fabricating electroluminescent devices with superior performance. The disclosed preparation method leverages a strategic combination of palladium-catalyzed cross-coupling and high-temperature thermal aromatization to construct the complex spiro-core with high precision. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a significant leap forward in creating reliable OLED material supplier pipelines that can deliver high-purity spiroalkene derivative products with consistent optical activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiroolefins with helical chirality has been plagued by significant technical hurdles that hinder their commercial viability in cost reduction in electronic chemical manufacturing. Traditional routes often suffer from low luminous quantum yields and, more critically, a propensity for chiral racemization under mild conditions, which destroys the desired CPL properties. Furthermore, the derivatization of higher-order spiroalkenes is frequently limited by difficult preparation protocols and inefficient chiral resolution processes. Many existing methods rely on harsh conditions that degrade the delicate aromatic systems or require expensive, non-recyclable chiral auxiliaries that drive up production costs. The inability to maintain configurational stability during device operation or post-processing remains a primary bottleneck, forcing manufacturers to compromise on device lifetime or brightness. Consequently, the industry has struggled to find a scalable pathway that balances structural complexity with the rigorous stability requirements of modern optoelectronic applications.
The Novel Approach
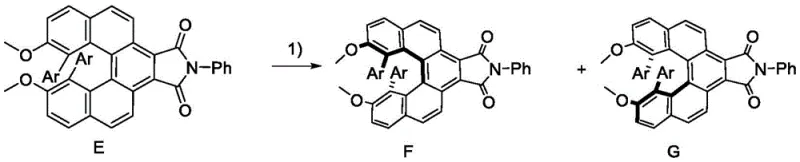
The methodology outlined in the patent introduces a transformative synthetic strategy that overcomes these legacy issues through a streamlined four-step sequence. By utilizing a dibromo-biaryl precursor and subjecting it to Suzuki-Miyaura coupling, the process efficiently installs diverse aryl groups to tune electronic properties without compromising the core architecture. The subsequent reaction with maleic anhydride followed by a unique thermal dehydrogenation step using sulfur and palladium on carbon creates a fully aromatized, rigid spiro-center. This specific thermal treatment is pivotal, as it locks the helical conformation, resulting in a product with an exceptionally high racemization energy barrier. Unlike conventional methods, this approach allows for the introduction of various functional groups (such as cyano, aldehyde, or amino substituents) on the peripheral phenyl rings, enabling fine-tuning of the HOMO-LUMO levels. The final imide formation and chiral resolution yield optically pure materials that are not only stable but also exhibit strong circular polarization luminescence, marking a distinct advantage for high-end display manufacturing.
Mechanistic Insights into Pd-Catalyzed Coupling and Thermal Aromatization
The core of this synthesis lies in the precise orchestration of transition metal catalysis and thermal elimination reactions to build the sterically crowded spiro-junction. The initial step involves a tetrakis(triphenylphosphine)palladium-catalyzed cross-coupling between the dibromo precursor and arylboronic acids in a toluene/ethanol/aqueous carbonate system. This reaction proceeds through a standard oxidative addition, transmetallation, and reductive elimination cycle, but the steric bulk of the biaryl system requires careful optimization of ligand loading (1-10 mol%) to ensure complete conversion. Following this, the Diels-Alder-type addition of maleic anhydride to the olefinic bond sets the stage for the critical aromatization. The subsequent heating at 270-300°C in the presence of sulfur and Pd/C facilitates a double dehydrogenation, effectively removing four hydrogen atoms to restore full aromaticity across the fused ring system. This high-temperature step is thermodynamically driven and is essential for planarizing the adjacent rings while maintaining the orthogonal spiro-geometry.
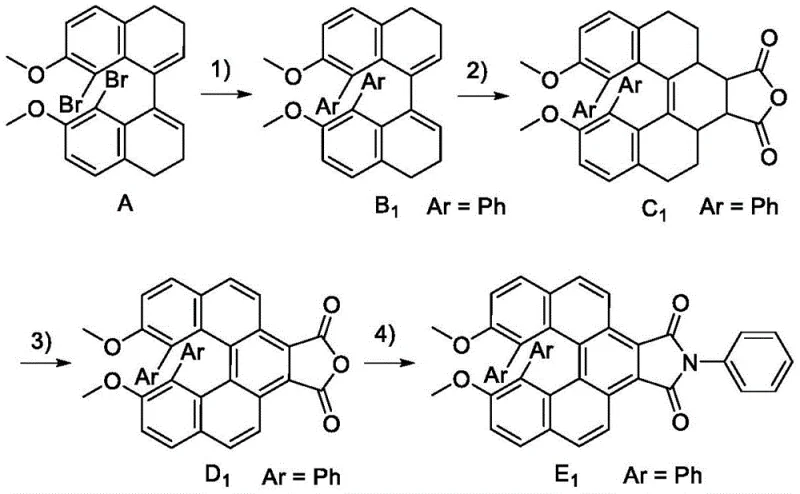
Controlling the impurity profile during this multi-step synthesis is paramount for achieving the high-purity spiroalkene derivative standards required in the semiconductor industry. The use of sulfur and palladium carbon in the aromatization step acts as a potent dehydrogenation system that minimizes the formation of partially saturated byproducts which could act as quenchers in the final luminescent device. Furthermore, the condensation of the resulting anhydride with amines in DMF at 90-110°C is highly selective, forming the phthalimide moiety without affecting the sensitive spiro-center. The final purification via preparative chiral HPLC using a CHIRALPAK® IE column ensures the separation of enantiomers with >99% ee, effectively removing any trace racemic material that could degrade the circular polarization ratio. The robustness of the spiro-structure is confirmed by thermal stress testing, where the compounds show no signs of racemization even after prolonged exposure to 300°C, validating the efficacy of the mechanistic design in locking chirality.
