Optimizing Vitamin A Intermediate Production: The DCDMH Chlorination Breakthrough
Optimizing Vitamin A Intermediate Production: The DCDMH Chlorination Breakthrough
The synthesis of Vitamin A esters, such as retinyl acetate and palmitate, relies heavily on the efficiency of producing key C5 building blocks. Patent CN101475471A introduces a transformative methodology for synthesizing 1-chloro-2-methyl-4-alkylacyloxy-2-butylene, a critical intermediate in the C15 + C5 Wittig reaction route. This innovation addresses long-standing inefficiencies in industrial chlorohydrination by replacing traditional sodium hypochlorite systems with a 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) and water reaction matrix. By shifting the chlorinating agent source, the process achieves a dramatic enhancement in product content and yield while simultaneously mitigating the environmental burden associated with saline wastewater and solid slag. For R&D directors and procurement strategists, this patent represents a viable pathway to secure high-purity pharmaceutical intermediates with a significantly optimized cost structure.
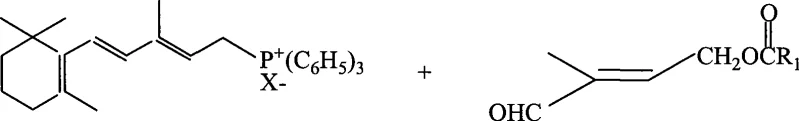
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of this crucial chloride intermediate has relied on the chlorohydrination of isoprene using sodium hypochlorite or calcium hypochlorite. As illustrated in the traditional reaction pathways, these methods suffer from inherent chemical instability and poor selectivity. The presence of free chlorine and various hetero-ions in commercial bleach solutions often triggers unwanted side reactions, such as dichloro-addition across the double bonds, which drastically lowers the purity of the desired 1,4-adduct. Furthermore, the pH control required during these reactions is notoriously difficult to maintain stable, leading to inconsistent batch quality. From a supply chain perspective, the reliance on low-concentration hypochlorite solutions (typically 10-15% available chlorine) necessitates the transport and storage of large volumes of water, increasing logistics costs and generating substantial amounts of salt-containing wastewater that requires expensive treatment before discharge.
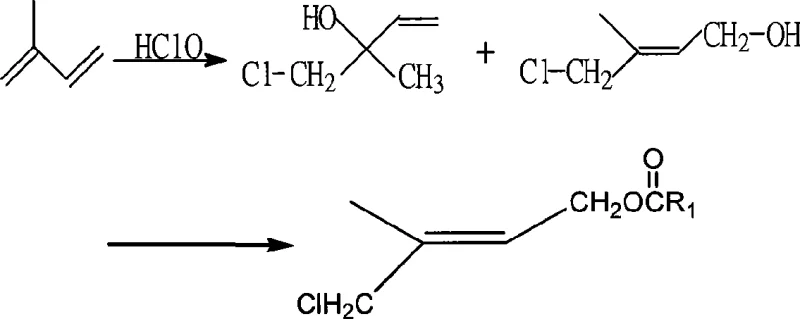
The Novel Approach
The patented methodology fundamentally alters the reaction landscape by utilizing 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) as the primary chlorinating source. This solid reagent possesses an available chlorine content exceeding 70%, which is markedly superior to liquid bleach. When introduced into the aqueous reaction system, DCDMH hydrolyzes to release hypochlorous acid in a controlled, steady manner, effectively eliminating the spikes of free chlorine that cause side reactions. This gentle release mechanism ensures that the electrophilic addition to the isoprene double bond proceeds with high regioselectivity towards the desired 1,4-adduct. Consequently, the process yields a crude product with significantly higher purity and content, reducing the burden on downstream purification steps like rectification. The shift to a solid, high-efficiency reagent also simplifies inventory management and reduces the volumetric footprint of raw material storage facilities.
Mechanistic Insights into DCDMH-Mediated Chlorohydrination
The core chemical advantage of this process lies in the hydrolysis mechanism of the hydantoin derivative. Unlike the chaotic mixture of ions present in bleaching powder or liquid chlorine water, DCDMH reacts with water to stoichiometrically generate two molecules of hypochlorous acid and one molecule of glycolylurea. This reaction is swift yet controllable, ensuring that the concentration of active chlorinating species remains optimal throughout the addition phase without accumulating excess free chlorine that could attack the product. The absence of metal cations (such as sodium or calcium) prevents the formation of insoluble salts and minimizes the ionic strength of the solution, which further suppresses competitive electrophilic attacks on the alkene. This clean reaction profile is critical for maintaining the integrity of the sensitive conjugated diene system found in isoprene derivatives.
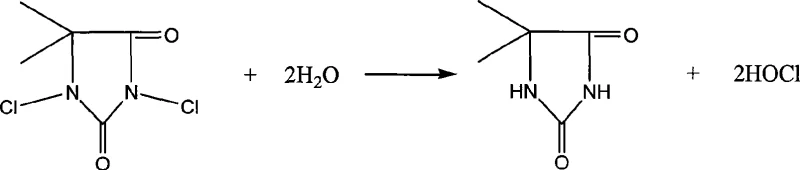
Impurity control is inherently built into this mechanistic design. In conventional processes, the variability of hypochlorite stability often leads to the formation of dichloro-isoprene and other polychlorinated byproducts that are difficult to separate due to similar boiling points. By contrast, the DCDMH system maintains a consistent chemical potential, favoring the formation of the mono-chlorohydrin species. Additionally, the byproduct glycolylurea is chemically distinct and easily separated into the aqueous phase, allowing for potential recovery and recycling. This separation efficiency means that the organic layer obtained after the reaction contains a much higher percentage of the target isomers, directly translating to higher throughput in the subsequent esterification step. For quality assurance teams, this translates to a more robust impurity profile and easier compliance with stringent pharmacopeial standards for Vitamin A precursors.
