Advanced Manufacturing of Hydrogenated Pyridine Derivatives for Anti-Thrombotic Applications
Advanced Manufacturing of Hydrogenated Pyridine Derivatives for Anti-Thrombotic Applications
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for safer and more effective anti-thrombotic agents. Patent CN101343278B introduces a significant advancement in the preparation of hydrogenated pyridine derivatives, specifically 2-acetoxy-5-(alpha-cyclopropyl carbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine and its salts. This compound exhibits potent platelet aggregation inhibitory effects with favorable oral absorption and reduced toxicity profiles compared to earlier generations of drugs. The technical breakthrough lies not just in the molecule itself, but in the robust, industrially viable synthetic pathway that overcomes the limitations of prior art. By establishing a reliable convergent synthesis strategy, this patent provides a foundation for high-purity pharmaceutical intermediate production that meets the stringent quality demands of modern drug development.
![Chemical structure of 2-acetoxy-5-(alpha-cyclopropyl carbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine](/insights/img/hydrogenated-pyridine-derivative-synthesis-pharma-supplier-20260307121450-01.png)
For R&D directors and procurement specialists, the value of this technology extends beyond the chemical structure to the manufacturability of the active ingredient. The patent details a method that simplifies the construction of the complex thienopyridine core while ensuring precise control over the stereochemistry and purity of the final salt form. This is critical for regulatory approval and batch-to-batch consistency. As a reliable pharmaceutical intermediate supplier, understanding these nuanced synthetic improvements allows us to offer superior cost reduction in API manufacturing by minimizing waste and maximizing yield through optimized reaction conditions. The ability to produce this key intermediate at scale positions it as a vital component in the supply chain for next-generation cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of similar thienopyridine derivatives was plagued by inefficiencies that hindered large-scale production. For instance, the methods described in EP-192535 involved an excessive number of synthetic steps, resulting in cumulatively low overall yields and prohibitively high synthetic costs. Such inefficiencies create bottlenecks in the supply chain, making it difficult to secure consistent volumes of high-purity material required for clinical trials and commercial launch. Furthermore, existing techniques often struggled with polymorphism; as noted in EP-542411, obtaining a single crystal form of the additive salt was challenging, leading to potential stability issues and bioavailability variations in the final drug product. These technical hurdles necessitated complex purification protocols and rigorous quality control measures that drove up the cost of goods sold.
The Novel Approach
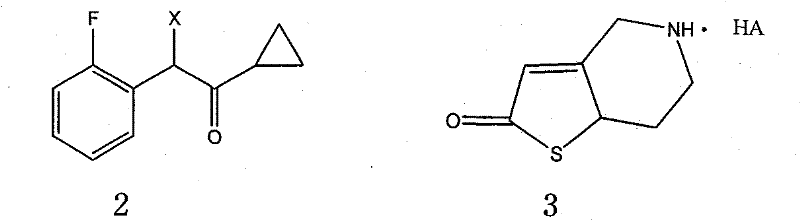
The methodology outlined in CN101343278B represents a paradigm shift towards a more streamlined and economically feasible production process. By decoupling the synthesis into two distinct, highly optimized branches—the preparation of the alpha-cyclopropyl carbonyl-2-fluorobenzyl halide intermediate and the 2-oxo-thienopyridine salt—the process achieves a level of modularity that enhances flexibility and control. This convergent strategy allows for the independent optimization of each fragment before the final coupling, significantly reducing the risk of impurity carryover. The novel approach also incorporates a specific crystallization protocol using seed crystals to guarantee a single crystal form, directly addressing the polymorphic instability of previous methods. This results in a product with superior physical properties and a more predictable performance profile in downstream formulation.
Mechanistic Insights into Convergent Synthesis and Functionalization
The core of this synthetic innovation lies in the meticulous construction of the side chain and the heterocyclic core. The synthesis of the alpha-cyclopropyl carbonyl-2-fluorobenzyl halide begins with a Grignard reaction, where 2-fluorobenzyl bromide reacts with magnesium to form a reactive organometallic species. This intermediate then undergoes nucleophilic addition to cyclopropyl cyanide, a transformation that requires precise temperature control between 0°C and 60°C to prevent side reactions and ensure high conversion. Following hydrolysis, the resulting ketone is subjected to halogenation using reagents like N-bromosuccinimide (NBS) or elemental bromine. This step is critical for activating the benzylic position for the subsequent nucleophilic substitution, and the patent specifies conditions that minimize poly-halogenation, a common impurity in such radical or electrophilic processes.
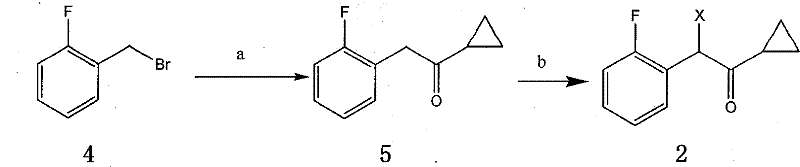
Parallel to the side chain synthesis, the construction of the thienopyridine core involves a sophisticated sequence of protection and oxidation steps. The amine functionality of the tetrahydrothieno[3,2-c]pyridine is first protected, often with bulky groups like trityl or Fmoc, to prevent unwanted alkylation during the oxidation phase. The subsequent oxidation at the 2-position, utilizing n-butyllithium and triethyl orthoformate followed by hydrogen peroxide hydrolysis, installs the crucial ketone functionality. This mechanistic pathway is designed to be highly regioselective, ensuring that the oxygen atom is introduced exclusively at the desired position on the thiophene ring. The final deprotection under acidic conditions reveals the free amine as a stable salt, ready for the convergent coupling step. This level of mechanistic control is essential for maintaining the integrity of the sensitive heterocyclic system throughout the synthesis.