How to Synthesize Chiral Spiroalkene Derivatives Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating these high-value intermediates, suitable for both laboratory scale-up and pilot production. The process begins with the coupling of the brominated biaryl scaffold, followed by anhydride addition and a high-temperature aromatization step that defines the molecular rigidity. The final amidation and chiral separation steps ensure the delivery of the specific enantiomer required for CPL applications. Detailed operational parameters, including solvent ratios and temperature gradients, are critical for maximizing yield and optical purity. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Perform Suzuki coupling of dibromo precursor with arylboronic acid using Pd(PPh3)4 catalyst in toluene/ethanol.
- React the biaryl intermediate with maleic anhydride in refluxing xylene, followed by thermal aromatization with sulfur and Pd/C.
- Condense the aromatic anhydride with an amine in DMF to form the phthalimide spiroalkene derivative.
- Separate enantiomers using preparative chiral HPLC (e.g., CHIRALPAK IE) to obtain optically pure CPL materials.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers substantial benefits regarding raw material accessibility and process scalability, directly addressing the needs of a reliable OLED material supplier. The starting materials, including substituted arylboronic acids and maleic anhydride, are commodity chemicals available from multiple global vendors, mitigating the risk of single-source dependency. The reaction conditions utilize common organic solvents such as toluene, xylene, and DMF, which simplifies solvent recovery and waste management protocols compared to exotic fluorinated solvents often used in fine chemical synthesis. Moreover, the elimination of sensitive organometallic reagents (like organolithiums) in favor of robust boronic acid coupling enhances operational safety and reduces the need for cryogenic infrastructure. This translates to a drastically simplified manufacturing process that lowers the barrier for commercial scale-up of complex polymer additives and small molecule emitters alike.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing earth-abundant catalysts and avoiding expensive chiral pool starting materials. Since the chirality is resolved at the final stage using chromatography rather than built-in via costly chiral auxiliaries, the overall material cost is significantly reduced. The high thermal stability of the intermediates allows for aggressive purification techniques like recrystallization and sublimation, which are far more economical than repeated chromatographic separations. Additionally, the high yield reported in the examples suggests minimal material loss, further driving down the cost per gram of the active pharmaceutical ingredient or electronic material.
- Enhanced Supply Chain Reliability: The robustness of the synthetic steps ensures consistent batch-to-batch quality, a critical factor for reducing lead time for high-purity spiroalkene derivatives. The tolerance of the reaction to various substituents (cyano, trifluoromethyl, methoxy) means that a single platform technology can produce a library of derivatives, allowing suppliers to pivot quickly based on customer demand without retooling entire production lines. The use of standard equipment (reflux condensers, high-temp reactors) means that contract manufacturing organizations (CMOs) can easily adopt this process without capital-intensive upgrades, ensuring a stable and continuous supply flow for downstream device manufacturers.
- Scalability and Environmental Compliance: The synthesis avoids the generation of heavy metal waste streams associated with stoichiometric oxidants, as the dehydrogenation is catalytic or uses recoverable sulfur. The solvent systems are amenable to distillation and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The high racemization barrier of the final product ensures long shelf-life and stability during transport, reducing losses due to degradation. This environmental and operational efficiency makes the process highly attractive for large-scale production facilities aiming to meet stringent regulatory standards while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and scalability of these novel spiroalkene derivatives. Understanding these aspects is crucial for integrating these materials into existing OLED fabrication lines and ensuring long-term device performance. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: What is the thermal stability of the synthesized chiral spiroalkene derivatives?
A: The derivatives exhibit exceptional thermal stability with a high racemization energy barrier. Experimental data indicates no racemization occurs even after heating at 300°C for 30 minutes, ensuring reliability in high-temperature device processing.
Q: Can this synthesis method be scaled for industrial production of OLED materials?
A: Yes, the method utilizes commercially available raw materials like arylboronic acids and maleic anhydride, and employs standard catalytic systems (Pd-catalyzed coupling) and solvent systems (toluene, xylene, DMF) that are amenable to large-scale manufacturing.
Q: What are the optical properties of these spiroalkene derivatives?
A: The compounds demonstrate strong Circular Polarization Luminescence (CPL) properties with high dissymmetry factors (glum). They also show good fluorescence emission in the visible range (approx. 540-580 nm), making them suitable for 3D display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiroalkene Derivative Supplier
As the demand for circularly polarized OLED materials surges, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the high-temperature aromatization steps and sensitive chiral resolutions required for these advanced materials, adhering to stringent purity specifications and rigorous QC labs to guarantee >99% ee and optical performance. We understand that consistency is key in the electronics industry, and our integrated supply chain management ensures that raw material fluctuations do not impact your production timelines.
We invite you to collaborate with us to accelerate your product development cycles. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, identifying opportunities to optimize the synthesis of these spiro-compounds. Please contact our technical procurement team today to request specific COA data for our catalog of chiral emitters and to discuss route feasibility assessments for your proprietary derivatives. Let us be your strategic partner in bringing the next generation of 3D display technology to market.
