Advanced Synthesis of Alkyl-Substituted α-Methylene-γ-Butyrolactones for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently, particularly those with significant biological potential. Patent CN114957174A introduces a groundbreaking approach for the synthesis of alkyl-substituted α-methylene-γ-butyrolactone derivatives, a structural motif prevalent in numerous natural products exhibiting potent anti-inflammatory, antitumor, and bactericidal activities. This innovation addresses a long-standing challenge in organic synthesis by enabling the direct coupling of phenylpropiolic acid derivatives with unactivated olefins, a transformation that was previously difficult to achieve with high selectivity and yield. By leveraging a synergistic palladium and copper catalytic system, this method operates under remarkably mild conditions, specifically within a temperature range of 20-70°C, and crucially, proceeds effectively under ambient air atmosphere. This represents a paradigm shift from traditional protocols that often demand rigorous exclusion of oxygen and moisture, thereby offering a pathway to high-purity pharmaceutical intermediates with significantly reduced operational overhead.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of functionalized γ-butyrolactone rings has relied heavily on the use of activated olefins, such as styrenes, enol ethers, or electron-deficient alkenes like acrylates, which possess inherent reactivity towards electrophilic metal species. Conventional transition metal-catalyzed cyclizations often suffer from poor substrate universality, failing to engage simple, unactivated aliphatic olefins due to their low nucleophilicity and lack of directing groups. Furthermore, prior art frequently necessitates the use of excessive amounts of additives, such as 12 equivalents of lithium chloride, to drive the reaction equilibrium, which complicates downstream purification and generates substantial inorganic waste. The requirement for inert gas protection (nitrogen or argon) in many existing protocols adds another layer of complexity and cost, making large-scale commercial scale-up of complex polymer additives or drug intermediates less economically viable. These limitations restrict the chemical space accessible to process chemists, forcing reliance on expensive starting materials and multi-step sequences to install the necessary functionality.
The Novel Approach
The methodology disclosed in CN114957174A overcomes these barriers by employing a unique chloropalladation-initiated cascade reaction that activates unactivated olefins directly. As illustrated in the general reaction scheme below, the process couples readily available phenylpropiolic acids with common long-chain alkenes in a single pot, eliminating the need for pre-functionalized substrates. 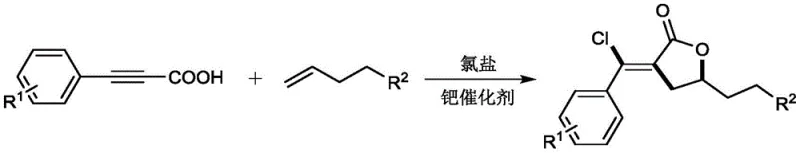 This novel approach utilizes a dual catalytic system involving palladium species and copper salts, which work in concert to facilitate the cyclization and subsequent oxidation steps without the need for external oxidants beyond atmospheric oxygen. The ability to use simple commodity chemicals like pentene or cyclohexyl-propene as starting materials drastically lowers the raw material cost profile. Moreover, the reaction demonstrates exceptional tolerance to various functional groups, allowing for the late-stage diversification of the lactone core, which is invaluable for medicinal chemistry campaigns aiming to optimize cost reduction in fine chemical manufacturing.
This novel approach utilizes a dual catalytic system involving palladium species and copper salts, which work in concert to facilitate the cyclization and subsequent oxidation steps without the need for external oxidants beyond atmospheric oxygen. The ability to use simple commodity chemicals like pentene or cyclohexyl-propene as starting materials drastically lowers the raw material cost profile. Moreover, the reaction demonstrates exceptional tolerance to various functional groups, allowing for the late-stage diversification of the lactone core, which is invaluable for medicinal chemistry campaigns aiming to optimize cost reduction in fine chemical manufacturing.
Mechanistic Insights into Pd/Cu-Catalyzed Chloropalladation Cyclization
The core of this synthetic breakthrough lies in the intricate interplay between the palladium catalyst and the chloride source, which initiates the reaction through a chloropalladation event across the triple bond of the phenylpropiolic acid. This step generates a reactive vinyl-palladium intermediate that is sufficiently electrophilic to intercept the unactivated olefin, a feat rarely achieved in standard cross-coupling scenarios. The presence of copper salts plays a critical role not only as a co-catalyst but potentially as a chloride reservoir and redox mediator, facilitating the turnover of the palladium cycle. The mechanism proceeds through a series of migratory insertions and β-hydride eliminations that are carefully balanced to favor the formation of the five-membered lactone ring over competing polymerization or isomerization pathways. Understanding this mechanistic nuance is vital for R&D Directors looking to adapt this chemistry for specific API intermediates, as it highlights the robustness of the catalytic cycle against common deactivation pathways.