How to Synthesize 2-acetoxy-5-(alpha-cyclopropyl carbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine Efficiently
The efficient synthesis of this complex anti-thrombotic intermediate relies on the seamless integration of the two precursor streams described above. The process begins with the condensation of the halogenated side chain and the thienopyridine salt under alkaline conditions, typically using triethylamine or pyridine as both solvent and acid scavenger. This nucleophilic substitution forms the carbon-nitrogen bond that links the pharmacophore elements. Following isolation, the intermediate undergoes esterification with acetic anhydride or acetyl chloride to install the acetoxy group, a modification that is crucial for the prodrug activity and metabolic stability of the final molecule. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures, are provided in the guide below to ensure reproducibility and safety in a pilot or plant setting.
- Synthesize alpha-cyclopropyl carbonyl-2-fluorobenzyl halide via Grignard reaction of 2-fluorobenzyl bromide with cyclopropyl cyanide, followed by halogenation.
- Prepare 2-oxo-2,4,5,6,7,7a-hexahydrothieno[3,2-c]pyridine salt through amine protection, oxidation substitution, and acidic deprotection.
- Condense the two key intermediates under alkaline conditions, followed by esterification with acetic anhydride and salt formation with acid seeding.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible benefits that extend far beyond simple chemistry. The primary advantage is the substantial cost savings achieved through yield improvement and process simplification. By eliminating the excessive steps associated with legacy methods, the overall material throughput is increased, meaning less raw material is required to produce the same amount of active pharmaceutical ingredient. This efficiency translates directly into a lower cost of goods, allowing for more competitive pricing in the global market. Furthermore, the use of common, commercially available reagents such as magnesium, cyclopropyl cyanide, and acetic anhydride reduces dependency on exotic or supply-constrained catalysts, thereby enhancing supply chain resilience and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive transition metal catalysts and complex purification sequences that were characteristic of older patents. By relying on robust organic transformations like Grignard additions and nucleophilic substitutions, the process utilizes cost-effective reagents that are readily available in bulk quantities. This shift significantly lowers the operational expenditure associated with raw material procurement and waste disposal. Additionally, the higher yields reported in the embodiments mean that the fixed costs of manufacturing—such as energy, labor, and equipment depreciation—are amortized over a larger output of product, driving down the unit cost and improving the overall margin profile for the final drug.
- Enhanced Supply Chain Reliability: A major risk in pharmaceutical manufacturing is the variability of raw material quality and the availability of specialized reagents. This process mitigates those risks by utilizing a chemistry platform based on stable, commodity chemicals. The robustness of the Grignard and halogenation steps ensures that the key intermediate can be produced consistently, regardless of minor fluctuations in feedstock quality. Moreover, the ability to store stable intermediates allows for a flexible production schedule, enabling manufacturers to build inventory buffers against market volatility. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages and ensuring that patient demand is met without interruption.
- Scalability and Environmental Compliance: The reaction conditions specified in the patent, ranging from ambient temperature to moderate heating (up to 100°C), are well within the operating parameters of standard stainless steel reactors found in most multipurpose chemical plants. This compatibility facilitates easy commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment. From an environmental perspective, the process generates fewer byproducts and utilizes solvents that can be efficiently recovered and recycled, such as dichloromethane and ethyl acetate. This alignment with green chemistry principles simplifies regulatory compliance regarding waste discharge and solvent emissions, reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this hydrogenated pyridine derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or long-term supply agreements.
Q: How does this patent improve upon previous methods like EP-192535?
A: Unlike EP-192535 which suffers from low yields and excessive synthetic steps, this method optimizes the route for higher efficiency and lower cost, making it suitable for industrial production.
Q: What is the significance of the crystallization step in this process?
A: The process utilizes specific crystal seeding during salt formation to ensure a single crystal form, addressing the polymorph instability issues found in prior art like EP-542411.
Q: Is this synthesis route scalable for commercial API manufacturing?
A: Yes, the reaction conditions are mild (typically 0°C to 80°C) and utilize common solvents and reagents, facilitating easy scale-up and robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-acetoxy-5-(alpha-cyclopropyl carbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular therapies. Our team of expert chemists has thoroughly analyzed the synthetic pathway described in CN101343278B and is fully prepared to execute this chemistry with precision and scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical supply to full commercialization. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-acetoxy-5-(alpha-cyclopropyl carbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine meets the highest industry standards for identity, strength, and purity.
We invite you to collaborate with us to leverage this advanced technology for your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep process knowledge and supply chain network, allowing you to optimize your manufacturing costs and secure a stable source of critical materials. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market.
