Advanced Zirconium-Catalyzed Amide Reduction for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Zirconium-Catalyzed Amide Reduction for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for synthesizing critical amine intermediates, which serve as the backbone for countless active pharmaceutical ingredients. Patent CN112299938B introduces a groundbreaking method for preparing amine compounds through the reduction of amide compounds, utilizing a zirconium metal catalyst system that operates under remarkably mild conditions. This innovation addresses the long-standing challenges associated with traditional reduction methods, such as harsh reaction environments and poor functional group tolerance, by leveraging the unique catalytic properties of zirconocene derivatives in conjunction with pinacolborane. The technology enables the selective reduction of the carbon-oxygen double bond in amides to methylene groups at room temperature, eliminating the need for high-pressure hydrogenation equipment or hazardous metal hydrides. For R&D directors and process chemists, this represents a significant leap forward in synthetic methodology, offering a robust route to high-purity amine compounds that can be seamlessly integrated into existing manufacturing workflows. As a reliable pharmaceutical intermediates supplier, understanding and adopting such advanced catalytic systems is crucial for maintaining competitiveness in a rapidly evolving market landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the reduction of amides to amines has relied heavily on stoichiometric metal hydrides such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4), which present significant safety and operational hazards on an industrial scale. These reagents often require strictly anhydrous conditions and low temperatures to control exothermic reactions, leading to complex process engineering requirements and increased operational costs for safety management. Furthermore, catalytic hydrogenation, while atom-economical, typically demands high temperatures and elevated pressures, necessitating specialized reactor infrastructure that many facilities lack. A major drawback of these conventional approaches is their narrow functional group tolerance, where sensitive moieties like nitro, cyano, or ester groups are frequently reduced alongside the amide, resulting in complex impurity profiles that are difficult and expensive to purify. This lack of selectivity often forces manufacturers to employ multi-step protection and deprotection strategies, drastically extending the overall synthesis timeline and reducing the final yield of the target high-purity amine compounds. Consequently, the search for a milder, more selective, and operationally simple reduction method has become a priority for cost reduction in fine chemical manufacturing.
The Novel Approach
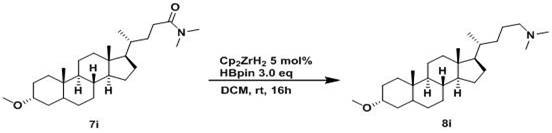
The novel approach detailed in the patent utilizes a zirconium metal catalyst, such as bis(cyclopentadienyl)zirconium dihydride, in combination with pinacolborane to achieve selective amide reduction under ambient conditions. This method operates effectively at room temperature and atmospheric pressure, removing the need for energy-intensive heating or high-pressure hydrogen gas, which fundamentally alters the safety profile and capital expenditure requirements of the process. The catalytic system exhibits exceptional functional group tolerance, allowing for the presence of halogens, esters, and other sensitive functionalities without side reactions, thereby simplifying the purification process and improving the overall purity of the final product. Additionally, the reaction can be performed under neat conditions or in common organic solvents like tetrahydrofuran, providing flexibility in process design that supports commercial scale-up of complex pharmaceutical intermediates. By avoiding the use of stoichiometric amounts of hazardous reducing agents, this technology not only enhances safety but also aligns with green chemistry principles by reducing waste generation. This shift towards catalytic efficiency represents a paradigm change in how amine intermediates are produced, offering a sustainable alternative that meets the rigorous demands of modern supply chains.
Mechanistic Insights into Zirconium-Catalyzed Hydroboration
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the zirconium catalyst, which activates the pinacolborane to selectively reduce the amide carbonyl group. The zirconium center coordinates with the oxygen atom of the amide, lowering the activation energy required for the hydride transfer from the boron species to the carbonyl carbon. This interaction proceeds through a well-defined catalytic cycle that avoids the formation of reactive intermediates capable of attacking other functional groups, ensuring high chemoselectivity throughout the transformation. The use of pinacolborane as the terminal reductant is particularly advantageous due to its stability and ease of handling compared to gaseous hydrogen or pyrophoric hydrides. Understanding this mechanism is vital for process optimization, as it allows chemists to fine-tune catalyst loading and reaction times to maximize conversion while minimizing catalyst residue in the final product. The ability to control the reaction at a molecular level ensures consistent quality across batches, which is a critical parameter for reducing lead time for high-purity amines in a regulated environment.

Impurity control is another critical aspect where this mechanistic understanding provides substantial benefits, particularly in the context of regulatory compliance for pharmaceutical intermediates. Traditional methods often generate inorganic salts and metal residues that require extensive workup procedures, such as aqueous quenching and filtration, which can lead to product loss and environmental burden. In contrast, the zirconium-catalyzed hydroboration pathway generates boron-containing byproducts that are generally easier to separate from the organic amine product, especially when the final product is isolated as a hydrochloride salt. The mild reaction conditions also prevent thermal degradation of the substrate or product, which is a common source of colored impurities and tars in high-temperature processes. By maintaining the integrity of the molecular structure throughout the reduction, the process ensures a cleaner crude reaction mixture, thereby reducing the load on downstream purification units like chromatography or crystallization. This inherent cleanliness of the reaction profile translates directly into higher overall yields and lower production costs, making it an attractive option for large-scale manufacturing.