How to Synthesize 1-chloro-2-methyl-4-alkylacyloxy-2-butylene Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for manufacturing this intermediate. The process begins with the preparation of a heterogeneous mixture containing isoprene, water, and a polymerization inhibitor such as hydroquinone to prevent premature curing of the diene. The DCDMH is then added in batches or continuously while strictly maintaining the reaction temperature between -5°C and 10°C, with 0-5°C being the optimal window for maximizing yield. Following the chlorohydrination, the organic layer is separated and subjected to an esterification rearrangement reaction with an acid anhydride or acid chloride under acid catalysis. This two-stage sequence allows for the direct conversion of the crude chlorohydrin mixture into the final acyloxy product with minimal intermediate isolation, streamlining the overall production timeline.
- Prepare the reaction system by mixing isoprene, water, and a polymerization inhibitor in a reactor equipped with temperature control.
- Gradually add 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) while maintaining the temperature between -5°C and 10°C to ensure controlled release of hypochlorous acid.
- Separate the organic layer containing the chlorohydrin mixture and proceed to esterification with acid anhydride under acid catalysis to form the final alkylacyloxy product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DCDMH-based technology offers compelling economic and operational advantages beyond simple yield improvements. The transition from low-concentration liquid oxidants to high-content solid reagents fundamentally changes the logistics equation, reducing the frequency of raw material deliveries and the associated transportation costs. Moreover, the significant reduction in wastewater salinity and the elimination of calcium slag simplify environmental compliance, potentially lowering the operational expenditure related to waste treatment facilities. These factors combine to create a more resilient and cost-effective supply chain for Vitamin A intermediates, insulating manufacturers from the volatility of bulk chemical markets and regulatory pressures.
- Cost Reduction in Manufacturing: The utilization of DCDMH eliminates the need for expensive pH buffering agents and reduces the consumption of raw materials per unit of output due to higher reaction efficiency. By avoiding the generation of large volumes of saline wastewater, manufacturers can significantly reduce the energy and chemical costs associated with effluent treatment. Furthermore, the ability to recycle the hydantoin byproduct back into the chlorinating agent creates a circular economy within the plant, drastically cutting the net consumption of chlorine sources and driving down the variable cost of goods sold over time.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the inconsistent quality of traditional chlorination processes. This novel method ensures a consistent supply of high-content crude product, reducing the risk of production delays caused by off-spec batches. The stability of the solid DCDMH reagent also allows for longer shelf-life and safer storage compared to unstable hypochlorite solutions, enabling facilities to maintain strategic stockpiles without degradation concerns. This reliability is crucial for meeting the rigorous delivery schedules of downstream pharmaceutical and nutraceutical clients.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, utilizing standard reactor configurations without the need for exotic corrosion-resistant materials often required for handling strong mineral acids or chlorine gas. The reduction in hazardous waste generation aligns with modern green chemistry principles, facilitating smoother permitting processes for capacity expansion. By minimizing the environmental footprint, manufacturers can future-proof their operations against tightening environmental regulations, ensuring long-term business continuity and brand reputation in the global fine chemicals market.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental conditions and beneficial effects detailed in the patent documentation, providing clarity on operational parameters and safety considerations. Understanding these nuances is essential for process engineers evaluating the feasibility of technology transfer.
Q: Why is 1,3-dichloro-5,5-dimethylhydantoin preferred over sodium hypochlorite for this synthesis?
A: DCDMH offers a significantly higher available chlorine content (>70%) compared to industrial bleach (~10-15%) and introduces far fewer hetero-ions, which minimizes side reactions like dichloro-addition and improves overall product purity.
Q: What are the critical temperature controls for the chlorohydrination step?
A: The reaction must be strictly maintained between -5°C and 10°C, ideally 0-5°C. Temperatures above this range promote byproduct formation, while lower temperatures increase refrigeration costs without significant yield benefits.
Q: Can the byproduct hydantoin be recycled in this process?
A: Yes, the hydrolysis byproduct, glycolylurea (hydantoin), can be recovered from the aqueous layer and re-chlorinated to regenerate DCDMH, creating a closed-loop system that drastically reduces solid waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-chloro-2-methyl-4-alkylacyloxy-2-butylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the synthesis of life-saving vitamins and nutraceuticals. Our technical team has extensively analyzed advanced synthetic routes like the DCDMH-mediated chlorohydrination described in CN101475471A to ensure our manufacturing capabilities align with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities or bulk supply, our output meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, guaranteeing that every batch of 1-chloro-2-methyl-4-alkylacyloxy-2-butylene supports your downstream synthesis with maximum efficiency.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while reducing overall production costs.