Furthermore, the structural integrity of the final product is maintained through a highly selective intramolecular esterification that locks the conformation into the thermodynamically stable α-methylene-γ-butyrolactone framework. 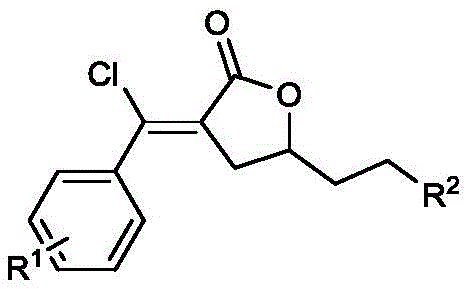 The versatility of this mechanism is evidenced by the wide range of substituents (R1 and R2) that can be accommodated, including electron-withdrawing groups like trifluoromethyl and nitro, as well as bulky groups like tert-butyl. This broad substrate scope ensures that impurity profiles remain manageable, as the reaction does not rely on fragile intermediates that might degrade into complex byproduct mixtures. For quality control teams, this translates to a cleaner crude reaction mixture, simplifying the purification process and ensuring that the final high-purity OLED material or pharmaceutical precursor meets stringent regulatory specifications with minimal effort.
The versatility of this mechanism is evidenced by the wide range of substituents (R1 and R2) that can be accommodated, including electron-withdrawing groups like trifluoromethyl and nitro, as well as bulky groups like tert-butyl. This broad substrate scope ensures that impurity profiles remain manageable, as the reaction does not rely on fragile intermediates that might degrade into complex byproduct mixtures. For quality control teams, this translates to a cleaner crude reaction mixture, simplifying the purification process and ensuring that the final high-purity OLED material or pharmaceutical precursor meets stringent regulatory specifications with minimal effort.
How to Synthesize Alkyl-Substituted α-Methylene-γ-Butyrolactone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the catalytic components, although the procedure is designed to be operationally simple. The patent outlines a straightforward protocol where the substrates are mixed with the catalyst system in a polar aprotic solvent such as acetonitrile or DMF, though greener alternatives like ionic liquids are also viable. The reaction is allowed to proceed with stirring for a duration of 12 to 20 hours, after which standard workup procedures involving concentration and column chromatography yield the pure product. Detailed standardized synthesis steps see the guide below.
- Combine phenylpropiolic acid derivatives and unactivated olefins in a suitable organic solvent such as acetonitrile or toluene within a standard reaction vessel.
- Introduce the catalytic system comprising a palladium source (e.g., PdCl2), a copper salt (e.g., CuCl2), and a chloride source under ambient air conditions without the need for inert gas protection.
- Stir the reaction mixture at mild temperatures ranging from 20°C to 70°C for 12 to 20 hours, followed by concentration and purification via column chromatography to isolate the target lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting the feedstock base from specialized, activated olefins to bulk commodity unactivated alkenes, manufacturers can decouple their production costs from the volatile pricing of fine chemical building blocks. The elimination of inert atmosphere requirements further reduces capital expenditure on specialized reactor infrastructure and lowers utility costs associated with gas purging and monitoring. This process intensification allows for higher throughput in existing facilities, effectively increasing capacity without the need for new construction, which is a critical factor for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the utilization of low-cost, widely available starting materials such as phenylpropiolic acid derivatives and simple aliphatic olefins, which are produced on a massive industrial scale globally. The removal of expensive ligands and the reduction of additive loading (avoiding the 12 equivalents of LiCl seen in prior art) significantly decrease the bill of materials for each batch. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals for the carbon backbone of the molecule mitigates the risk of supply disruptions that often plague the sourcing of exotic or highly functionalized reagents. Since the reaction tolerates air and moisture to a significant degree, the logistical constraints on raw material storage and handling are relaxed, allowing for more flexible inventory management. This robustness ensures a consistent supply of critical intermediates, safeguarding production schedules against the variability often encountered in complex multi-step syntheses dependent on sensitive reagents.
- Scalability and Environmental Compliance: The mild reaction conditions (20-70°C) and the absence of hazardous high-pressure reagents make this process inherently safer and easier to scale from gram to ton quantities. The use of air as the terminal oxidant eliminates the generation of stoichiometric heavy metal waste associated with chemical oxidants, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. This ease of scale-up facilitates the rapid transition from clinical trial material to commercial production, supporting the agile development timelines required by the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived from the detailed experimental data provided in the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of adopting this route for their specific project needs.
Q: What distinguishes this synthesis method from conventional lactone production?
A: Unlike traditional methods that require activated olefins like styrenes or acrylates and harsh conditions, this patented process successfully utilizes inexpensive, unactivated long-chain olefins under mild air conditions, significantly broadening substrate scope and reducing raw material costs.
Q: Does this process require specialized equipment for inert atmospheres?
A: No, a key advantage of this technology is its operation under standard air conditions. This eliminates the need for expensive nitrogen or argon purging systems, simplifying the engineering requirements for commercial scale-up and reducing operational complexity.
Q: What is the functional group tolerance of this catalytic system?
A: The method exhibits exceptional functional group tolerance, accommodating diverse substituents on the aromatic ring including halogens, esters, ketones, and trifluoromethyl groups, making it highly versatile for synthesizing complex pharmaceutical intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl-Substituted α-Methylene-γ-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd/Cu-catalyzed cyclization technology for the development of next-generation therapeutics and functional materials. As a leading CDMO partner, we possess the technical expertise to translate this academic innovation into a robust, GMP-compliant manufacturing process tailored to your specific volume requirements. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of alkyl-substituted α-methylene-γ-butyrolactone delivered meets the highest industry standards for potency and impurity control.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your upcoming projects. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel methodology for your specific application. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can accelerate your path to market while optimizing your overall production costs.
