Revolutionizing Gamma-Gamma-Diaryl Ester Production: A Sustainable Route for Complex Pharmaceutical Intermediates
Revolutionizing Gamma-Gamma-Diaryl Ester Production: A Sustainable Route for Complex Pharmaceutical Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and operationally simple methodologies. A groundbreaking development in this sector is detailed in Chinese Patent CN113387799A, which discloses a novel preparation method for gamma,gamma-diaryl ester and gamma,gamma,gamma-triaryl ester compounds. These structural motifs are ubiquitous in bioactive molecules and serve as critical scaffolds in the construction of advanced pharmaceutical intermediates. The patent introduces a direct cross-coupling strategy that bypasses traditional, hazardous pre-functionalization steps, utilizing readily available aryl-substituted methanols instead of toxic organic halides. This technological leap not only streamlines the synthetic pathway but also aligns perfectly with modern green chemistry principles, offering a compelling value proposition for R&D directors and procurement managers alike who are seeking to optimize their supply chains for high-purity organic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of gamma,gamma-diaryl ester skeletons has relied heavily on the use of organic halides as electrophilic coupling partners. While effective in many contexts, this conventional approach suffers from significant drawbacks that impact both economic efficiency and environmental safety. The synthesis of the requisite organic halides often involves additional synthetic steps starting from alcohols, which are naturally abundant, thereby increasing the overall step count and reducing atom economy. Furthermore, organic halides are inherently toxic and pose serious handling hazards on a commercial scale, necessitating stringent safety protocols and specialized waste disposal procedures. The reliance on these hazardous reagents also introduces potential impurities related to halogenated byproducts, complicating the purification process and potentially affecting the quality of the final active pharmaceutical ingredient (API). For large-scale manufacturers, these factors translate into higher operational costs, longer lead times, and increased regulatory burdens associated with the management of halogenated waste streams.
The Novel Approach
In stark contrast to the legacy methods, the technology described in CN113387799A utilizes aryl-substituted methanols directly as the electrophilic source, effectively eliminating the need for pre-conversion to halides. This direct cross-coupling with cyclopropanol derivatives represents a paradigm shift in synthetic efficiency. By leveraging the inherent reactivity of the hydroxyl group under specific Lewis acidic conditions, the process achieves high conversion rates under remarkably mild conditions, specifically at room temperature. This eliminates the energy-intensive heating or cooling cycles often required in traditional cross-coupling reactions. The simplicity of the post-treatment procedure, involving standard aqueous quenching and extraction, further underscores the practicality of this method for industrial application. The ability to access complex diaryl and triaryl ester structures directly from stable alcohol precursors significantly reduces the material footprint and simplifies the logistical challenges associated with sourcing and storing hazardous halogenated intermediates.
Mechanistic Insights into TiCl4-Catalyzed Cross-Coupling
The success of this transformation hinges on the precise selection of the catalytic system and reaction parameters. Extensive screening revealed that Titanium Tetrachloride (TiCl4) acts as a superior additive compared to other Lewis acids such as Indium Trichloride, Zinc Chloride, or Aluminum halides, which yielded less than 5% of the desired product. The mechanism likely involves the coordination of TiCl4 to the hydroxyl oxygen of the aryl-substituted methanol, enhancing its leaving group ability and generating a reactive carbocation or ion-pair intermediate. Simultaneously, the strained three-membered ring of the cyclopropanol derivative undergoes ring-opening, driven by the relief of ring strain, to form a nucleophilic species that attacks the activated alcohol center. This synergistic activation allows for the formation of the new carbon-carbon bond with high regioselectivity.
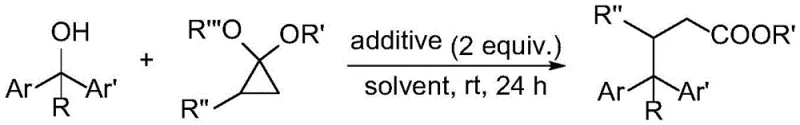
The optimization of reaction stoichiometry is also critical for maximizing yield and minimizing waste. The patent data indicates that a molar ratio of aryl-substituted carbinol to cyclopropanol derivative between 1:2 and 1:2.5 is optimal, with 2.5 equivalents of the cyclopropanol derivative providing the best results. Additionally, the choice of solvent plays a pivotal role; while coordinating solvents like THF and DMF suppress the reaction, non-coordinating solvents like dichloromethane (DCM) facilitate the ionic mechanism effectively, achieving yields up to 60% at room temperature. The tolerance of this system towards various functional groups, including halogens (F, Cl, Br, I) and electron-donating groups (Me, OMe), suggests a robust mechanistic pathway that is not easily perturbed by electronic variations on the aromatic rings, making it highly versatile for diverse substrate scopes.