How to Synthesize Amine Compounds Efficiently
Implementing this synthesis route requires careful attention to the mixing order and atmospheric conditions to ensure optimal catalyst performance and reproducibility. The process begins by establishing an inert atmosphere, typically using nitrogen or argon, to protect the zirconium catalyst from oxidation before the addition of the amide substrate and pinacolborane. Detailed standardized synthesis steps are provided below to guide process engineers in replicating the high yields reported in the patent literature, ensuring that the transition from laboratory scale to pilot plant is smooth and efficient. Adhering to these protocols allows manufacturers to leverage the full potential of this technology, achieving consistent results that meet the stringent quality specifications required by global pharmaceutical clients.
- Mix amide compound, zirconium metal catalyst (e.g., Cp2ZrH2), and pinacolborane in a protective atmosphere.
- Stir the mixture at room temperature for 12 to 48 hours to allow the reduction reaction to proceed completely.
- Perform post-treatment using an ether solution of hydrogen chloride to isolate the amine hydrochloride compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this zirconium-catalyzed reduction technology offers transformative benefits that extend beyond mere chemical efficiency. The elimination of high-pressure equipment and hazardous reagents significantly lowers the barrier to entry for manufacturing these intermediates, allowing for more flexible sourcing strategies and reduced dependency on specialized contract manufacturing organizations. The ability to run reactions at room temperature drastically cuts energy consumption, contributing to substantial cost savings in utility expenses over the lifecycle of the product. Furthermore, the broad substrate scope means that a single catalytic platform can be used to produce a wide variety of amine derivatives, simplifying inventory management and reducing the need for multiple dedicated production lines. This versatility enhances supply chain reliability by mitigating the risk of bottlenecks associated with specific reagent shortages or equipment maintenance downtime. For supply chain heads, this technology represents a strategic asset that improves agility and resilience in the face of market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic method is profound, primarily driven by the removal of expensive and hazardous stoichiometric reducing agents from the bill of materials. Traditional hydride reagents not only carry a high purchase price but also incur significant disposal costs due to their reactive nature and the generation of hazardous waste streams. By utilizing a catalytic amount of zirconium and a stable borane reagent, the material costs per kilogram of product are significantly reduced, improving the overall gross margin of the manufacturing process. Additionally, the simplified workup procedure reduces labor hours and solvent usage, further driving down the variable costs associated with production. These cumulative savings allow companies to offer more competitive pricing to their customers while maintaining healthy profit margins, which is essential for cost reduction in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents and the capacity of high-pressure hydrogenation facilities, both of which are mitigated by this new approach. Pinacolborane is a commercially available reagent with a stable supply chain, and the zirconium catalysts are robust and easy to store, reducing the risk of production delays due to raw material shortages. The mild operating conditions mean that the reaction can be performed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure autoclaves, increasing the number of eligible manufacturing sites. This flexibility allows for better geographic distribution of production capacity, reducing logistics risks and ensuring a steady flow of high-purity amine compounds to downstream customers. Consequently, procurement managers can negotiate better terms and secure longer-term contracts with greater confidence in delivery performance.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and safety, but the room temperature nature of this reaction minimizes these risks significantly. The absence of exothermic spikes associated with hydride additions makes the process inherently safer to scale from grams to tons, facilitating a smoother technology transfer from R&D to commercial production. Moreover, the reduced generation of hazardous waste aligns with increasingly strict environmental regulations, lowering the compliance burden and potential liability for the manufacturing facility. The ability to operate under neat conditions further minimizes solvent emissions, supporting corporate sustainability goals and enhancing the company's reputation as a responsible chemical producer. These factors combined make the commercial scale-up of complex pharmaceutical intermediates not only feasible but also environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zirconium-catalyzed reduction technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent, providing a reliable foundation for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this method into their current production portfolios and evaluate the potential return on investment. Clear communication of these technical advantages is key to building trust with partners and clients who prioritize quality and consistency in their supply chain.
Q: What are the advantages of using zirconium catalysts over traditional hydrides?
A: Zirconium catalysts offer superior functional group tolerance compared to LiAlH4, allowing sensitive groups like esters and nitro groups to remain intact during reduction.
Q: Can this method be performed without organic solvents?
A: Yes, the patent demonstrates that the reaction can proceed under neat conditions, which significantly simplifies downstream processing and reduces solvent waste.
Q: Is this process suitable for large-scale manufacturing?
A: The mild reaction conditions (room temperature, atmospheric pressure) and high yields make this method highly scalable for commercial production of amine intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to deliver superior value to our global partners in the pharmaceutical and agrochemical sectors. Our team of expert chemists has extensively evaluated the zirconium-catalyzed reduction pathway described in CN112299938B and confirmed its viability for producing high-quality amine intermediates at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of amine compounds meets the highest industry standards. By leveraging this advanced chemistry, we can offer our clients a reliable amine compounds supplier partnership that combines technical excellence with commercial reliability.
We invite you to collaborate with us to explore how this innovative reduction technology can optimize your specific synthesis routes and drive down your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, highlighting the specific economic benefits of switching to this catalytic method. Please contact us to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our expertise can accelerate your development timelines. Together, we can build a more efficient and sustainable supply chain for the next generation of life-saving medicines and advanced materials.