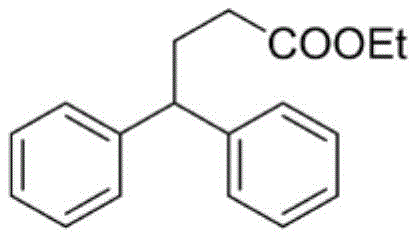
How to Synthesize Ethyl 4,4-Diphenylbutyrate Efficiently
To demonstrate the practical application of this technology, we examine the synthesis of Ethyl 4,4-Diphenylbutyrate, a representative gamma,gamma-diaryl ester. This specific transformation serves as a benchmark for the efficacy of the TiCl4-catalyzed protocol. The process begins with the rigorous exclusion of moisture, utilizing a nitrogen atmosphere and oven-dried glassware to prevent the hydrolysis of the sensitive titanium catalyst. The sequential addition of reagents ensures controlled initiation of the reaction, preventing exotherms that could lead to side products. Following the reaction period, a straightforward workup involving neutralization and extraction allows for the isolation of the crude material, which is then purified to high standards suitable for pharmaceutical applications.
- Under a nitrogen atmosphere, sequentially add aryl-substituted methanol, anhydrous dichloromethane solvent, cyclopropanol derivative (2.5 equiv.), and titanium tetrachloride additive (2.0 equiv.) to a reaction vessel.
- Stir the reaction mixture at room temperature (25°C) for 24 hours to ensure complete conversion of the starting materials into the target ester compound.
- Quench the reaction with saturated sodium bicarbonate solution, extract with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. By utilizing aryl-substituted methanols, which are commodity chemicals often derived from natural sources or bulk petrochemical processes, manufacturers can bypass the volatile market for specialized organic halides. This shift not only stabilizes raw material costs but also mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the elimination of the halide synthesis step removes an entire unit operation from the production schedule, effectively shortening the overall manufacturing lead time and allowing for faster response to market demands for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive and toxic halogenating agents and the associated waste treatment costs. Traditional routes require the conversion of alcohols to halides, a step that consumes additional reagents and generates stoichiometric amounts of salt waste. By skipping this step, the process achieves significant savings in both material costs and environmental compliance expenditures. Moreover, the use of Titanium Tetrachloride, a relatively inexpensive and widely available Lewis acid, ensures that catalyst costs remain low even at large scales. The mild reaction conditions (room temperature) further contribute to cost efficiency by reducing energy consumption for heating or cooling, resulting in a lower overall cost of goods sold (COGS) for the final ester products.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it by relying on robust, commercially available starting materials. Aryl-substituted methanols and cyclopropanol derivatives are stable solids or liquids that can be sourced from multiple global suppliers, reducing dependency on single-source vendors. The operational simplicity of the reaction, which does not require specialized high-pressure equipment or cryogenic conditions, means that it can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant capital investment. This flexibility ensures continuity of supply even in the face of geopolitical or logistical challenges, securing the production pipeline for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen complexities, but this protocol is inherently designed for scalability. The use of dichloromethane as a solvent, while requiring careful management, is a well-understood industrial practice with established recovery and recycling infrastructure. More importantly, the reduction in hazardous waste generation—specifically the avoidance of halogenated byproducts from the coupling step—simplifies the environmental permitting process and lowers the burden on wastewater treatment facilities. The high purity of the crude product, as evidenced by the clean reaction profiles in the patent data, minimizes the need for extensive chromatographic purification, which is often a bottleneck in large-scale production. This combination of safety, efficiency, and environmental stewardship makes the process highly attractive for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this technology for their own production lines.
Q: Why is Titanium Tetrachloride preferred over other Lewis acids for this transformation?
A: According to patent CN113387799A, screening of various additives including indium trichloride, zinc chloride, and aluminum halides resulted in yields below 5%. Titanium tetrachloride uniquely facilitates the activation of the hydroxyl leaving group and the subsequent ring-opening of the cyclopropanol derivative, achieving yields up to 78%.
Q: What are the optimal solvent conditions for scaling up this reaction?
A: While solvents like tetrahydrofuran and acetonitrile resulted in negligible yields (<5%), dichloromethane (DCM) proved to be the optimal medium, providing a 60% yield at room temperature. Non-coordinating solvents like cyclohexane showed moderate activity but DCM remains the standard for highest efficiency.
Q: Can this method tolerate diverse functional groups on the aryl rings?
A: Yes, the methodology demonstrates excellent functional group tolerance. It successfully accommodates substrates with fluoro, chloro, bromo, iodo, methyl, methoxy, and even heterocyclic substituents like furan and thiophene, making it highly versatile for synthesizing complex pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Gamma-Diaryl Ester Supplier
The technological advancements detailed in CN113387799A represent a significant opportunity for the fine chemical industry to produce high-value intermediates more efficiently. At NINGBO INNO PHARMCHEM, we recognize the potential of this TiCl4-catalyzed cross-coupling strategy to redefine the production standards for gamma,gamma-diaryl and gamma,gamma,gamma-triaryl esters. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying the identity and purity of every batch, guaranteeing that our clients receive materials that meet the highest pharmaceutical grade standards.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this innovative synthesis route for their specific projects. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
